Overview
Best practices for medical device trial protocols underscore the critical need for structured frameworks that guarantee compliance and efficiency in clinical research. This article emphasizes that well-defined protocols—encompassing clear participant criteria and robust statistical analysis—are vital for regulatory adherence and the integrity of research outcomes. Such elements not only facilitate the successful advancement of medical devices but also enhance the overall credibility of clinical trials.
Introduction
In the realm of medical device research, clinical trial protocols serve as the backbone of successful studies, guiding every aspect from objectives to methodologies. As the industry evolves, the emphasis on meticulous protocol design has never been more critical, particularly in ensuring regulatory compliance and safeguarding participant integrity. This article delves into the essential components of effective trial protocols, the intricacies of navigating regulatory landscapes, and the importance of patient safety and ethical compliance.
Furthermore, it highlights innovative strategies that enhance research efficiency, paving the way for advancements in medical technologies. By understanding these elements, stakeholders can better navigate the complexities of clinical trials and contribute to the successful development of life-saving devices.
Understanding Medical Device Trial Protocols
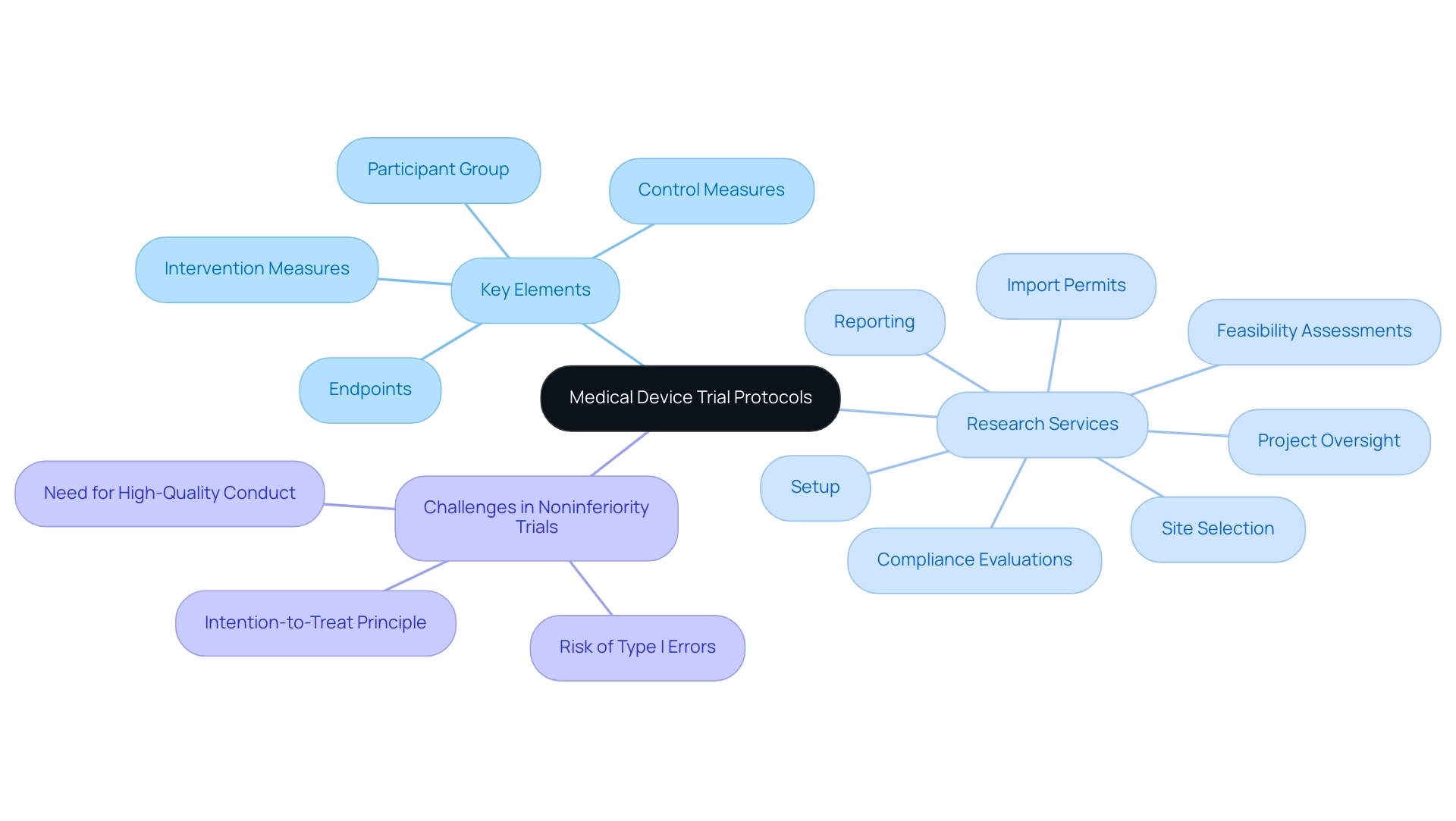
Medical device trial protocols serve as crucial frameworks for examinations, meticulously detailing the objectives, structure, methodology, and statistical aspects. A well-structured medical device trial protocol is essential not only for regulatory compliance but also for upholding the integrity of research. Key elements of effective medical device trial protocols include:
- A precise definition of the participant group
- Comprehensive descriptions of intervention and control measures
- Clearly established endpoints for assessing the system's safety and efficacy
In 2025, the emphasis on organized protocols has intensified, with statistics indicating that adherence to these guidelines can significantly enhance compliance rates across medical studies. This systematic approach not only streamlines the execution of research related to medical device trial protocols but also bolsters the reliability of results, ultimately contributing to the successful advancement of medical devices.
