Overview
AI-driven medtech trial design employs innovative strategies that enhance clinical research efficiency, reduce costs, and improve patient outcomes through advanced data analytics and predictive modeling. This article underscores the transformative role of AI in optimizing study frameworks and patient recruitment. It also addresses ethical and regulatory challenges, showcasing the potential for AI to revolutionize the Medtech sector.
As we explore the Medtech landscape, it is crucial to recognize how these advancements can address key challenges in clinical research, fostering a collaborative environment for future innovations.
Introduction
The integration of Artificial Intelligence (AI) into the Medtech sector is ushering in a new era for clinical trials, fundamentally transforming traditional methodologies into innovative, data-driven approaches. By harnessing technologies such as machine learning and predictive analytics, researchers can delve into vast datasets to extract insights that were previously unattainable. This capability optimizes trial designs and enhances patient outcomes.
This evolution not only promises to streamline processes and reduce costs but also raises critical questions about the ethical and regulatory implications of AI applications in healthcare. As organizations navigate this complex landscape, understanding the potential benefits and challenges of AI in clinical trials becomes essential for stakeholders aiming to leverage its capabilities effectively.
With a projected surge in AI adoption by 2025, the Medtech industry stands on the brink of a significant transformation that could redefine the future of clinical research.
Understanding AI in Medtech: A New Paradigm for Clinical Trials
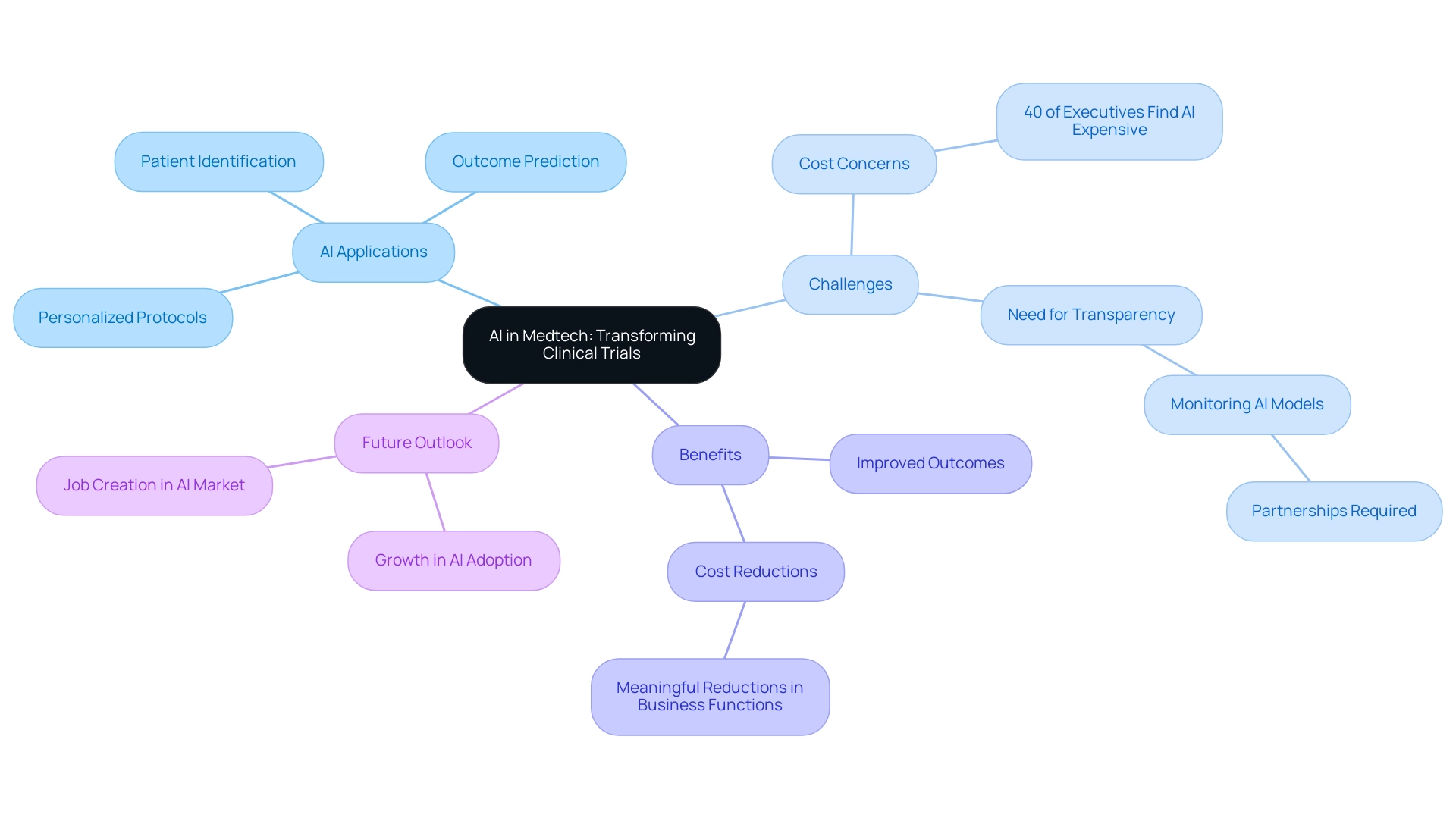
Artificial Intelligence (AI) is fundamentally transforming the Medtech sector by implementing AI-driven trial designs that introduce innovative approaches, significantly enhancing clinical research. This transformation encompasses a range of technologies, including machine learning, natural language processing, and predictive analytics, capable of analyzing extensive datasets to uncover patterns and insights that may elude human researchers. The shift from conventional research methodologies to more adaptive and efficient AI-driven trial designs enables researchers to optimize study frameworks and improve patient outcomes.
