Overview
The article outlines best practices for clinical trial strategies specific to Brazil, underscoring the importance of understanding the local regulatory environment, cultural nuances, and effective recruitment techniques. It supports these insights by detailing the roles of regulatory bodies such as ANVISA and CONEP, emphasizing the necessity for community engagement, and highlighting the benefits of leveraging technology. All these factors significantly enhance the likelihood of successful clinical trials in the country.
Introduction
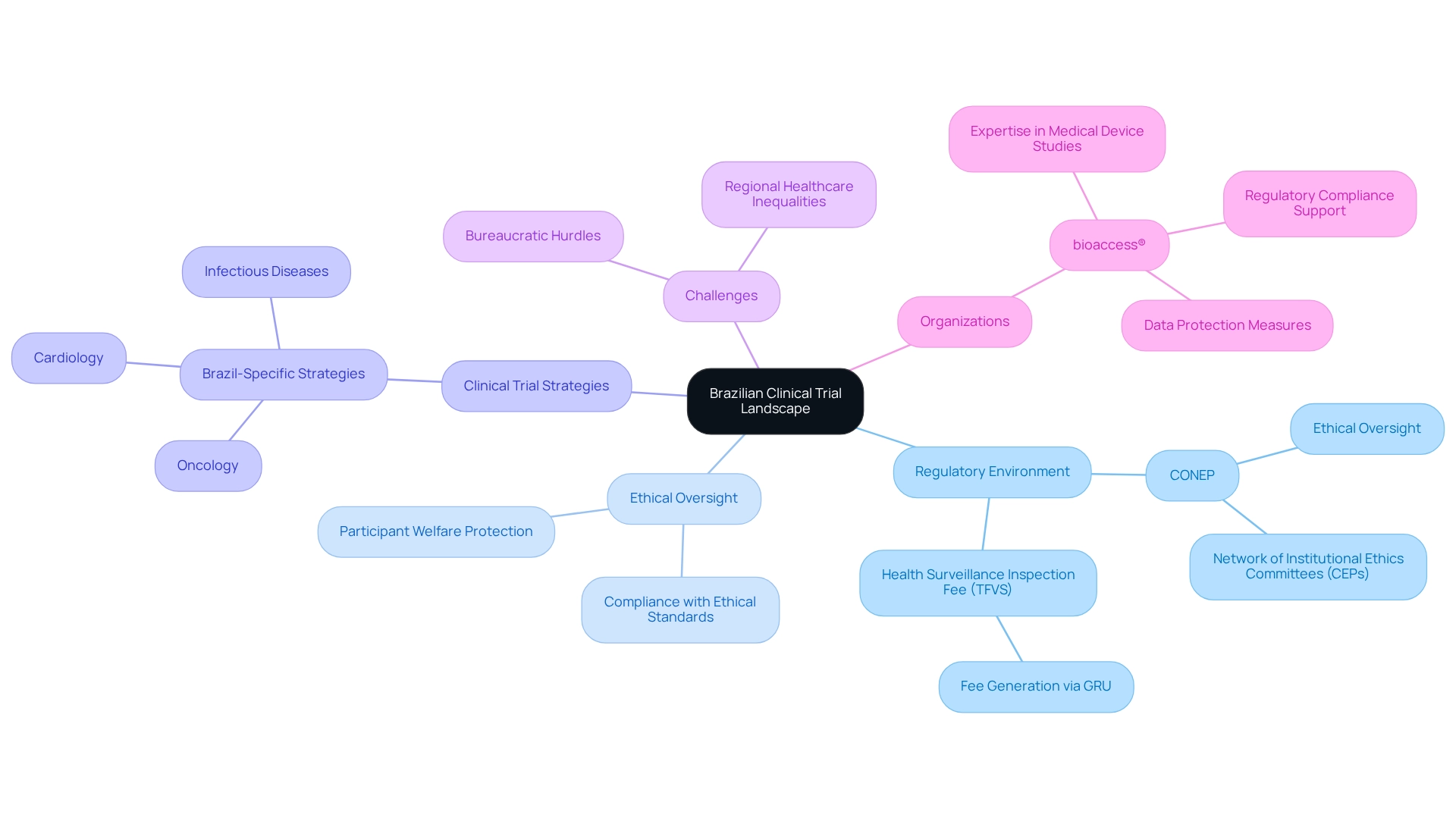
Brazil's clinical trial landscape stands as a vibrant tapestry, intricately woven from its rich diversity and complex regulatory environment, establishing it as a pivotal hub for medical research in Latin America. With nearly 10,000 clinical studies registered as of early 2024, the country demonstrates a robust commitment to advancing healthcare through research.