At bioaccess®, we specialize in comprehensive research management services, encompassing:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Setup
- Import permits
- Project oversight
- Reporting
Our expertise in managing diverse research types—including Early-Feasibility Research (EFS), First-In-Human Research (FIH), Pilot Research, Pivotal Research, and Post-Market Follow-Up Research (PMCF)—ensures that your evaluations are conducted efficiently and effectively.
Expert opinions underscore the critical nature of medical device trial protocols; clinical research leaders advocate for thorough documentation and strict adherence to best practices to mitigate risks associated with their execution. For instance, recent case analyses have revealed that noncompliance with experimental protocols can lead to significant biases and erroneous conclusions. The study titled 'Challenges in Noninferiority Studies' emphasizes that applying the intention-to-treat principle in noninferiority studies may increase the risk of type I errors, further reinforcing the necessity for high-quality conduct and reporting.
As the landscape of medical equipment research evolves, remaining informed about current best practices and integrating expert insights into medical device trial protocols will be vital for successful navigation of regulatory pathways and effective evaluation of innovative medical technologies. By leveraging our 20+ years of experience in Medtech, bioaccess® is dedicated to advancing healthcare through rigorous and compliant clinical research.
Navigating Regulatory Requirements for Medical Device Trials
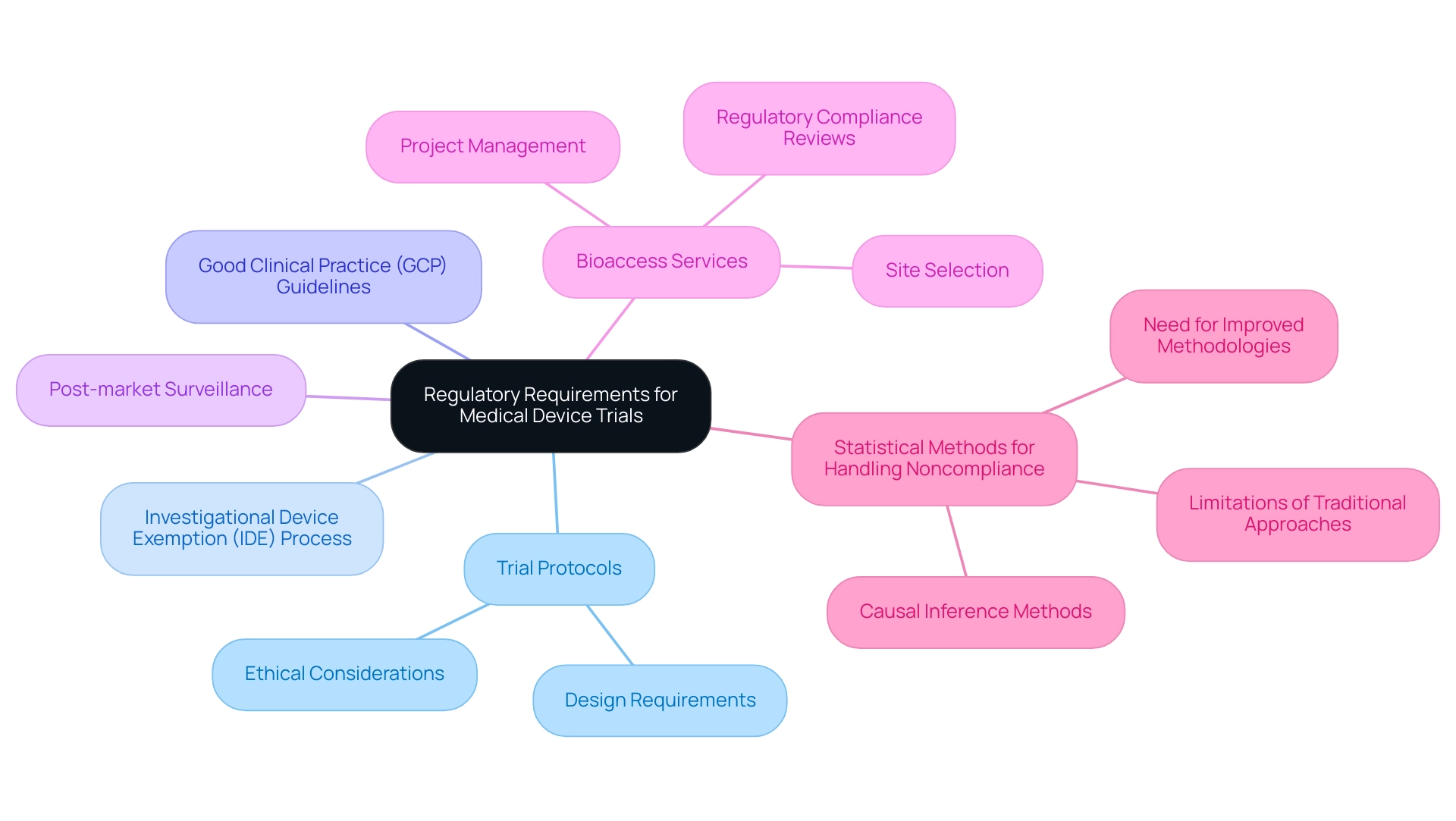
Regulatory requirements for medical device trial protocols are crucial and vary significantly across regions, primarily overseen by the FDA in the United States and the EMA in Europe. Clinical researchers must possess a comprehensive understanding of these regulations, which encompass:
- Medical device trial protocols
- The Investigational Device Exemption (IDE) process
- Good Clinical Practice (GCP) guidelines
- Post-market surveillance obligations
Adhering to these guidelines is essential, as it not only protects participants but also enhances the reliability of research outcomes.