For instance, AI plays a crucial role in:
- Identifying suitable patient populations
- Predicting study outcomes
- Personalizing treatment protocols through real-time data analysis
Recent advancements in AI have demonstrated its potential to reduce costs and streamline processes, with the majority of executives acknowledging meaningful cost reductions from generative AI applications across various business functions. However, it is important to note that 40% of executives perceive AI as prohibitively expensive, highlighting a significant challenge in its adoption.
Moreover, the incorporation of AI in medical studies presents its own difficulties. Experts emphasize the necessity for transparency in AI model performance and ongoing monitoring post-implementation. As Brian Anderson, CEO of the nonprofit Coalition for Health AI, states, "Monitoring these models is not something you can do on your own, and you need that partnership."
Establishing strong vendor partnerships and utilizing tools like model cards can enhance procurement processes and ensure sustained performance, as highlighted by healthcare professionals in the case study titled "Transparency and Monitoring of AI Tools."
Looking ahead to 2025, the adoption of AI technologies in the Medtech sector is expected to grow significantly, with many organizations recognizing the need for effective implementation strategies. Successful case studies illustrate how AI-driven trial designs can lead to more efficient studies, ultimately benefiting both researchers and patients alike. Furthermore, the extensive medical study management services provided by bioaccess®, including feasibility assessments, site selection, compliance evaluations, and project oversight, are essential in navigating the complexities of research studies.
With over 20 years of experience in Medtech, bioaccess® is well-equipped to address the challenges faced by medical device startups, such as regulatory hurdles and competition. The AI market is projected to create more startups and jobs than it destroys, indicating a promising future for AI in Medtech. Understanding these foundational concepts of AI is crucial for stakeholders aiming to effectively harness its capabilities in the evolving landscape of Medtech, while also acknowledging the positive impact of research studies on local economies through job creation and healthcare enhancement.