However, navigating this landscape presents challenges, from bureaucratic hurdles to regional disparities in healthcare capabilities. The National Research Ethics Commission (CONEP) plays a crucial role in ensuring ethical oversight, while organizations like bioaccess® are stepping up to streamline processes and enhance compliance. This proactive approach ultimately drives innovation in critical therapeutic areas such as oncology and cardiology.
This article delves into the intricacies of conducting clinical trials in Brazil, exploring effective recruitment strategies, cultural considerations, and the vital role of local partnerships in fostering successful research outcomes.
Understanding the Brazilian Clinical Trial Landscape
Brazil's experimental landscape is marked by its diverse population and a dynamic regulatory environment, establishing the nation as a pivotal player in Latin America for medical studies, particularly through Brazil-specific clinical trial strategies. As of early 2024, around 10,000 medical studies have been registered, underscoring a strong commitment to advancing medical research. The Brazilian healthcare system, encompassing both public and private sectors, presents a promising backdrop for research studies, especially when leveraging Brazil-specific clinical trial strategies in critical therapeutic areas such as oncology, cardiology, and infectious diseases.
However, navigating this landscape requires acute awareness of various challenges, including bureaucratic hurdles and Brazil-specific clinical trial strategies aimed at addressing inequalities in regional healthcare capabilities. These elements can significantly influence recruitment strategies, data collection processes, and the overall execution of Brazil-specific clinical trial strategies in medical studies. For instance, the Health Surveillance Inspection Fee (TFVS) serves as a crucial mechanism for generating charges related to health monitoring, adding an extra layer of regulatory consideration for sponsors.
The National Research Ethics Commission (CONEP) plays a vital role in overseeing the ethical dimensions of studies involving human participants, ensuring compliance with established ethical standards and safeguarding participant welfare. CONEP's framework not only protects participants but also enhances the credibility of clinical studies conducted in Brazil, as it orchestrates a network of institutional ethics committees (CEPs) that evaluate study protocols. Moreover, recent developments in Brazil's clinical study environment reflect a growing emphasis on compliance and ethical oversight, which are essential for the integrity of Brazil-specific clinical trial strategies.
Organizations like bioaccess®, Latin America's foremost contract research organization (CRO), utilize their expertise and tailored approach to navigate these complexities, facilitating the expedited advancement of medical devices. bioaccess® offers a comprehensive process for optimizing medical device studies, covering study set-up, start-up, approval, site feasibility, investigator selection, regulatory compliance, project management, and reporting. By understanding the regulatory framework and ethical context, bioaccess® refines Brazil-specific clinical trial strategies for conducting research studies in South America, thereby enhancing the likelihood of successful outcomes while ensuring information security and client trust through robust data protection measures.