At bioaccess, we provide a thorough process for advancing medical assessments, which includes feasibility evaluations, site selection, and investigator recruitment. Our services encompass:
- Regulatory compliance reviews
- Setup
- Import permits
- Nationalization of investigational devices
- Project management
This ensures that all necessary approvals are obtained efficiently. Recent evaluations reveal that only 9% of research considered compliance as a factor in their analysis, underscoring a significant gap in addressing its impact on treatment estimates.
This underscores the importance for researchers to engage with regulatory bodies early in the study design process. By doing so, they can proactively address potential compliance issues and streamline their path to market. Furthermore, a systematic review of statistical methods for managing noncompliance in randomized controlled experiments demonstrated that while many studies employed causal inference methods, none utilized Complier Average Causal Effect (CACE) analysis specifically for intervention products.
This gap emphasizes the need for improved techniques and reporting standards in studies involving medical device trial protocols. Mostazir et al. (2019) evaluated these statistical methods, highlighting the limitations of conventional approaches and the necessity for enhanced methodologies.
Successful case examples illustrate that navigating the regulatory landscape efficiently can yield positive outcomes. For instance, studies that closely followed FDA and EMA guidelines not only achieved compliance but also accelerated the path to market for innovative medical devices. As Professor A Davies, lead researcher of the NESIC project funded by the National Institute for Health Research (NIHR) Efficacy and Mechanism Evaluation (EME), noted, collaborating with specialists in Regulatory Affairs, such as Ana Criado and Katherine Ruiz, can provide invaluable insights into the latest requirements and best practices, ensuring that research activities are both compliant and efficient.
Moreover, bioaccess emphasizes the importance of comprehensive reporting, which includes research status, inventory, and serious and non-serious adverse events, to maintain transparency and accountability throughout the process.
Key Components of an Effective Clinical Trial Protocol
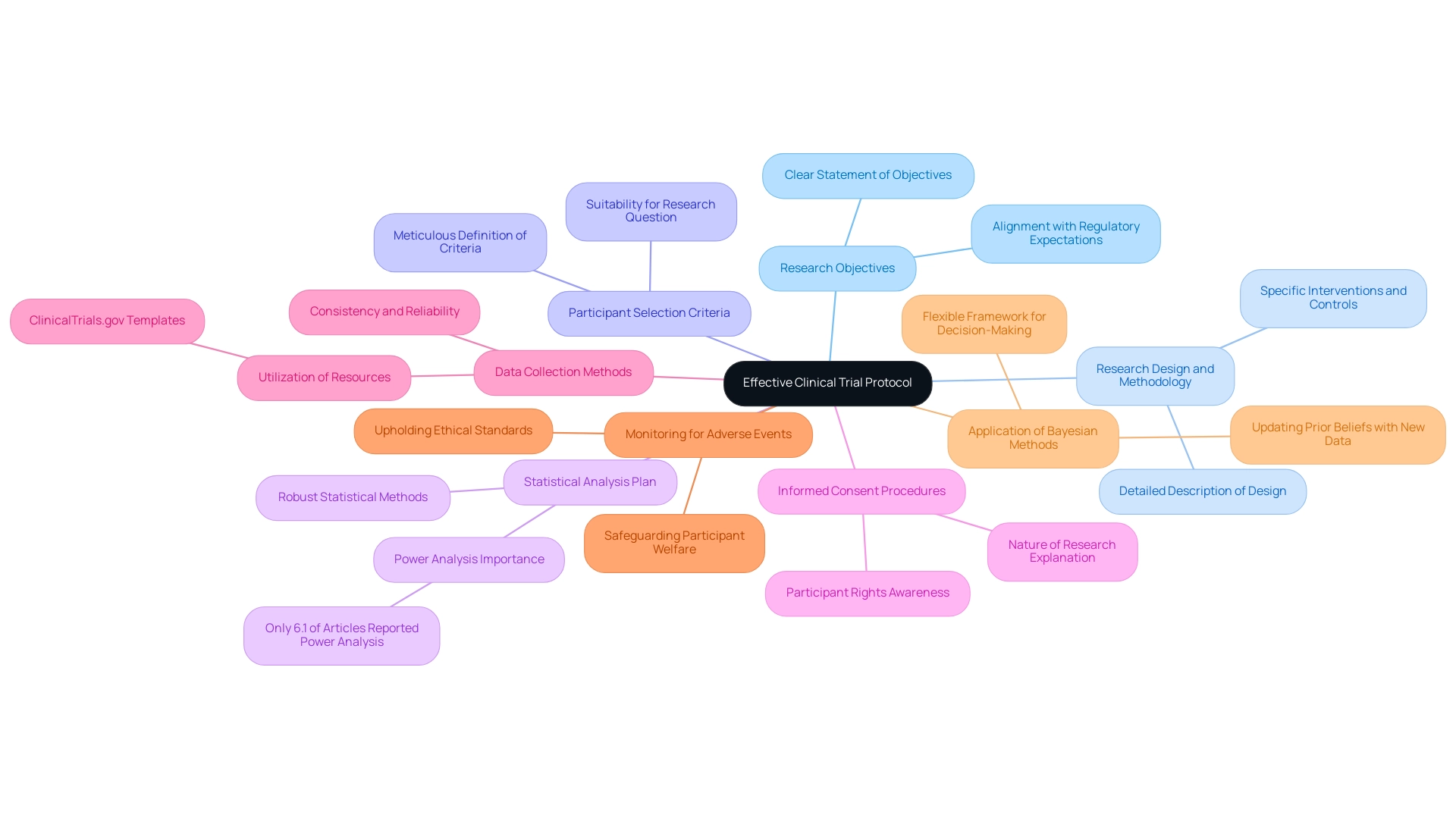
A well-organized clinical research protocol is essential for the success of medical device investigations and must adhere to established medical device trial protocols, encompassing several key elements. First and foremost, a clear statement of the research objectives lays the foundation for the entire protocol, directing the research and ensuring alignment with regulatory expectations. Detailed descriptions of the research design and methodology are crucial, as they delineate how the trial will be conducted, including the specific interventions and controls involved.