The Transformative Benefits of AI in Clinical Trial Design
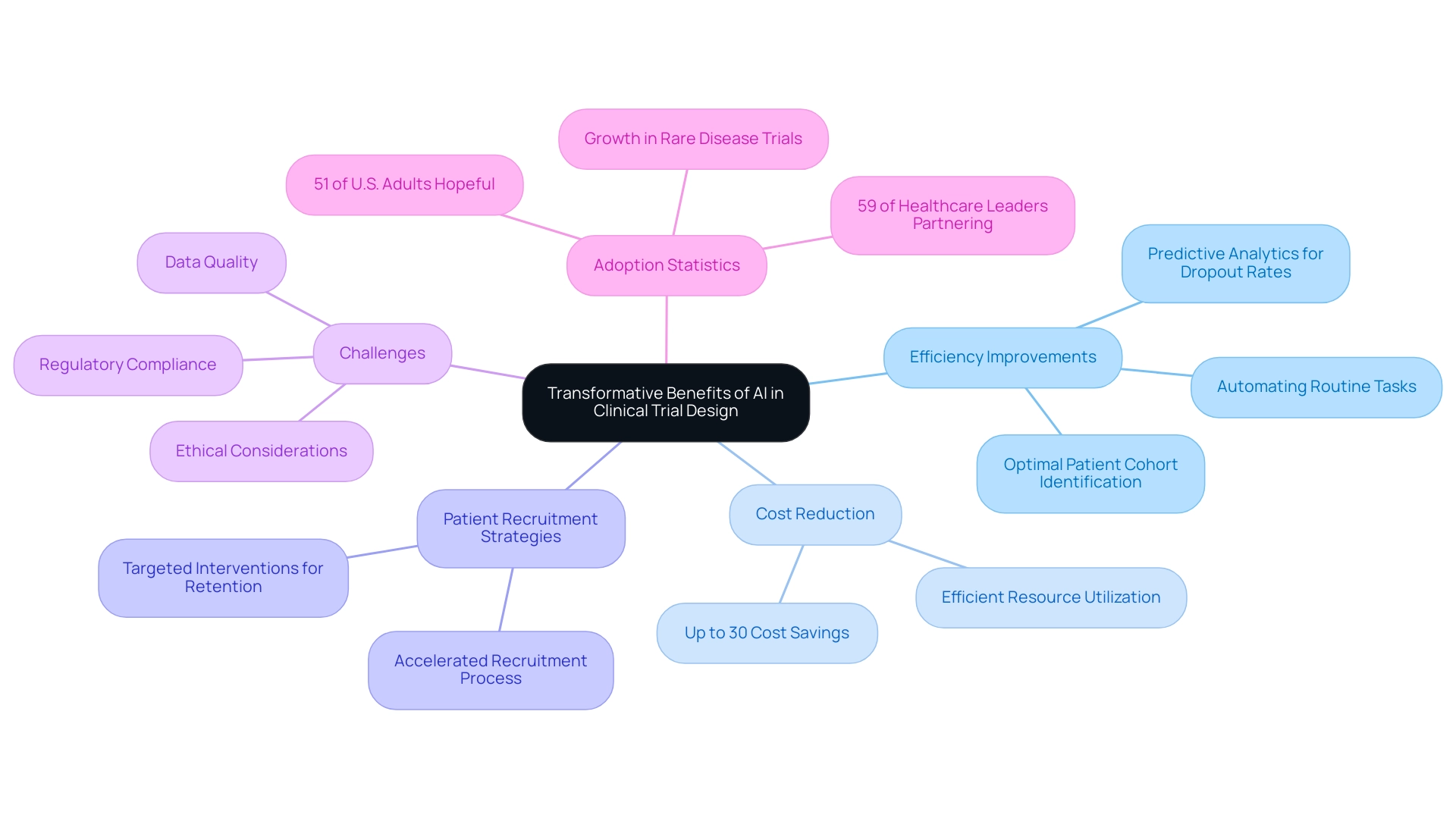
AI is revolutionizing clinical study design through AI-driven medtech trial design, delivering transformative benefits that enhance efficiency, reduce costs, and improve patient recruitment strategies. By automating routine tasks such as information gathering and analysis, AI enables researchers to focus on the more complex elements of management. For instance, advanced AI algorithms can sift through historical data to pinpoint optimal patient cohorts, significantly accelerating the recruitment process.