For any inquiries or concerns regarding data protection, clients are encouraged to contact bioaccess®'s Grievance Officer. To explore how bioaccess® can support your study requirements, SCHEDULE A MEETING.
Navigating Brazil's Regulatory Framework for Clinical Trials
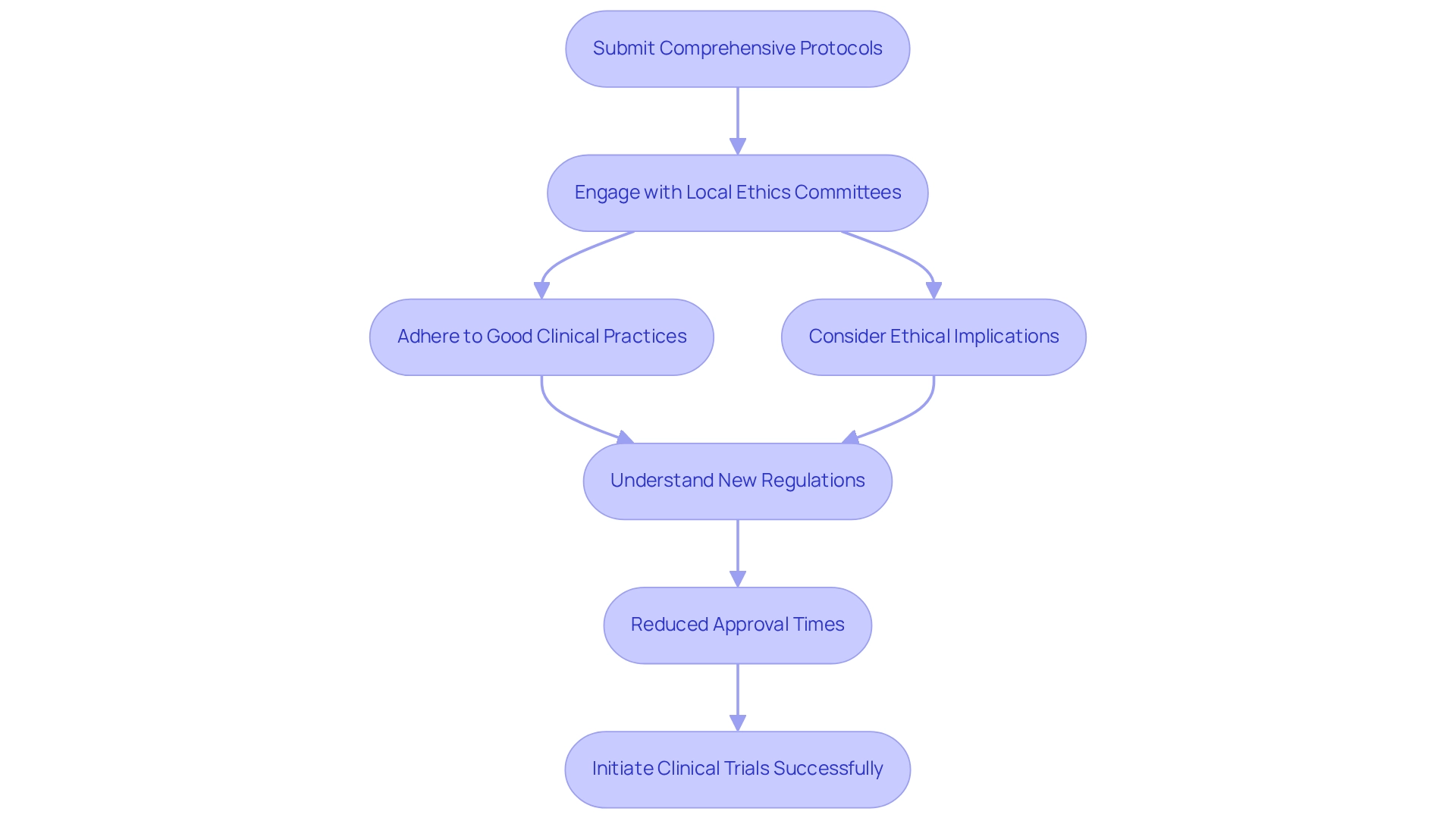
The Brazilian National Health Surveillance Agency (ANVISA) serves as the cornerstone of medical study regulation in the country. With the introduction of Resolution No. 945/2024 in January 2025, the approval process for research studies has been significantly streamlined, thereby enhancing efficiency for researchers.
To navigate this landscape successfully, it is imperative that researchers submit comprehensive protocols that adhere to Good Clinical Practices (GCP) and address all ethical considerations. Early engagement with local ethics committees is crucial, as their input plays a vital role in the approval process. Familiarity with these regulatory intricacies not only facilitates prompt trial initiation but also ensures the successful implementation of studies employing Brazil-specific clinical trial strategies.
Moreover, understanding the implications of the new regulations can lead to reduced approval times, ultimately accelerating the transition from research to market for innovative medical technologies through the application of Brazil-specific clinical trial strategies. It is essential to recognize that the withdrawal of consent is valid from the date that the decision is communicated, underscoring the significance of participant rights. Furthermore, research studies involving women of childbearing age or expectant mothers require additional safeguards to ensure participants are fully informed of potential risks, as emphasized in the relevant case study.
Investigators must prioritize the rights of female participants, ensuring they can engage in studies without being compelled to use contraceptives unless pregnancy is the study's primary focus. As Santiago Pena, President of Paraguay, has noted, the independence and institutional strengthening of DINAVISA is crucial for the continued development of the pharmaceutical sector, further highlighting the necessity of a robust regulatory environment. At bioaccess®, we leverage over 20 years of experience in overseeing research projects throughout Latin America, encompassing Early-Feasibility Studies, First-In-Human Studies, and Post-Market Follow-Up Studies, to ensure compliance and success in this evolving landscape.
Our accelerated medical device research study services are specifically designed to assist clients in navigating these regulatory changes effectively, while our flexible and customized approach guarantees that we meet the unique needs of each study.