Criteria for participant selection must be meticulously defined to ensure that the population is suitable for the research question. Notably, statistics reveal that only 6.1% of articles in British orthopedic journals included a statistical power analysis, highlighting a common oversight in medical research that can impact the validity of findings. Consequently, a robust statistical analysis plan is indispensable, outlining the methods for data analysis and ensuring that the research is adequately powered to detect meaningful effects.
As Howard Barkan, an affiliated researcher and consulting statistician, emphasizes, "Statistics in clinical research: Important considerations" must be acknowledged to enhance the reliability of results.
In addition to these components, the protocol should clearly outline procedures for obtaining informed consent, ensuring that participants are fully aware of their rights and the nature of the research. Data collection methods must be specified to maintain consistency and reliability, while monitoring for adverse events is critical to safeguard participant welfare and uphold ethical standards. Consistency is also vital, and utilizing resources like clinicaltrials.gov for templates and past protocols can streamline the protocol writing process and ensure adherence to best practices.
Incorporating these elements not only fulfills regulatory requirements but also enhances the efficiency of study execution and the integrity of the collected data. For instance, the application of Bayesian methods in medical research demonstrates how adaptable frameworks can improve decision-making processes, particularly in dose-finding studies. By revising previous beliefs with recent information, researchers can refine their methods and more accurately assess dose-toxicity connections, ultimately resulting in more informed and efficient medical studies.
Focusing on these vital elements enables researchers to create medical device trial protocols that are not only compliant but also optimized for success in advancing medical devices swiftly. This aligns with bioaccess®'s mission to offer extensive research management services, leveraging their expertise in Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies to promote successful outcomes in the Medtech sector. With over 20 years of experience in Medtech, bioaccess® distinguishes itself as a reliable partner in managing the complexities of research studies, employing unique approaches that enhance the overall research process.