This capability is especially vital in the context of rare disease studies, where identifying suitable participants can be challenging. In fact, the potential for greater adoption and growth of AI applications in rare disease studies is noteworthy. Moreover, AI's predictive analytics can forecast patient dropout rates, enabling researchers to implement targeted interventions that enhance retention. This proactive method not only results in more robust outcomes but also aids in a more efficient use of resources. Research suggests that AI-driven medtech trial design can realize cost savings of up to 30%, making it an appealing choice for Medtech firms aiming to optimize their operations.
In Latin America, bioaccess® provides extensive management services for studies that encompass feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting. These services are essential for guaranteeing that research studies not only fulfill regulatory standards but also positively impact local economies through job creation and healthcare enhancements. The incorporation of AI-driven medtech trial design into these processes improves the overall efficiency of medical studies, aligning with the area's objective of changing lives through advanced Medtech solutions.
However, the incorporation of AI into research processes is not without its challenges. The need for high-quality data, regulatory compliance, and ethical considerations regarding patient consent and data privacy must be addressed to ensure successful implementation. As healthcare leaders increasingly acknowledge the potential of AI, with 51% of U.S. adults hopeful that new applications of AI will result in significant progress in healthcare in 2024, the momentum for AI adoption in medical studies is poised to expand.
Significantly, 59% of healthcare leaders utilizing generative AI are collaborating with external vendors to create tailored solutions, highlighting the trend of partnership in AI adoption. Moreover, Medidata AI offers solutions to expedite drug development and enhance patient outcomes in research studies, consistent with the advantages mentioned. This trend underscores the importance of leveraging AI as a vital tool for Medtech companies aiming to expedite the introduction of innovative solutions to the market.
Integrating AI: Methodologies and Frameworks for Effective Trial Design
Incorporating AI-driven medtech trial design into clinical research planning necessitates a systematic approach that integrates various methodologies and frameworks. A notably successful strategy is the application of adaptive study frameworks, which allow for adjustments based on interim outcomes. This inherent flexibility is significantly enhanced through AI-driven medtech trial design, which forecasts outcomes across diverse scenarios, empowering researchers to make data-informed decisions throughout the experimental process.
Moreover, the utilization of machine learning algorithms to analyze real-time data can unveil trends and anomalies, enabling timely adjustments to study protocols. For instance, a study monitoring the usage trends of adaptive approaches from 2006 to 2021 revealed a consistent increase in the acceptance of these methodologies, particularly in oncology research, with a significant spike noted in 2020. This trend is further underscored by the identification of 36 infectious disease research studies, 16 of which focused specifically on COVID-19 vaccines, illustrating the critical role of adaptive approaches in addressing urgent health challenges.
In this context, comprehensive research management services, such as those offered by bioaccess, are pivotal. Their capabilities encompass feasibility studies, site selection, compliance reviews, experiment setup, import permits, project management, and reporting. These services ensure that all elements of the study are optimized for success, facilitating the integration of AI and flexible frameworks into research studies.
Examples of successful adaptive frameworks, including the CARISA study, Tailor study, and a study on acute myeloid leukemia, demonstrate the tangible benefits of these methodologies in medical research. Frameworks like the AI-Driven Clinical Study Framework (AI-CTF) serve as comprehensive guides for researchers, aiding in the seamless incorporation of AI, ultimately enhancing the efficiency and effectiveness of studies. As the landscape of medical research evolves, the successful integration of AI-driven medtech trial design methodologies will be crucial in advancing healthcare technology and improving patient outcomes.
Furthermore, the need for guidelines on creating and communicating with external adaptation committees to oversee study modifications is essential, as it highlights the importance of structured oversight in adaptive study frameworks. As noted by Salaheddin M Mahmud, a specialist in the field, the incorporation of adaptive designs and AI can significantly enhance the quality and responsiveness of medical studies.
Data Management and Analytics: The Backbone of AI-Driven Trials
Data management and analytics are fundamental to the success of AI-driven medtech trial design, serving as the backbone that supports the entire research process. High-quality information is crucial for training AI models, which in turn ensures accurate predictions and outcomes. To maintain information integrity, researchers must establish robust governance frameworks that encompass comprehensive protocols for collection, storage, and processing. This structured approach not only safeguards data quality but also enhances compliance with regulatory standards.
