Overview
Medical device clinical trial design is paramount for ensuring the safety and effectiveness of new medical technologies. It serves as a guiding force in regulatory approval processes and plays a crucial role in improving patient outcomes. Adhering to rigorous research protocols and optimizing study processes are vital for navigating the complex regulatory landscapes that characterize this field. These elements enhance overall trial efficiency, contributing significantly to successful market entry and ongoing product safety monitoring.
Introduction
In the rapidly evolving landscape of medical technology, the significance of clinical trials cannot be overstated. These trials serve as the bedrock for assessing the safety and efficacy of new medical devices, guiding developers through a complex regulatory maze while ensuring that innovations translate into tangible benefits for patients. As the biopharma industry faces increasingly crowded clinical markets in 2025, understanding the intricacies of clinical trials has become more crucial than ever.
From early-feasibility studies to post-market surveillance, each phase plays a pivotal role in refining devices and enhancing patient outcomes. This article delves into the multifaceted world of clinical trials, exploring their importance, the challenges faced during execution, and innovative strategies that can propel medical device development forward.
Understanding the Importance of Clinical Trials in Medical Device Development
Medical device clinical trial design is crucial in the development process of medical equipment, serving as the foundation for assessing safety and effectiveness. In 2025, as the biopharma sector navigates congested research markets, the importance of these studies is more evident than ever. They provide essential information that guides regulatory bodies, healthcare professionals, and patients regarding the potential advantages and risks associated with new equipment.
By adhering to strict research protocols, developers ensure their products meet the stringent standards required for market approval, which is vital for improving patient outcomes and advancing healthcare solutions.
The success rates of studies focusing on medical device clinical trial design have shown a positive trend, with innovative methodologies and regulatory advancements streamlining processes. For instance, the FDA's proposed mandate for single Institutional Review Board (IRB) reviews aims to alleviate administrative burdens, expediting study initiation while upholding high ethical standards. This regulatory shift illustrates a broader commitment to enhancing the efficiency of medical device clinical trial design, which is essential in a landscape where pharmaceutical companies increasingly concentrate on fewer, high-value therapeutic areas.
In fact, in 2025, the overall number of medical studies is expected to decline as companies focus their efforts on these high-value areas.
Recent insights from industry specialists underscore the importance of evaluations in the context of medical device clinical trial design to ensure device safety and effectiveness. As emphasized by a Senior Director within a global biopharma’s clinical sciences group, optimizing study processes can significantly reduce workload. For example, eliminating one 20-minute task per visit across 130,000 visits saves 43,000 hours of work, allowing Clinical Research Associates (CRAs) to concentrate on essential tasks that enhance study integrity.
Eliminating even minor inefficiencies can yield substantial time savings across thousands of visits, ultimately improving overall success.
Practical examples further illustrate how medical device clinical trial design research studies have bolstered medical equipment safety and effectiveness. By systematically evaluating equipment in diverse populations within the framework of medical device clinical trial design, developers can identify potential issues early, leading to timely modifications that enhance product performance. This proactive approach not only safeguards patient health but also fosters trust among stakeholders, reinforcing a commitment to ethical research practices.
Moreover, bioaccess® has established itself as a recognized CRO and consulting partner for U.S. medical device firms in Colombia, facilitating accelerated clinical research services that contribute to local economic growth and healthcare enhancement.
Additionally, bioaccess® specializes in various types of research, including Early-Feasibility Assessments (EFS), First-In-Human Trials (FIH), Pilot Evaluations, Pivotal Analyses, and Post-Market Clinical Follow-Up Evaluations (PMCF). These investigations not only confirm the safety and effectiveness of new technologies but also significantly contribute to job creation and economic growth in the regions where they are conducted.
The case study titled "Regulatory Innovation in Research" highlights how regulatory bodies like the FDA are modernizing regulations to facilitate efficient study conduct while maintaining high standards, which is crucial in today's complex research environment. The recent presentation by Dr. Jorge Hernando Ulloa at the Charing Cross International Symposium, showcasing one-year first-in-human data for the VenoValve®, exemplifies the advancements in vascular medicine that arise from thorough research studies.