Ensuring Patient Safety and Ethical Compliance in Trials
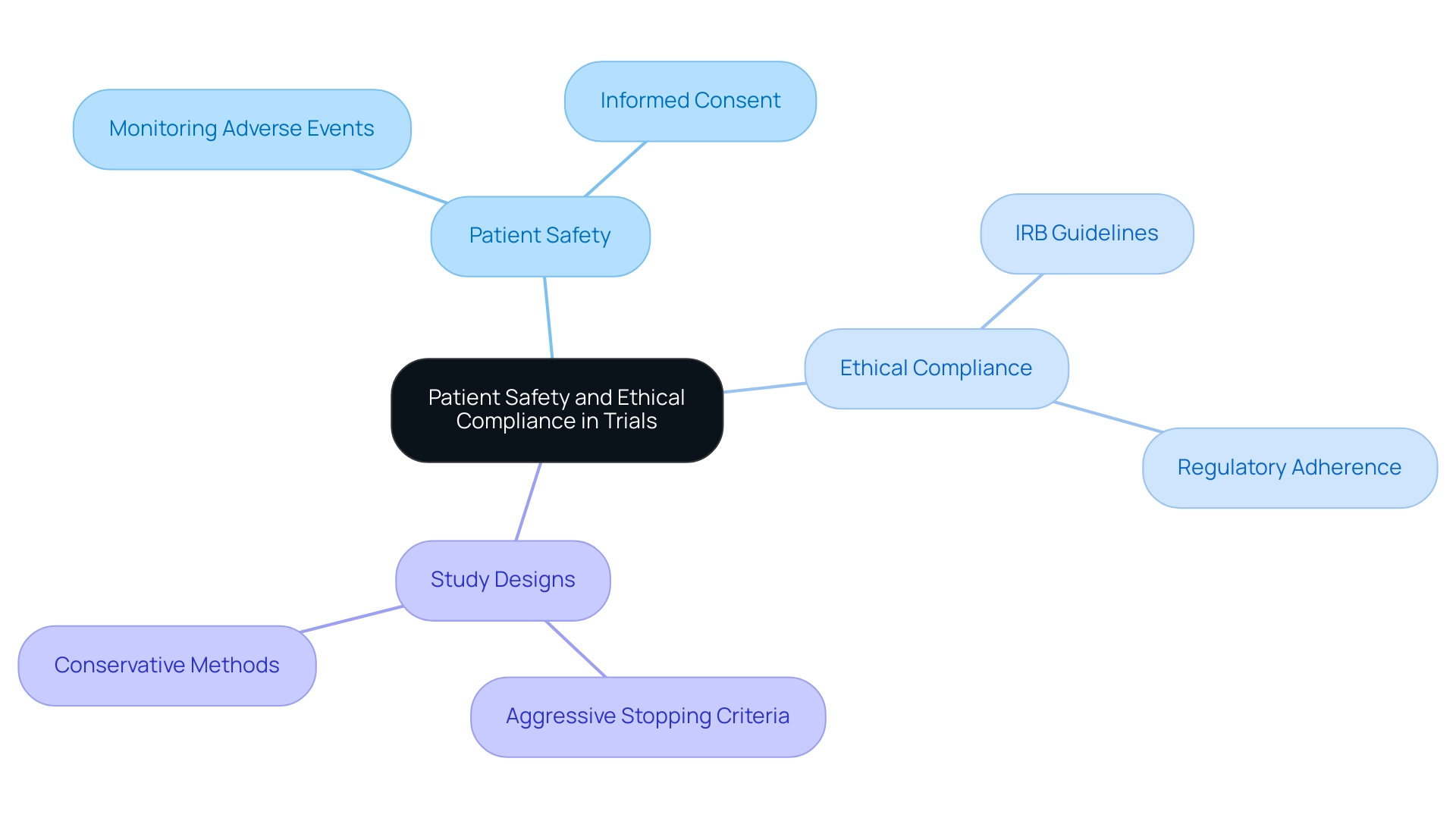
Patient safety remains the foremost concern in medical studies, necessitating the implementation of stringent protocols for monitoring adverse events and securing informed consent from all participants. Ethical compliance is essential, requiring adherence to guidelines established by Institutional Review Boards (IRBs) and regulatory agencies that govern the ethical conduct of research. Creating a Data Safety Monitoring Board (DSMB) is a best practice that offers independent oversight of the study's safety data, ensuring that any potential risks are swiftly addressed.
In 2025, the landscape of patient safety in research is evolving, with an increasing emphasis on innovative study designs that prioritize ethical compliance. Recent findings indicate that aggressive stopping criteria can lead to fewer expected toxicities, although they may compromise statistical power. Specifically, the modified SPRT method has been shown to have similar or worse power than the modified SPoRTs while exhibiting higher expected events than the SPh method.
Conversely, more conservative methods may enhance power but result in higher expected toxicities. This balance is essential for ensuring patient safety while achieving legitimate research outcomes. Moreover, insights from ethical compliance specialists underscore the significance of cultivating trust and transparency throughout the research process.
Erin Grace, M.H.A., Acting Director of AHRQ’s Center for Quality Improvement and Patient Safety, emphasizes that when we collaborate towards a common goal and rely on evidence, we can achieve remarkable results. By prioritizing patient safety and adhering to ethical standards within medical device trial protocols, researchers can significantly improve the success rates of medical studies. The incorporation of robust patient safety measures not only protects participants but also enhances the overall integrity and credibility of medical device trial protocols.
Additionally, bioaccess provides extensive research management services, including feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting. These services are designed to ensure that medical device trial protocols are conducted efficiently and ethically, ultimately contributing to the enhancement of healthcare results and economic growth in local communities. The continuous efforts of the Patient Safety Authority to promote high-quality reporting and patient safety practices in Pennsylvania healthcare facilities emphasize the existing commitment to enhancing patient safety in medical studies.
This ensures that the discussion remains timely and relevant.