Effective Recruitment Strategies for Brazilian Clinical Trials
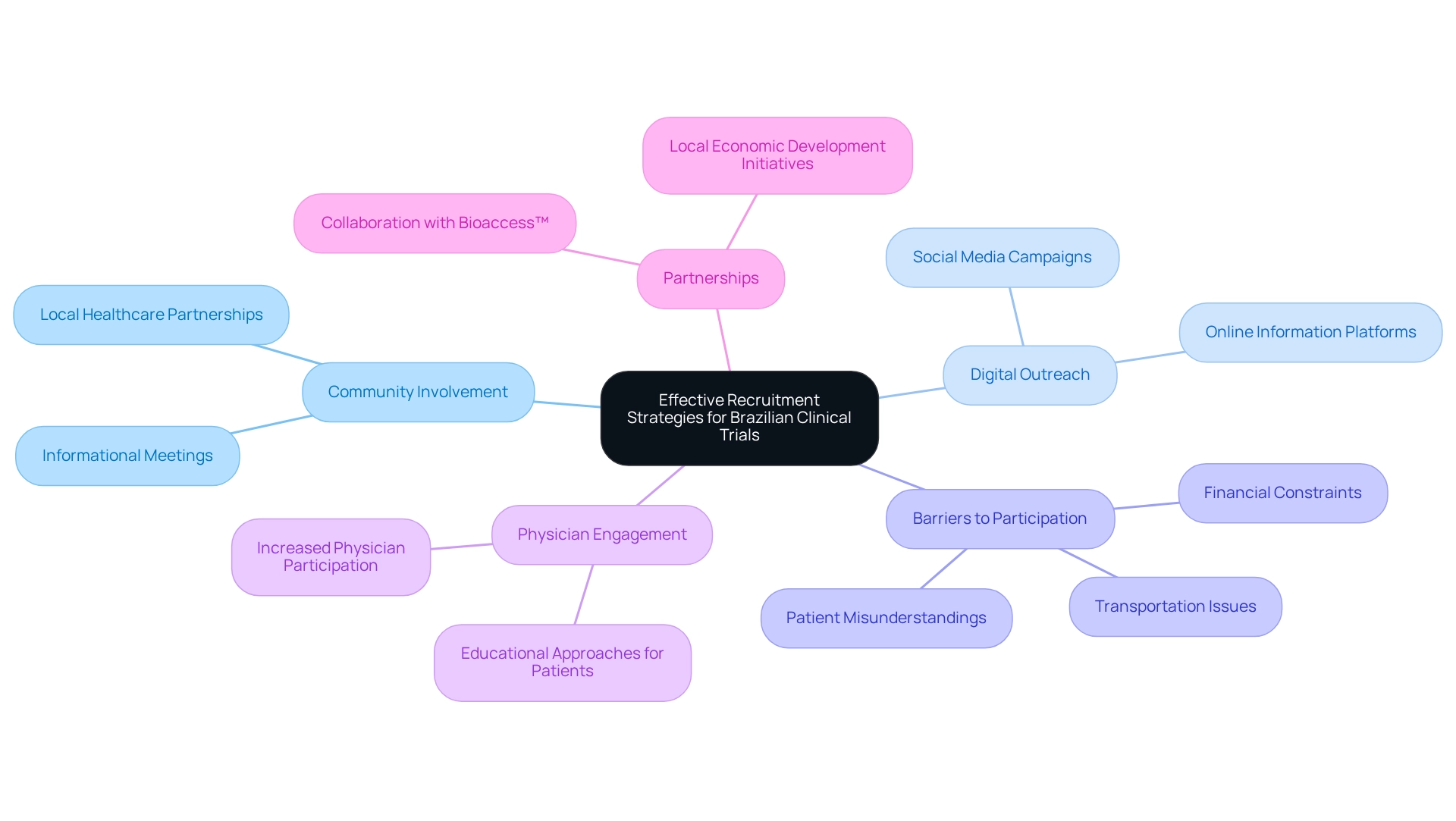
Improving recruitment for research studies in Brazil necessitates the implementation of Brazil-specific clinical trial strategies that align with the local context. Community involvement programs, such as informational meetings and partnerships with local healthcare providers, play an essential role in increasing awareness and generating interest in research studies. These efforts can be complemented by leveraging digital platforms and social media, which are particularly effective in reaching younger demographics. Addressing barriers to participation is equally important.
Factors such as transportation issues and financial constraints can deter potential participants, with dropout rates often attributed to patient misunderstandings, schedule conflicts, and personal matters. Offering support services to mitigate these challenges is essential. Moreover, comprehending the local culture and nurturing trust within communities can greatly improve recruitment results.
Recent findings emphasize the need for greater physician participation in research activities, indicating that educational approaches directed at patients could boost recruitment rates. A study on patient awareness revealed that many doctors recognized low awareness as a major obstacle to recruitment, highlighting the necessity for platforms that share information about ongoing studies and enhance public knowledge.