In conclusion, the importance of research studies in the context of medical device clinical trial design for advancing medical equipment cannot be overstated. They are essential for validating the safety and efficacy of new technologies, ensuring that innovations translate into real-world benefits for patients and healthcare systems alike. As Max Baumann from Treehill Partners cautions, the congested medical markets present significant business model challenges for biotech, making the role of studies even more critical in navigating these complexities.

Navigating the Stages of Medical Device Clinical Trials
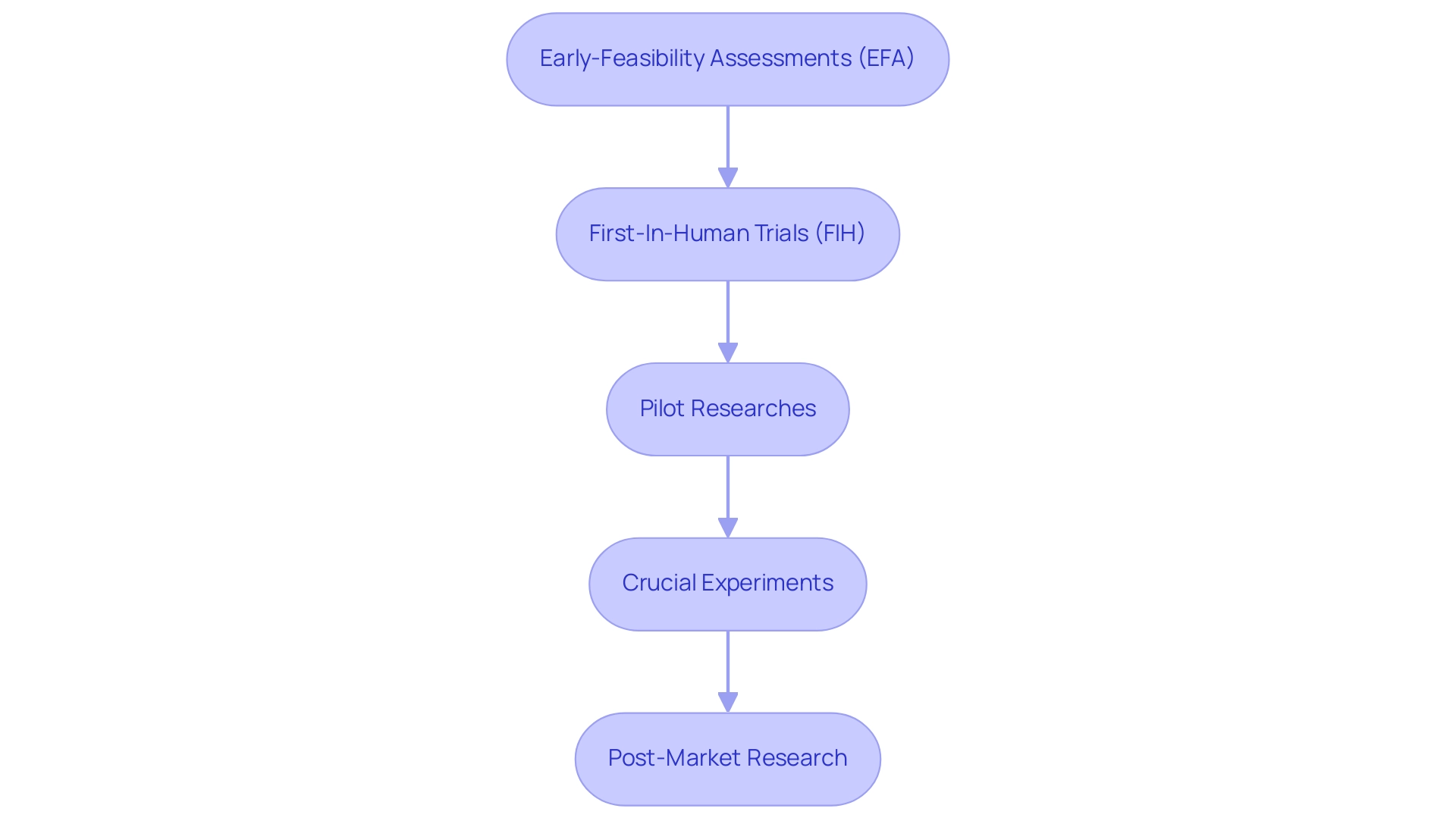
Medical device clinical trials are structured into several critical stages, each serving a distinct purpose in the development and validation of new technologies. Bioaccess® is here to guide you through each phase with our comprehensive clinical trial management services:
-
Early-Feasibility Assessments (EFA): These preliminary evaluations are essential for assessing the potential of a medical device. They help gather preliminary data on safety and efficacy, informing the design of subsequent trials. At bioaccess®, we leverage over 20 years of Medtech expertise to ensure these analyses are conducted effectively.