At bioaccess, our extensive research study management services encompass feasibility assessments, site selection, compliance evaluations, setup, import permits, project management, and reporting on serious and non-serious adverse events. These services are designed to streamline the clinical study process, ensuring that all aspects are meticulously managed to foster successful outcomes.
The adoption of cloud-based platforms is becoming increasingly essential for AI-driven medtech trial design, as they enable real-time information sharing and collaboration among all parties involved in the experiment. This smooth merging of information sources greatly enhances the overall effectiveness of experiment operations, enabling faster decision-making and flexible experiment designs. Significantly, the AI-driven medtech trial design can facilitate the integration of information from various sources in clinical study management, ensuring smooth information flow and consistency.
Moreover, advanced analytics tools play a pivotal role in extracting actionable insights from the data. These tools can examine patient demographics, treatment responses, and potential adverse events, enabling researchers to make informed, data-driven decisions that improve outcomes. For example, the application of AI-driven medtech trial design and machine learning technologies has been demonstrated to enhance research processes, as illustrated by Saama Technologies' platform, which automates resource-intensive steps and boosts productivity. Saama's platform has shown the capability to save considerable time in query identification and information transformation, thus enhancing the efficiency of research studies.
As conventional medical studies encounter an average 30% dropout rate due to inconvenience, intricate protocols, and insufficient support, prioritizing efficient information management and analytics becomes vital. As Guillaume Gigon, VP of Technology and AI, states, "Clinical studies are becoming progressively intricate and information-heavy, and CROs must navigate an environment that requires efficiency, transparency, and high-quality information." By fully utilizing the capabilities of AI and ensuring immediate access to information, bioaccess can lead to more successful clinical studies, ultimately contributing to job creation, economic growth, healthcare enhancement, and international cooperation in the Medtech sector.
Navigating Ethical and Regulatory Challenges in AI-Driven Trials
Navigating the ethical and regulatory challenges of AI-driven medtech trial design is essential for maintaining public trust and ensuring compliance with established guidelines. Key factors include patient privacy, informed consent, and robust security measures. At bioaccess®, we are committed to safeguarding information security and fostering client trust through our comprehensive protection procedures.
Clients are encouraged to contact our Grievance Officer at IMH ASSETS CORP (doing business as "bioaccess®"), located at 1200 Brickell Avenue, Suite 1950 #1034, via email at info@bioaccessla.com, for any concerns regarding their information. This ensures transparency and compliance with applicable laws.
Researchers must implement transparent processes that empower participants to understand how their information will be utilized, ensuring that AI algorithms are free from bias. Recent statistics reveal that ethical challenges in AI-driven medical studies are under increasing scrutiny, exemplified by a notable case where a hospital in Portugal faced a €400,000 fine for data protection breaches. This underscores the critical importance of strict adherence to privacy laws.
Moreover, staying informed about evolving regulatory frameworks, such as the FDA's guidance on decision support software, is vital for ensuring that AI applications meet safety and efficacy standards. Engaging regulatory bodies early in the study planning phase can facilitate smoother approvals and promote collaboration between researchers and regulators. This proactive approach not only enhances the credibility of AI-driven medical studies but also addresses the ethical challenges that may arise, ensuring that AI technologies are applied fairly and beneficially across diverse healthcare settings.
The case study titled "Global Implications of AI in Healthcare" illustrates the potential of AI to democratize healthcare, particularly in low-resource environments, while also highlighting the risks of exacerbating existing inequalities if AI solutions are not adapted to varied contexts. As one participant noted, "If the AI is efficacious, it’s an accurate diagnostic, but is this cost-effective if it costs a million dollars to run?" This raises significant economic considerations that must be addressed in the design of AI-driven medtech trials.