In Colombia, the partnership between bioaccess™ and Caribbean Health Group seeks to establish Barranquilla as a premier location for research in Latin America, backed by Colombia's Minister of Health. This initiative not only boosts awareness of medical studies but also aids in local economic development through job creation and enhanced healthcare services. Media attention from Clinical Leader enhances this visibility, underscoring the significance of research studies in the region.
Furthermore, GlobalCare Clinical Studies' collaboration with bioaccess™ has shown substantial enhancements in ambulatory services for studies, achieving over a 50% decrease in recruitment time and an impressive 95% retention rate. As Clarissa Mathias, MD, PhD, noted in her observations on establishing a medical study network in the country, tackling these obstacles is essential for effective recruitment. By applying Brazil-specific clinical trial strategies, research studies in the country can attain higher success in participant recruitment.
Cultural Considerations in Clinical Trial Design
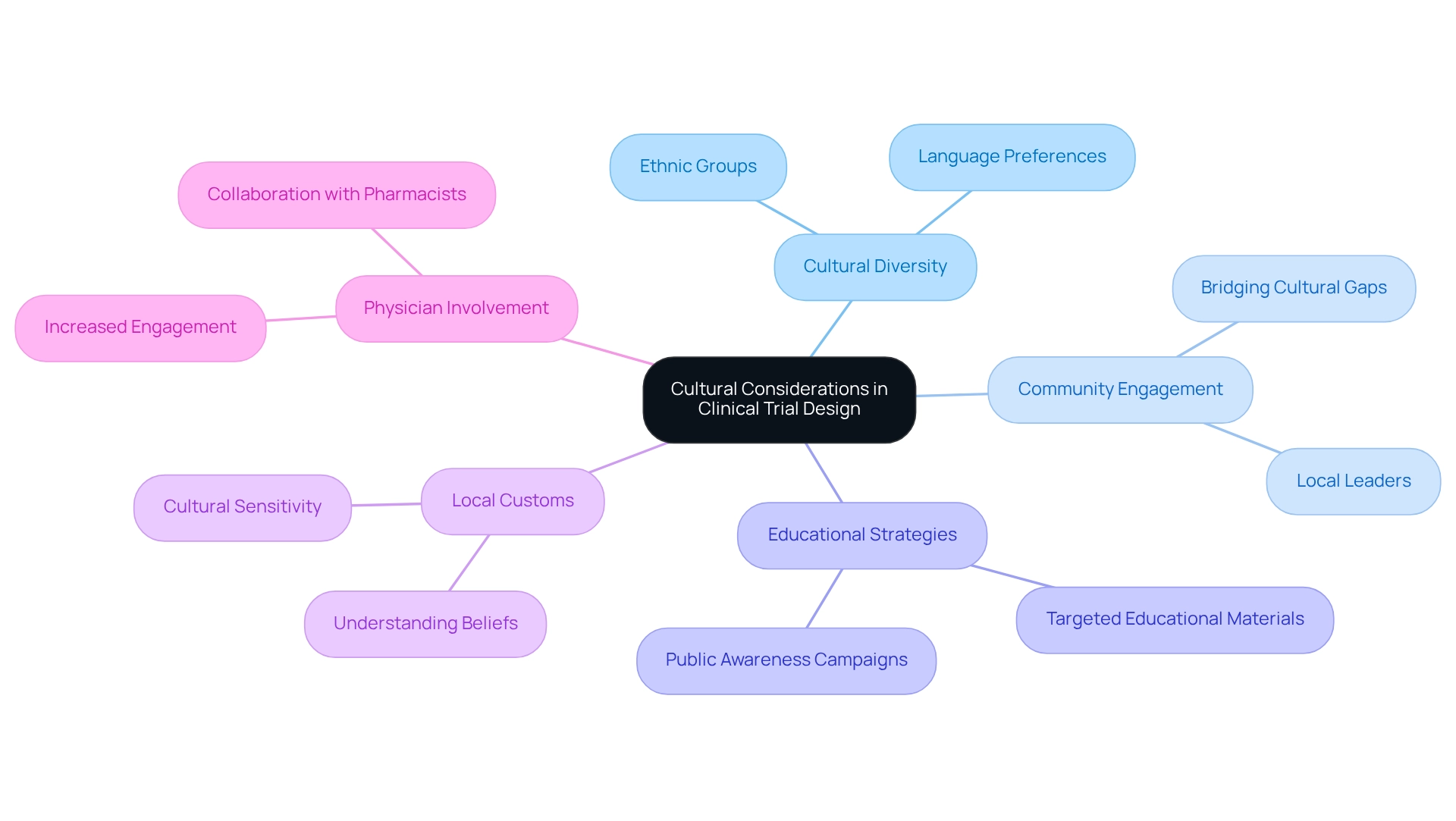
Cultural factors are pivotal in shaping Brazil-specific clinical trial strategies, considering the country's rich diversity of ethnic groups and traditions that profoundly influence patient perspectives on healthcare and medical experimentation. A thorough understanding of local customs, beliefs, and language preferences is essential for enhancing communication and fostering trust between researchers and participants. Engaging local community leaders in the testing process serves as an effective means to bridge cultural gaps and stimulate greater participation, as underscored by Steve Garchow's emphasis on the importance of local perspectives in navigating the complexities of the Latin American market.