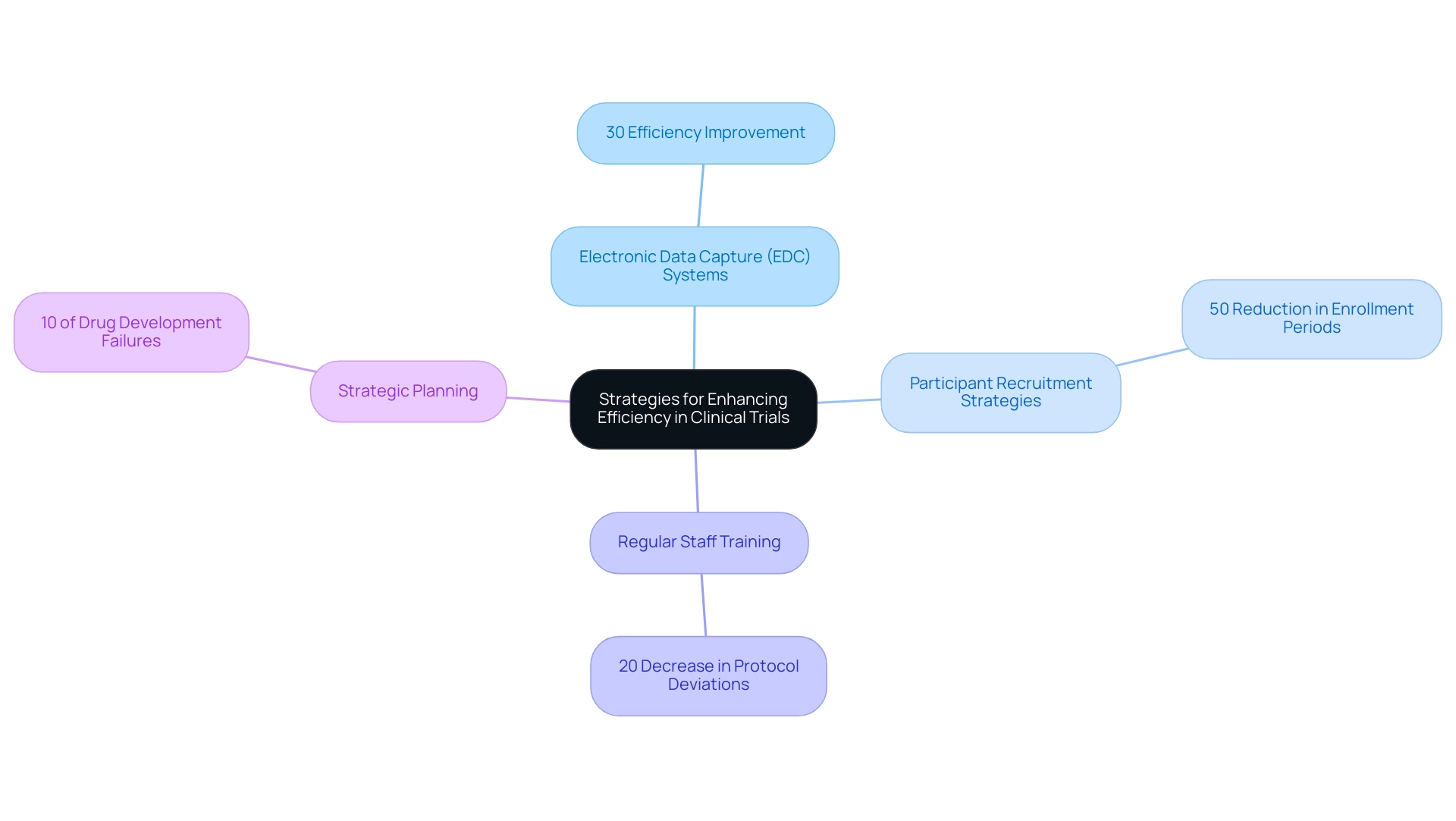
Strategies for Enhancing Efficiency in Clinical Trials
To significantly enhance efficiency in medical studies, the adoption of medical device trial protocols and electronic data capture (EDC) systems is crucial. These systems streamline data collection and management, reducing the likelihood of errors and expediting the overall process. Studies indicate that the adoption of EDC can result in efficiency enhancements of up to 30% in medical studies, emphasizing its essential role in medical device trial protocols and contemporary research methodologies.
As highlighted by TransCelerate Biopharma, the urgency to accelerate research processes has become increasingly important, underscoring the need for effective solutions like EDC systems.
In addition to EDC systems, enhancing participant recruitment strategies is vital. Utilizing patient registries and engaging in community outreach can dramatically shorten enrollment timelines, which often serve as a bottleneck in clinical trials. Research has shown that focused recruitment efforts can reduce enrollment periods by up to 50%, enabling faster initiation and completion of research.
Eliminating these bottlenecks is critical for conserving resources and accelerating research progress. Regular training for study staff on medical device trial protocols, protocol adherence, and data management practices is also essential. This training not only reduces errors but also guarantees adherence to regulatory standards, which is vital for preserving the integrity of the study. Specialists in medical research emphasize that continuous education and training related to medical device trial protocols can lead to a 20% decrease in protocol deviations, further enhancing study efficiency.