As the landscape of medical research continues to evolve, prioritizing these ethical considerations, alongside public education on data security and ethical AI usage, will be crucial for the successful integration of AI in Medtech. At bioaccess®, our extensive research management services encompass feasibility studies, site selection, compliance reviews, setup, import permits, project oversight, and reporting, all designed to assist researchers in navigating these complexities effectively.
Collaboration and Stakeholder Engagement: Keys to Successful AI Implementation
Collaboration and stakeholder involvement are paramount for the successful execution of AI-driven medtech trial design in clinical studies within the Medtech sector. Establishing robust collaborations among researchers, technology suppliers, regulatory agencies, and patient advocacy organizations fosters innovative solutions that significantly enhance study outcomes. Effective communication and continuous feedback cycles among stakeholders are essential, ensuring that diverse perspectives are integrated into the project development process.
This collaborative environment not only promotes knowledge sharing but also facilitates problem-solving, which is vital for navigating the complexities of clinical research.
Engaging patients early in the study design can yield invaluable insights into their needs and preferences, ultimately leading to more patient-centered research. Statistics reveal that studies characterized by strong stakeholder engagement enjoy higher success rates, underscoring the critical role of collaboration in achieving favorable outcomes. Notably, only 6% of experiments specified the type of information accessed, highlighting a significant gap in clarity that can be addressed through effective stakeholder engagement.
A recent study highlighted that experiments marked by active collaboration between academia and industry reported enhanced transparency and reliability in results, which is essential for maintaining public trust in clinical research. As one Lead Academic Author noted, "I had access to all the data but did not have access to the database. Any analysis that we desired was performed in the sponsor's dataset," illustrating the challenges faced in study planning and the crucial need for cooperation.
Furthermore, as the landscape of medical studies evolves, particularly with the integration of AI technologies, the significance of collaboration becomes increasingly apparent. Effectively engaging stakeholders not only improves the design and execution of studies but also ensures that the innovations developed align with the real-world needs of patients and healthcare providers. The partnership between bioaccess™ and Caribbean Health Group to establish Barranquilla as a premier research location in Latin America exemplifies this approach, supported by Colombia's Minister of Health.
Additionally, collaborations with IDx Technologies and GlobalCare Clinical Trials further demonstrate how bioaccess™ is advancing global health improvement through international collaboration and innovation in Medtech.
Moreover, bioaccess™ offers comprehensive research study management services, including feasibility assessments, site selection, compliance evaluations, study setup, import permits, project management, and reporting. These capabilities are crucial for ensuring that research studies are conducted efficiently and effectively, ultimately contributing to job creation and economic development in local communities. The issue of ghost authorship in industry-sponsored studies, where key contributors remain unrecognized, further underscores the need for transparency and accountability in research.
By emphasizing these collaborative initiatives, Medtech firms can leverage the collective expertise of various stakeholders, driving the effective integration of AI-driven medtech trial design in healthcare studies and ultimately advancing the field of medical technology.
Future Trends in AI-Driven Medtech Trials: Innovations on the Horizon
The future of AI-driven medtech trial design is poised for remarkable transformations, propelled by several emerging trends. A significant advancement is the rise of decentralized clinical studies, which leverage digital health technologies to enhance remote patient monitoring and streamline data collection. This innovative approach, utilizing AI-driven medtech trial design, not only boosts patient engagement but also broadens access to diverse populations, thereby improving the generalizability of study outcomes.
Furthermore, the incorporation of generative AI and advanced analytics will elevate AI-driven medtech trial design by facilitating sophisticated simulations and predictive modeling. This empowers researchers to refine study designs prior to execution. As AI technologies advance, Medtech firms must remain agile and adaptable, embracing these innovations to maintain a competitive edge in the evolving research landscape. With over 20 years of experience in the Medtech sector, bioaccess® recognizes the critical importance of these advancements.
By 2025, it is anticipated that 45% of medical data will be recorded on the same day as patient visits, underscoring the significance of real-time data management in enhancing study efficiency and precision. bioaccess® specializes in comprehensive trial management services, including:
- feasibility studies
- site selection
- compliance reviews
- trial setup
- import permits
- project management
- reporting
ensuring that trials are executed smoothly and effectively. However, as Max Baumann from Tree Hill Partners warns, the medical markets are becoming increasingly crowded, presenting fundamental business model challenges for biotech companies.