-
First-In-Human Trials (FIH): Following successful EFA, FIH trials are conducted to evaluate the apparatus's performance in humans for the first time. This phase is critical in medical device clinical trial design for understanding the safety profile and initial effectiveness of the apparatus, laying the groundwork for further development.
-
Pilot Researches: These initial investigations are essential for evaluating the viability of a medical apparatus. They collect essential information on safety and efficacy, offering insights that inform later phases of the study. The success rates for pilot investigations in medical device evaluations can vary significantly, with approximately 5-14% of treatments advancing through all phases to obtain regulatory approval. This emphasizes the significance of comprehensive pilot investigations in reducing risks early in the development process. At bioaccess®, we leverage over 20 years of Medtech expertise to ensure these analyses are conducted effectively.
-
Crucial Experiments: After successful preliminary tests, crucial experiments are carried out on a larger scale. These experiments aim to generate definitive evidence regarding the device's effectiveness and safety, which is often a prerequisite for regulatory approval. The design of crucial experiments, especially in medical device clinical trial design, must be robust, incorporating insights from pilot studies to ensure comprehensive evaluation. For instance, the integration of electronic health records (EHR) and other data sources is increasingly recognized as essential for contemporary research methodologies, enhancing data accuracy and reliability. Additionally, as noted by Rebeca Sanz Barriuso, "through the Collective Minds Research for CROs platform, we support all clinical trial phases I-IV," emphasizing the importance of comprehensive support throughout the trial process, which bioaccess® provides.
-
Post-Market Research: After a product gains approval, post-market research plays a crucial role in observing its performance in actual environments. These investigations are designed to ensure ongoing safety and efficacy, providing valuable data that can inform future iterations of the device or its application. The Medical Device Regulation (MDR) highlights the significance of data standards in these analyses, particularly as electronic data capture (EDC) systems become more prevalent. Effective management of metadata is crucial for integrating research design, data collection, analysis, and submission processes. Furthermore, there is a growing trend for sponsors to consolidate data management in-house, aiming for greater control and transparency over their medical data, which is relevant to the methodologies employed in post-market studies.
Comprehending these phases is crucial for enhancing medical device clinical trial design and implementation, ultimately aiding the effective progression of medical innovations from concept to market. With bioaccess® as your vetted CRO and consulting partner in Colombia, you can navigate these complexities with confidence.
Regulatory Frameworks and Compliance in Medical Device Trials
Navigating the regulatory landscape is paramount for the success of medical device clinical trial design. Regulatory agencies such as the FDA in the United States and the EMA in Europe establish extensive guidelines that govern the conduct of these studies. Following these regulations not only ensures ethical execution but also guarantees the reliability of the data generated.
In 2025, the FDA's approval rates for medical device assessments have shown a notable increase, reflecting a more streamlined review process that emphasizes safety and efficacy. According to the FDA Adverse Event Reporting System (FAERS), supported by over 150 countries, the global implications of these regulations are significant, highlighting the importance of compliance on an international scale.