Furthermore, customizing educational materials to reflect cultural nuances not only enhances comprehension but also elevates engagement among potential participants. A recent survey has illuminated the critical need for increased public awareness regarding trials and their benefits, revealing that numerous patients remain unaware of available study opportunities. This gap underscores the necessity for targeted educational strategies that align with Brazil-specific clinical trial initiatives and resonate with the cultural context of the Brazilian populace.
The case study titled "Public Awareness and Education in Trials" illustrates that many physicians have noted patients' lack of understanding about trial opportunities, thereby reinforcing the need for effective educational outreach. Statistics reveal that there are over 2.3 billion citations within the academic community; however, the call for enhanced physician involvement in medical studies remains evident. This statistic accentuates the importance of fostering stronger connections between physicians and medical research. By addressing the cultural elements that influence research participation, investigators can enhance recruitment rates and ultimately refine Brazil-specific clinical trial strategies in the realm of medical research.
Moreover, the Brazilian-Portuguese version of the scale for evaluating collaborative attitudes among pharmacists and physicians highlights the ongoing efforts to improve inter-professional cooperation, which is crucial in the context of cultural factors in research. Understanding these dynamics is vital for the effective implementation of research studies and for promoting medical technology in the region, positioning bioaccess® as a key player in supporting these initiatives. Additionally, based on Steve Garchow's experiences, it is imperative to adopt Brazil-specific clinical trial strategies that address local needs and regulatory challenges, ensuring that studies are not only culturally attuned but also strategically aligned with market demands.
This comprehensive approach will enhance the efficacy of research studies and foster greater confidence and involvement among the Brazilian population.
Building Partnerships with Local Institutions and Stakeholders
Establishing collaborations with nearby hospitals, universities, and academic institutions is crucial for the successful implementation of Brazil-specific clinical trial strategies. These partnerships not only provide access to a diverse patient population but also enhance the credibility and visibility of the study. Involving local stakeholders, including patient advocacy organizations and regulatory authorities, is essential to promote more efficient study operations and ensure adherence to Brazil-specific clinical trial strategies.
As Heloisa Resende noted, "The landscape is excessively complex and needs further engagement of all policymakers, pharmaceutical companies, investigators, cooperative groups, medical societies, non-governmental organizations, government and general society to transform it into a more inclusive scenario." Remarkably, a substantial 32.1% of surveyed physicians indicated a deficiency in prior experience with research during their specialty training, underscoring the necessity for strong collaborations to bridge this gap. Moreover, with 4,730 doctors listed at the Federal Council of Medicine engaged in oncology practice, there exists a significant group of qualified experts ready to assist research studies.
Building these relationships early in the trial planning process can enhance recruitment strategies and participant retention when utilizing Brazil-specific clinical trial strategies. Effective partnerships have been demonstrated through initiatives such as the National Program of Oncological Attention (PRONON), which supports Brazil-specific clinical trial strategies and financing for pertinent projects. For instance, the NEOSAMBA project, which explores chemotherapy protocols, illustrates how government assistance can augment research opportunities and ultimately enhance patient care.
Furthermore, the partnership between bioaccess™ and Caribbean Health Group, revealed on March 29, 2019, aims to establish Barranquilla as a leading location for research studies in Latin America, bolstered by the support of Colombia's Minister of Health. By prioritizing local partnerships and utilizing extensive research management services—including feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting—research initiatives in the country can achieve greater success and contribute to the advancement of medical technology.
Leveraging Technology for Enhanced Clinical Trial Efficiency
Incorporating technology into research studies is essential for enhancing both effectiveness and data integrity, particularly in the evolving landscape of the region. At bioaccess®, we offer comprehensive research study management services, which include:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Study setup
- Import permits
- Project management
- Reporting
Our expertise encompasses various types of studies, such as Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH), ensuring we address the diverse needs of our clients.