Embracing these trends will be essential for achieving commercial success alongside regulatory approval, particularly as the research environment grows more complex with the introduction of AI-driven medtech trial design. As Joseph Tucker, CEO of Enveric, observes, the regulatory landscape is evolving, and companies must navigate these challenges meticulously to thrive. Moreover, the shift towards greater ownership and transparency, as highlighted in recent case studies, emphasizes the necessity for sponsors to have direct access to real-time information, thereby enhancing quality and control over clinical outcomes.
This focus on data management will be vital for optimizing trial designs and ensuring successful outcomes, especially in the context of bioaccess®'s expertise in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies.
Conclusion
The integration of Artificial Intelligence in the Medtech sector is fundamentally reshaping clinical trials, transforming traditional methodologies into innovative, data-driven approaches. By utilizing technologies such as machine learning and predictive analytics, researchers can optimize trial designs and enhance patient outcomes, all while achieving significant cost reductions. However, as AI adoption continues to grow, stakeholders must proactively address the ethical and regulatory challenges that accompany this evolution.
Collaboration among researchers, technology providers, and patient advocacy groups is essential for improving trial outcomes. Engaging patients early in the design process ensures that research aligns with their needs, resulting in more patient-centered studies. Furthermore, maintaining high data quality and transparency is crucial for building public trust and meeting regulatory standards.
As the industry anticipates advancements such as decentralized trials and real-time data management, agility and responsiveness will be paramount. The future of AI in clinical trials holds immense potential for increased efficiency and improved treatment protocols. By embracing these innovations and fostering a collaborative environment, the Medtech sector can fully harness AI's capabilities, driving advancements that benefit both researchers and patients.
In summary, the successful integration of AI in clinical trials hinges on ethical considerations, transparency, and stakeholder engagement, ensuring that these technologies lead to equitable and impactful healthcare solutions.
Frequently Asked Questions
How is AI transforming the Medtech sector?
AI is fundamentally transforming the Medtech sector by implementing AI-driven trial designs that enhance clinical research through innovative approaches, utilizing technologies like machine learning, natural language processing, and predictive analytics to analyze extensive datasets.
What are some applications of AI in clinical research?
AI plays a crucial role in identifying suitable patient populations, predicting study outcomes, and personalizing treatment protocols through real-time data analysis.
What benefits does AI offer in medtech trial design?
AI-driven medtech trial design enhances efficiency, reduces costs, and improves patient recruitment strategies, with potential cost savings of up to 30%.
What challenges do organizations face in adopting AI?
Organizations face challenges such as high costs, the need for transparency in AI model performance, regulatory compliance, and ethical considerations regarding patient consent and data privacy.
How does AI improve patient recruitment in clinical studies?
Advanced AI algorithms can sift through historical data to identify optimal patient cohorts, significantly accelerating the recruitment process, especially in studies involving rare diseases.
What is the role of partnerships in AI adoption in healthcare?
Many healthcare leaders are collaborating with external vendors to create tailored AI solutions, highlighting the importance of partnerships in the successful adoption of AI technologies.
What services does bioaccess® provide in Latin America?
bioaccess® offers extensive management services for studies, including feasibility assessments, site selection, compliance evaluations, project oversight, and reporting, ensuring that research studies meet regulatory standards and positively impact local economies.
What is the future outlook for AI in Medtech?
The adoption of AI technologies in the Medtech sector is expected to grow significantly, with many organizations recognizing the need for effective implementation strategies to enhance research efficiency and patient outcomes.
What is the significance of monitoring AI models post-implementation?
Ongoing monitoring of AI models is essential to ensure their performance and effectiveness, as indicated by experts in the field, emphasizing the need for partnerships in this process.
How does AI contribute to local economies through research studies?
Research studies incorporating AI can create jobs and enhance healthcare, positively impacting local economies as organizations implement advanced Medtech solutions.