Key components of regulatory compliance include:
- Obtaining necessary approvals
- Adhering to Good Clinical Practice (GCP) standards
- Maintaining meticulous documentation throughout the study process
bioaccess provides extensive research study management services that encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Study setup
- Import permits
- Project management
- Reporting
The FDA's recent Cybersecurity Guidance mandates that manufacturers develop a robust cybersecurity risk management plan, while the EU MDR requires rigorous testing of connected product security throughout the product lifecycle.
These advancements underscore the rising significance of cybersecurity in medical studies, particularly as medical devices become more interconnected. Additionally, AI can streamline compliance processes, enhance analytics-driven strategies, and improve fraud detection in health plans, further emphasizing the role of technology in regulatory adherence. Moreover, case studies on emerging regulations for cell and gene therapies illustrate the evolving nature of regulatory frameworks. These therapies have prompted regulators to create expedited pathways for promising innovations, thereby enhancing the approval process for advanced therapies.
Grasping these guidelines is crucial for reducing risks and enabling smoother execution in medical device clinical trial design, ultimately resulting in successful navigation of FDA and EMA regulations in research studies. Data suggest that effective regulatory adherence can significantly minimize delays in studies and enhance overall results, establishing it as a vital priority for research leaders in the Medtech field. With specialists like Ana Criado, Director of Regulatory Affairs at Mahu Pharma, and Katherine Ruiz, an authority in Regulatory Affairs for Medical Devices and In Vitro Diagnostics in Colombia, bioaccess is well-prepared to assist research directors in achieving compliance and enhancing study success.
Key Considerations in Designing Medical Device Clinical Trials
Creating a clinical evaluation for a medical device necessitates careful thought regarding several fundamental components that can significantly influence the success of the research.
Medical device clinical trial design: The selection of experimental design—whether a randomized controlled experiment, observational research, or another format—plays a pivotal role in effectively addressing the research question. Selecting the right design not only aligns with the study's objectives but also enhances the credibility of the findings.
At bioaccess®, we leverage over 20 years of Medtech expertise to guide you in selecting the most appropriate medical device clinical trial design tailored to your specific needs.
Endpoint Definitions: Precise definitions of primary and secondary endpoints are essential for measuring outcomes that accurately reflect the performance of the instrument. For instance, in 2025, examples of endpoint definitions may include specific metrics such as device efficacy, safety profiles, and patient-reported outcomes.
Clear endpoint definitions facilitate better data interpretation and regulatory compliance, ensuring that your study meets the rigorous standards set by authorities like INVIMA, Colombia's National Food and Drug Surveillance Institute.
Statistical Methods: Utilizing robust statistical techniques is crucial for analyzing experimental data and deriving meaningful conclusions. The application of appropriate statistical methods ensures that the results are valid and dependable, which is especially crucial in the context of medical equipment where regulatory scrutiny is high.
In 2025, advancements in statistical methodologies are expected to further enhance the analysis of clinical study data, a focus area for bioaccess® as we strive to optimize your study outcomes.
Key Considerations: When developing medical device clinical trial design, several key considerations must be taken into account, including patient population selection, sample size determination, and the regulatory landscape. These factors can affect the study's feasibility and the generalizability of its results.
Our comprehensive services for medical device clinical trial design encompass feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting, ensuring a thorough approach to your study.
Expert Advice: Engaging with specialists in the field can provide invaluable insights into best practices for study design. For example, Danish Mairaj, a principal engineer of medical device design, emphasizes the importance of implementing relevant use cases in facilities to leverage data effectively.
This method can result in better results and more effective research processes. Furthermore, timely data entry is essential; for example, 45% of Alcon's data is recorded on the same day as the visit date, highlighting the significance of real-time data management in improving study quality.
Case Studies: Examining case studies on medical device clinical trial design selection can provide practical examples of effective strategies in medical equipment studies.
For example, the U.S. medical apparatus research market produced a revenue of USD 6,335.0 million in 2024, with forecasts suggesting an increase to USD 8,678.4 million by 2030. This growth reflects a compound annual growth rate (CAGR) of 5.6% from 2025 to 2030, highlighting the increasing significance of well-structured studies in the Medtech sector. Effective experimental design not only contributes to the success of individual investigations but also plays a significant role in the overall market leadership and growth of the medical equipment industry, fostering job creation and economic development in local economies.