Moreover, inadequate strategic planning, including alterations in therapeutic emphasis and corporate mergers, contributes to 10% of medication development failures, highlighting the importance of effective planning in improving overall study success.
By implementing these strategies, researchers can enhance the overall efficiency of studies while ensuring timely and dependable outcomes, ultimately promoting the advancement of medical solutions and therapies that can significantly impact patient care. bioaccess® plays a pivotal role in connecting Medtech firms in Latin America, facilitating the swift progress of medical instruments through its proficiency in research, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Research Follow-Up Studies. With an emphasis on innovation and regulatory excellence, bioaccess® stands at the forefront of navigating the Latin American Medtech landscape, offering extensive trial management services that address the distinct challenges and opportunities for market access.
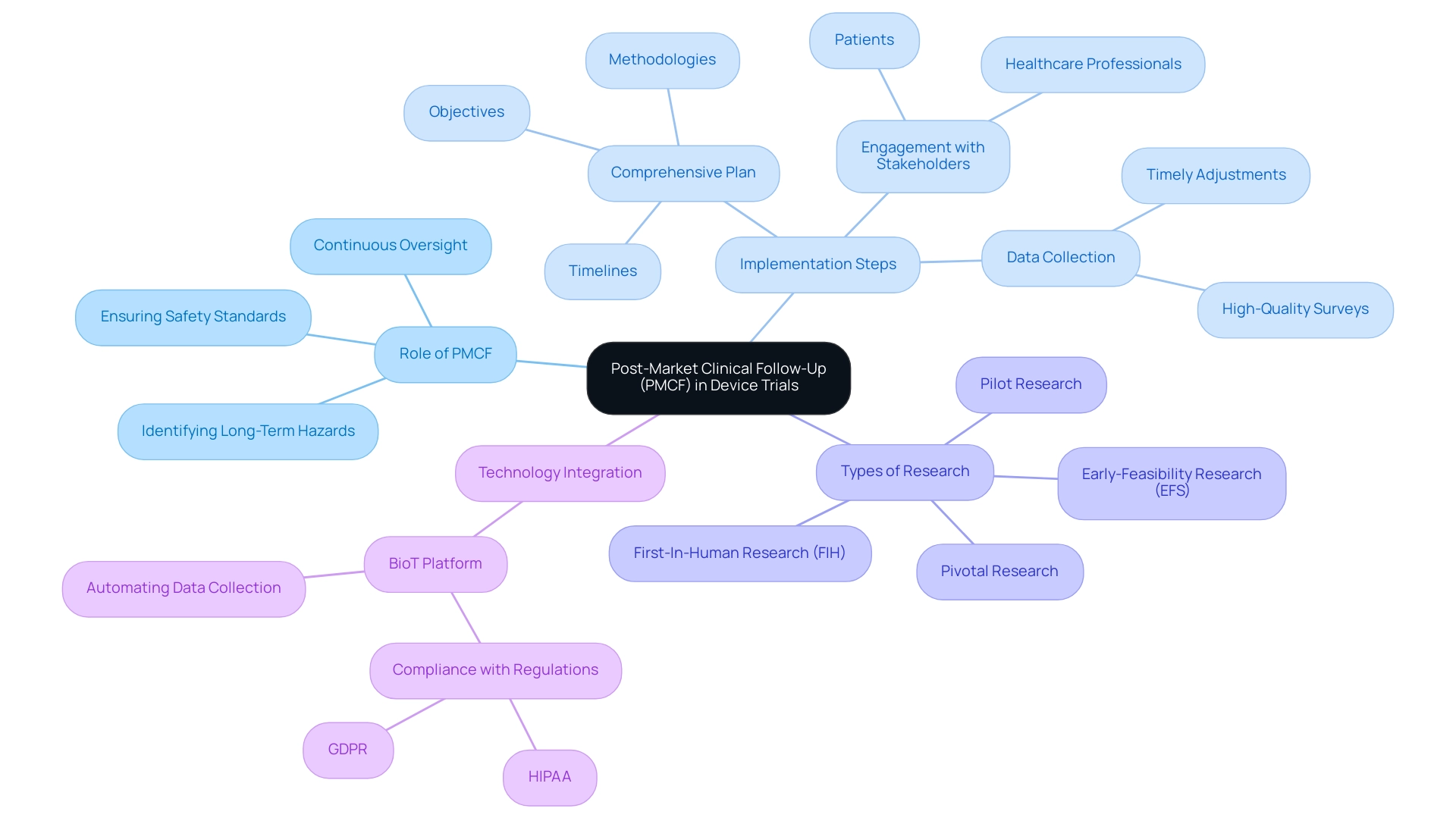
The Role of Post-Market Clinical Follow-Up in Device Trials
Post-market clinical follow-up (PMCF) evaluations are crucial for the continuous oversight of medical tools' safety and performance after market approval. These investigations play an essential role in identifying long-term hazards associated with equipment, ensuring that they consistently meet established safety standards. In 2025, the Medical Device Co-ordination Group (MDCG) underscores the significance of PMCF research, evaluating data collection techniques for legacy equipment and noting that PMCF surveys typically provide Level 4 Evidence. This classification indicates that PMCF surveys serve as a reliable source of information for assessing the safety and effectiveness of medical products in real-world environments.