The adoption of electronic data capture (EDC) systems has been shown to streamline data collection processes, significantly reducing the errors often linked to manual data entry. In 2024, statistics reveal that a considerable portion of medical studies in Brazil is employing EDC as part of Brazil-specific clinical trial strategies, highlighting a growing trend towards digitization in research methodologies.
Mobile health (mHealth) applications further enhance this efficiency by facilitating participant monitoring and engagement. These tools enable real-time data gathering and communication, which are vital for sustaining participant engagement and ensuring data integrity throughout the study process. Additionally, the rise of telemedicine solutions has expanded access for participants in remote areas, effectively broadening the recruitment pool and enhancing diversity in research studies.
It is crucial to note that clinical study data must be retained for a minimum of two years if the investigational product's clinical development is suspended, underscoring a significant compliance factor in the process.
The impact of these technological advancements is profound, resulting in improved trial management and outcomes. Authorities in the field have observed that the transition to electronic data capture not only accelerates the study timeline but also enhances the overall quality of the information collected. As Jennifer Mendoza, a specialist in health, pharma, and medtech, states, "Overall, figures indicate that the medical technology market in the country is continuing to expand."
With over 20 years of experience in Medtech, bioaccess® is committed to supporting medical device research studies in Latin America, ensuring our clients navigate the complexities of this dynamic landscape with efficiency. As the nation continues to advance technologically, particularly in the implementation of AI and data analytics, the adoption of Brazil-specific clinical trial strategies positions the country at the forefront of healthcare research in Latin America. This dedication to leveraging technology highlights the country’s potential to drive significant progress in the medical technology sector.
The case study on Brazil's leadership in AI adoption exemplifies this commitment, as the nation significantly outpaces its regional peers in investing in AI and data analytics, further enhancing its research environment. At bioaccess®, we are dedicated not only to promoting medical studies but also to fostering job creation and economic development in the local context.
Training and Development for Clinical Research Teams
Funding the training and development of medical study teams is essential for upholding high standards in execution. Regular workshops and training sessions are vital for ensuring team members remain informed about the latest regulatory changes, Good Clinical Practice (GCP) guidelines, and technological advancements. Moreover, encouraging team members to pursue certifications and engage in industry conferences significantly enhances their skills and knowledge base.
A well-prepared group is better equipped to identify and address potential obstacles, which is critical for the successful execution of research studies. Recent findings indicate that improving compensation structures for healthcare investigators could attract more physicians to participate in medical activities, thereby enhancing both the quality and quantity of studies conducted in Brazil.
Additionally, a survey revealed that a lack of understanding among both doctors and patients regarding the benefits of medical studies serves as a significant barrier to participation. This highlights the urgent need for targeted educational initiatives aimed at promoting awareness and training for both groups. The case study titled "Awareness and Education as Barriers to Clinical Research Participation" underscores this issue, suggesting that educational initiatives are crucial for improving participation rates.
With new legislation being proposed to streamline and expedite regulatory approvals, there is potential for an increase in Brazil-specific clinical trial strategies tailored for Brazilian patients. Furthermore, financial support for medical research, as evidenced by Gustavo Gössling's funding from organizations such as AstraZeneca/Merck and Janssen Oncology, can further bolster the training and development of research teams. By leveraging the comprehensive research study management services provided by bioaccess®, including feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting, the country can enhance participation rates in research studies.
Ultimately, this leads to more successful outcomes and advancements in medical technology, driving economic growth and healthcare improvement through international collaboration.
Key Takeaways for Successful Clinical Trials in Brazil
To achieve successful clinical studies in Brazil, stakeholders must prioritize strategies tailored to the local context, alongside a comprehensive understanding of the regulatory frameworks and cultural nuances. As Heloisa Resende aptly observed, "The landscape is excessively complex and needs further engagement of all policymakers, pharmaceutical companies, investigators, cooperative groups, medical societies, non-governmental organizations, government, and general society to transform it into a more inclusive scenario." Navigating Brazil-specific clinical trial strategies is critical, as they significantly affect timelines and outcomes.
Organizations should focus on integrating Brazil-specific clinical trial strategies into study designs, ensuring that protocols align with local practices and beliefs. This alignment can enhance participant engagement and retention, which are vital for the success of clinical trials.
Establishing partnerships with local institutions is another essential aspect of Brazil-specific clinical trial strategies. Collaborating with universities, hospitals, and research organizations can provide access to diverse patient populations and valuable insights into local healthcare dynamics. Furthermore, leveraging technology—such as electronic data capture and remote monitoring—can streamline processes, improve data accuracy, and enhance overall study efficiency.