By focusing on these critical aspects, researchers can optimize their experimental designs, ultimately leading to faster approvals and reduced costs while ensuring high-quality data that supports the advancement of innovative medical equipment. Rely on bioaccess® to guide your research projects towards success.
Overcoming Challenges in Clinical Trial Execution
Conducting research studies for medical instruments presents various challenges that can significantly impact the success of the investigation. Key areas of concern include participant recruitment, retention, and protocol complexity.
Participant Recruitment: Identifying and enrolling suitable participants can be particularly challenging, especially for niche medical devices. To enhance recruitment efforts, strategies such as targeted outreach, leveraging social media platforms, and engaging with local healthcare communities are essential. Research indicates that 80% of internet users actively search for health information online, underscoring the importance of a robust online presence to attract potential participants. bioaccess® is dedicated to innovative solutions for recruitment and retention, ensuring efficient clinical research processes, particularly in collaboration with local institutions like Caribbean Health Group. This partnership aims to position Barranquilla as a leading destination for clinical studies in Latin America, supported by Colombia's Minister of Health.
Retention of Participants: Maintaining participant engagement throughout the study is critical for ensuring data integrity and achieving reliable outcomes. Effective retention strategies include personalized communication tailored to individual participants, flexible monitoring options, and the provision of incentives. By reducing the burden of participation and fostering a sense of community, researchers can cultivate loyalty and commitment among participants. Examples have demonstrated that implementing these strategies can result in significantly higher retention rates. For instance, the collaboration between bioaccess™ and GlobalCare Clinical Trials has achieved over a 50% reduction in recruitment time and 95% retention rates, demonstrating the effectiveness of these approaches. Furthermore, satisfied patients are more involved and likely to finish clinical studies, becoming advocates for future medical research.
Protocol Complexity: The complexity of study protocols can create confusion and hinder compliance among both participants and investigators. Simplifying protocols where feasible can enhance understanding and adherence, thereby improving overall execution. Tackling these challenges in advance not only simplifies the process but also enhances the dependability of the outcomes.