To effectively implement PMCF studies, manufacturers must devise a comprehensive plan that clearly outlines the objectives, methodologies, and timelines for medical device trial protocols and follow-up studies. Engaging healthcare professionals and patients during this phase is vital, as their feedback can offer invaluable insights into real-world performance. High-quality surveys can facilitate data collection within weeks or, at most, months, enabling timely adjustments and improvements—an essential aspect of maintaining safety and efficacy.
bioaccess® possesses the expertise and tailored strategy necessary to navigate the complexities of PMCF evaluations, ensuring that manufacturers can efficiently oversee their products post-market. With over 20 years of experience in the Medtech field, bioaccess® specializes in a variety of research, including:
- Early-Feasibility Research (EFS)
- First-In-Human Research (FIH)
- Pilot Research
- Pivotal Research
A notable case study is Jasper's experience in the MedTech sector, where his extensive background in behavioral economics and consultancy has enabled him to generate insights that drive innovation in medical equipment.
As Jasper Van de Sande articulates, "Integrating market insights into PMCF strategies is essential for ensuring that devices not only comply with regulatory requirements but also meet the evolving needs of users." His expertise highlights the critical nature of this integration in enhancing the overall effectiveness of PMCF research.
Moreover, the emergence of platforms like BioT, a cloud-based solution designed for automating PMCF data collection, accentuates the importance of compliance and efficiency in these investigations. By leveraging such technologies, manufacturers can streamline their data collection processes while ensuring adherence to regulations like HIPAA and GDPR.
As the landscape of medical equipment regulation continues to evolve, the role of PMCF research in safety monitoring within medical device trial protocols becomes increasingly vital. By prioritizing these studies, manufacturers can bolster their compliance efforts and contribute to the ongoing improvement of medical devices, ultimately enhancing patient safety and healthcare outcomes.
Conclusion
The realm of medical device clinical trials underscores the essential role of well-structured protocols, regulatory compliance, and a commitment to patient safety. Effective trial protocols provide a solid foundation for studies, incorporating clearly defined objectives, methodologies, and statistical analyses. Adherence to these protocols enhances research reliability and facilitates regulatory navigation, ultimately expediting the market entry of innovative medical technologies.
Regulatory compliance is crucial; proactive engagement with regulatory bodies and adherence to guidelines ensure participant protection and credible outcomes. Expert evaluations indicate a gap in compliance considerations among researchers, highlighting the need for early engagement in trial design to mitigate risks.
Patient safety and ethical compliance are paramount. The establishment of independent monitoring bodies and rigorous safety protocols safeguard participants and uphold the integrity of research. As the field evolves, integrating innovative trial designs and robust reporting mechanisms will further enhance credibility and success rates.
In conclusion, advancements in clinical trial methodologies, combined with a focus on patient safety and regulatory adherence, are vital for the future of medical device research. By leveraging expert insights and adopting best practices, stakeholders can effectively navigate the complexities of clinical trials, fostering innovation in medical technologies and improving healthcare outcomes for patients.
Frequently Asked Questions
What is the purpose of medical device trial protocols?
Medical device trial protocols serve as essential frameworks for examinations, detailing the objectives, structure, methodology, and statistical aspects necessary for regulatory compliance and maintaining research integrity.
What are the key elements of effective medical device trial protocols?
Key elements include a precise definition of the participant group, comprehensive descriptions of intervention and control measures, and clearly established endpoints for assessing the safety and efficacy of the system.
How has the emphasis on organized protocols changed in 2025?
In 2025, there has been an intensified emphasis on organized protocols, with statistics indicating that adherence to these guidelines can significantly enhance compliance rates across medical studies.
What services does bioaccess® provide in relation to medical device trials?
bioaccess® offers comprehensive research management services, including feasibility assessments, site selection, compliance evaluations, project oversight, and reporting, among others.
What types of research does bioaccess® manage?
bioaccess® manages various types of research, including Early-Feasibility Research (EFS), First-In-Human Research (FIH), Pilot Research, Pivotal Research, and Post-Market Follow-Up Research (PMCF).
Why is thorough documentation important in medical device trials?
Thorough documentation is critical to mitigate risks associated with trial execution, as noncompliance with experimental protocols can lead to significant biases and erroneous conclusions.
What regulatory bodies oversee medical device trial protocols?
In the United States, the FDA oversees medical device trial protocols, while in Europe, it is managed by the EMA.
What are some key regulatory requirements for medical device trials?
Key regulatory requirements include understanding medical device trial protocols, the Investigational Device Exemption (IDE) process, Good Clinical Practice (GCP) guidelines, and post-market surveillance obligations.
What is the significance of engaging with regulatory bodies early in the study design process?
Engaging with regulatory bodies early allows researchers to proactively address potential compliance issues, streamlining their path to market.
What does bioaccess® emphasize regarding reporting in medical device trials?
bioaccess® emphasizes the importance of comprehensive reporting that includes research status, inventory, and adverse events to maintain transparency and accountability throughout the process.