Bioaccess offers comprehensive clinical study management services that include:
- Feasibility studies
- Site selection
- Compliance reviews
- Study setup
- Import permits
- Project management
- Reporting
These services are crucial for navigating the regulatory landscape and ensuring that Brazil-specific clinical trial strategies are executed efficiently and in compliance with local regulations. Continuous training for investigation groups is vital to uphold high standards and ensure adherence to both local and international regulations, particularly in the context of Brazil-specific clinical trial strategies.
Ongoing education regarding Brazil-specific clinical trial strategies and best practices in medical studies will empower teams to adapt effectively to the evolving landscape. Additionally, initiatives led by LACOG and CURA aim to educate the public about Brazil-specific clinical trial strategies, dispel misconceptions, and encourage participation, which is essential for enhancing diversity in medical study populations. The estimated time to complete the survey questionnaire was 10 minutes, underscoring the significance of efficient data collection processes.
By implementing these best practices, including support from SBOC's annual research fund to finance projects addressing disparities in cancer treatment access, organizations can optimize their clinical trial processes using Brazil-specific clinical trial strategies. This optimization ultimately contributes to the advancement of medical research in Brazil and improves patient outcomes.
Conclusion
Brazil's clinical trial landscape presents a unique blend of opportunities and challenges that necessitate a nuanced approach for success. The country's diverse population and evolving regulatory framework establish it as a vital hub for medical research in Latin America, exemplified by nearly 10,000 clinical studies registered to date. However, navigating this landscape requires an understanding of the intricate regulatory environment, cultural considerations, and effective recruitment strategies.
Successful clinical trials depend on building strong partnerships with local institutions and stakeholders, enhancing credibility and facilitating access to diverse patient populations. Additionally, integrating technology into clinical trial processes—such as electronic data capture and mobile health applications—can significantly improve efficiency and data quality. The emphasis on training and development for research teams further underscores the importance of maintaining high standards in trial execution.
To optimize clinical trial outcomes in Brazil, organizations must prioritize cultural sensitivity, regulatory compliance, and community engagement. By leveraging these strategies and the expertise of organizations like bioaccess®, stakeholders can enhance participant recruitment, ensure ethical oversight, and ultimately drive advancements in medical technology. As Brazil continues to evolve its clinical research capabilities, the commitment to innovation and collaboration will be crucial in shaping the future of healthcare in the region.
Frequently Asked Questions
What is the current state of medical studies in Brazil?
As of early 2024, around 10,000 medical studies have been registered in Brazil, highlighting the country's commitment to advancing medical research through Brazil-specific clinical trial strategies.
What are some critical therapeutic areas for research studies in Brazil?
Key therapeutic areas for research studies in Brazil include oncology, cardiology, and infectious diseases.
What challenges do researchers face when conducting studies in Brazil?
Researchers encounter challenges such as bureaucratic hurdles and the need to address inequalities in regional healthcare capabilities, which can affect recruitment strategies, data collection, and execution of clinical trials.
What is the role of the Health Surveillance Inspection Fee (TFVS)?
The TFVS is a mechanism that generates charges related to health monitoring, adding an extra layer of regulatory consideration for sponsors of medical studies.
How does the National Research Ethics Commission (CONEP) contribute to medical studies?
CONEP oversees the ethical dimensions of studies involving human participants, ensuring compliance with ethical standards and safeguarding participant welfare, thereby enhancing the credibility of clinical studies in Brazil.
What is the significance of the Brazilian National Health Surveillance Agency (ANVISA)?
ANVISA serves as the cornerstone of medical study regulation in Brazil, and the introduction of Resolution No. 945/2024 has streamlined the approval process for research studies, improving efficiency for researchers.
Why is early engagement with local ethics committees important?
Early engagement with local ethics committees is crucial as their input significantly influences the approval process and helps ensure adherence to ethical considerations.
What are the additional safeguards required for studies involving women of childbearing age or expectant mothers?
Research studies involving these participants must ensure they are fully informed of potential risks and prioritize their rights, including not compelling them to use contraceptives unless pregnancy is the study's primary focus.
How does bioaccess® support medical research in Brazil?
bioaccess® provides expertise and tailored approaches to navigate regulatory complexities, offering services that cover study set-up, regulatory compliance, project management, and reporting to enhance the likelihood of successful outcomes.
What is the focus of bioaccess®'s accelerated medical device research study services?
These services are designed to assist clients in effectively navigating regulatory changes while ensuring that the unique needs of each study are met through a flexible and customized approach.