By concentrating on these essential areas—recruitment, retention, and protocol management—researchers can navigate the intricacies of medical device clinical trial design more effectively. This approach results in successful outcomes and advancements in medical technology. bioaccess® specializes in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), backed by over 20 years of experience in Medtech.
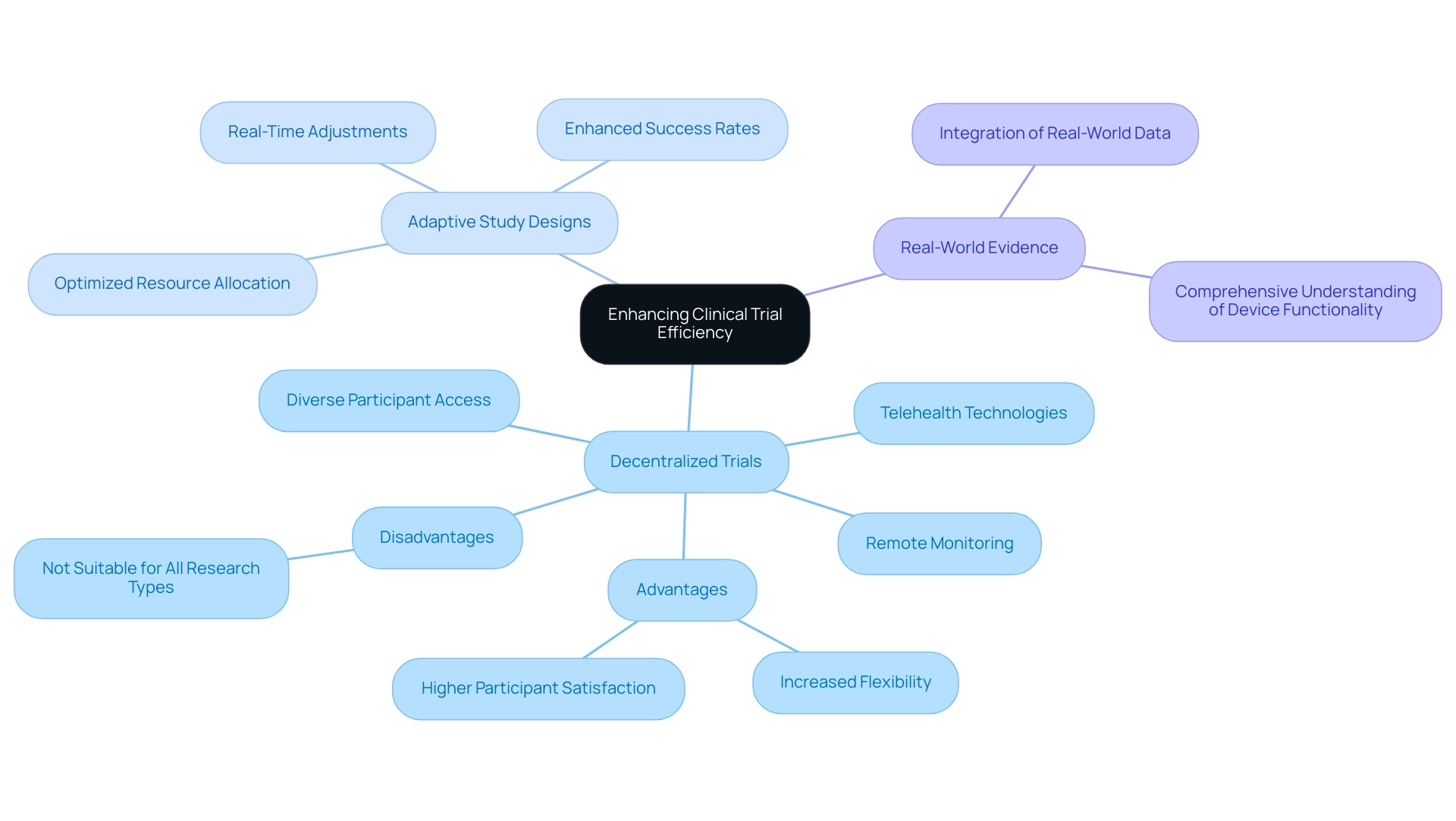
Innovative Approaches to Enhance Clinical Trial Efficiency
To enhance the efficiency of clinical trials, several innovative approaches can be employed:
-
Decentralized Trials: The adoption of remote monitoring and telehealth technologies has revolutionized participant engagement and data collection. This model not only increases flexibility but also broadens access to diverse populations, which is crucial for obtaining representative data. The popularity of decentralized studies surged following the COVID-19 pandemic, demonstrating their effectiveness in quickly recruiting and enrolling participants while maintaining high satisfaction levels. As highlighted by Ravindran et al., "Overall, this decentralized research demonstrated the capability to swiftly recruit, enroll, and complete the clinical investigation with a diverse and satisfied participant pool." However, it is important to consider both the advantages and disadvantages of this model within medical device clinical trial design, as it may not be suitable for all types of research.
-
Adaptive Study Designs: These designs allow for modifications to the protocol based on interim results, fostering a more responsive and efficient study execution. By enabling real-time adjustments, adaptive studies can enhance success rates and optimize resource allocation, ultimately leading to faster and more reliable outcomes.
-
Real-World Evidence: Integrating real-world information into medical studies offers valuable perspectives on equipment effectiveness and patient results. This approach enhances the findings of the study, providing a more comprehensive understanding of how medical devices function in everyday settings.
By adopting these innovations, bioaccess® connects innovative medtech companies with the opportunity for medical device clinical trial design research projects in Latin America. With more than 20 years of experience in Medtech and extensive management services for studies—including feasibility assessments, site selection, compliance evaluations, setup, import permits, project management, and reporting—bioaccess® significantly simplifies the process. Additionally, its partnership with Caribbean Health Group establishes Barranquilla as a prominent location for medical studies in Latin America, backed by Colombia's Minister of Health.
This strategic partnership improves the trial landscape, allowing U.S. medical equipment companies to utilize bioaccess® as a vetted CRO and consulting ally, ultimately achieving over 50% reduction in recruitment time and 95% retention rates. Additionally, bioaccess® specializes in managing Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH), ensuring a robust approach to clinical research.
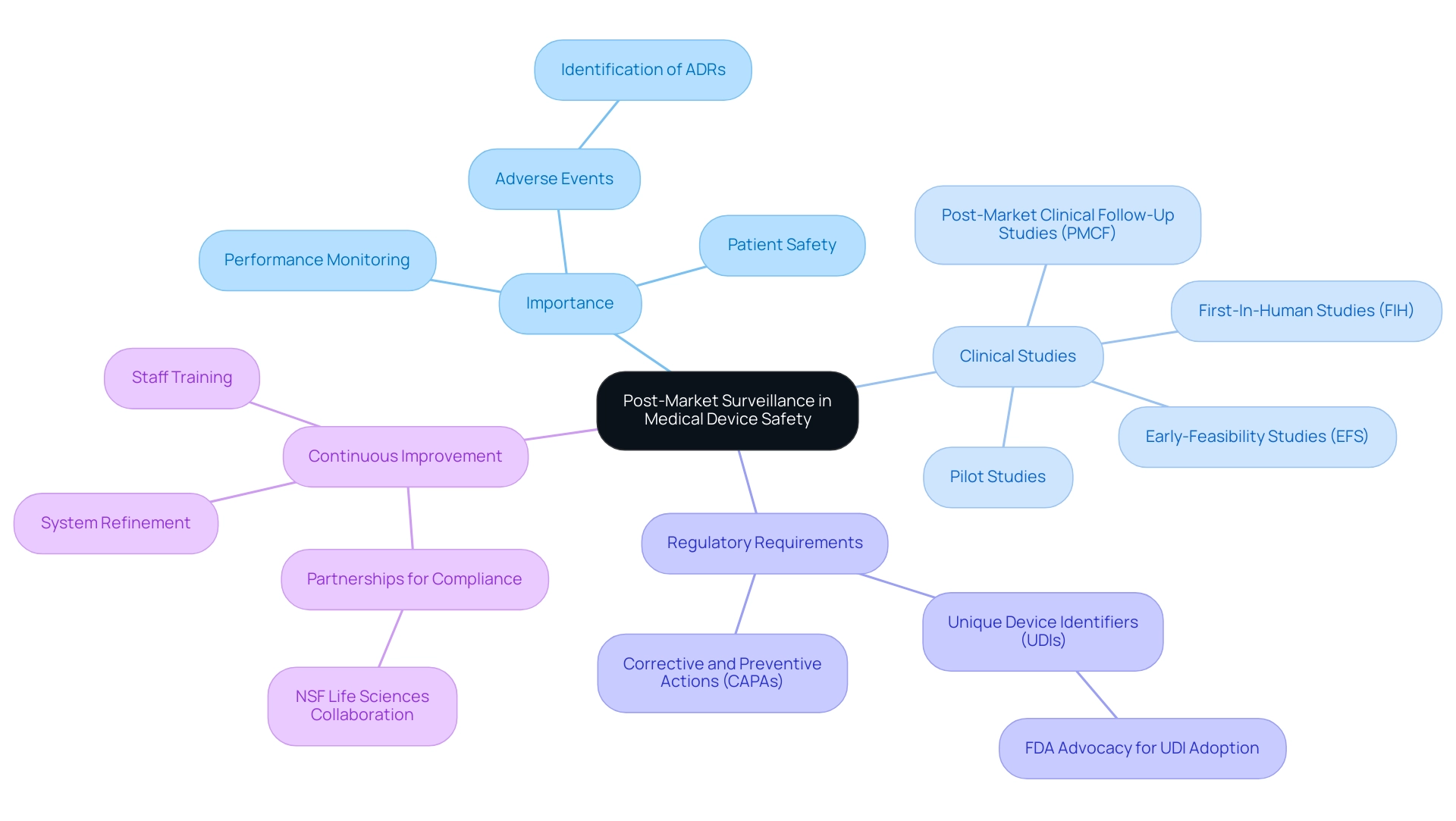
The Role of Post-Market Surveillance in Medical Device Safety
Post-market surveillance is an essential element of the medical product lifecycle, playing a critical role in ensuring that products remain safe and effective after reaching the market. This ongoing process necessitates meticulous performance monitoring, systematic data collection on adverse events, and comprehensive assessment of long-term outcomes. Regulatory authorities mandate that manufacturers conduct post-market research to gather real-world evidence regarding the safety and effectiveness of their products.
At bioaccess®, we excel in managing a variety of clinical studies, including:
- Medical Device Clinical Trial Design for Post-Market Clinical Follow-Up Studies (PMCF)
- Early-Feasibility Studies (EFS)
- First-In-Human Studies (FIH)
- Pilot Studies
as part of our extensive clinical trial management services in Latin America. Our team leverages over 20 years of Medtech expertise to ensure that your products are continuously monitored and evaluated, in alignment with regulatory requirements, thereby enhancing patient safety.
The significance of post-market surveillance is further emphasized by recent statistics indicating the growing integration of artificial intelligence and machine learning in analyzing post-market surveillance data, which enhances the ability to detect and respond to potential safety issues. Additionally, the FDA has acknowledged the limited adoption of Unique Device Identifiers (UDIs) as a barrier to effective surveillance. In response, the agency is actively advocating for the inclusion of UDIs in health records and billing claims, which is anticipated to improve tracking and safety evaluations.
This initiative exemplifies the evolving regulatory efforts aimed at strengthening post-market surveillance.
Regular training for staff on post-market surveillance procedures is crucial for effective implementation. Continuous refinement of these systems in response to new insights and regulatory changes is equally vital. For instance, a recent partnership between NSF Life Sciences Pharma Biotech and leading pharmaceutical and biotechnology companies aims to provide expert consulting and training services to enhance regulatory compliance and operational excellence.
This collaboration underscores the importance of ongoing education and support in the realm of post-market surveillance.
The importance of post-market studies cannot be overstated; they are instrumental in identifying adverse events and ensuring efficacy. As Meri Beckwith, Co-Founder, articulates, "One of the crucial roles of post-market surveillance is the identification of adverse drug reactions (ADRs)," which can significantly impact patient safety. By implementing robust post-market surveillance systems, manufacturers can proactively identify potential issues, enabling timely corrective and preventive actions (CAPAs) to address quality concerns and prevent recurrence, as outlined in CFR 21 820.100.
This proactive approach not only safeguards patients but also reinforces the credibility and reliability of medical devices in the marketplace.
Conclusion
The multifaceted world of clinical trials is integral to the advancement of medical device development, providing a framework for assessing the safety and efficacy of innovations that ultimately benefit patients. From early-feasibility studies to post-market surveillance, each phase is crucial in refining devices and ensuring they meet the rigorous standards set by regulatory bodies. As the biopharma industry navigates the complexities of crowded clinical markets, understanding and optimizing these trials becomes paramount.
The challenges faced in clinical trial execution, such as participant recruitment and protocol complexity, underscore the necessity for innovative strategies to enhance efficiency and data integrity. Embracing decentralized trials, adaptive designs, and real-world evidence can significantly improve participant engagement and streamline processes. Furthermore, the role of post-market surveillance is vital, ensuring that devices remain safe and effective in real-world settings, thereby fostering trust among stakeholders.
In conclusion, the journey from concept to market for medical devices is paved with the rigorous demands of clinical trials. By prioritizing robust trial designs, adhering to regulatory frameworks, and embracing innovative approaches, the industry can navigate these complexities effectively. This commitment not only advances healthcare solutions but also reinforces the essential role of clinical trials in safeguarding patient health and enhancing the credibility of medical devices in the marketplace.




