Overview
The primary objective of this article is to deliver a comprehensive understanding of the clinical trial requirements for Medtech in Brazil, with a keen emphasis on the regulatory frameworks and recent changes that bolster research initiatives.
By detailing the significance of compliance with regulations established by ANVISA and CONEP, alongside recent legislative updates, the article illustrates how these factors enhance the efficiency of clinical trials.
This positions Brazil as an appealing landscape for Medtech innovation and development, underscoring the vital role of collaboration in advancing the field.
Introduction
Brazil is rapidly emerging as a pivotal player in the global landscape of clinical trials, driven by a combination of regulatory advancements and a commitment to ethical research practices.
With the National Health Surveillance Agency (ANVISA) and the National Research Ethics Commission (CONEP) leading the way, the regulatory framework is meticulously designed to safeguard participant welfare while fostering innovation in the Medtech sector.
The recent enactment of laws aimed at streamlining the approval process has opened new avenues for Medtech startups, enabling them to navigate the complexities of clinical trials with greater ease.
As the country continues to evolve its regulatory environment, understanding the intricacies of conducting clinical trials in Brazil becomes essential for companies seeking to leverage its diverse patient population and robust healthcare infrastructure.
This article delves into the key regulations, challenges, and opportunities that define Brazil's clinical trial landscape, offering insights for Medtech companies aiming to thrive in this dynamic market.
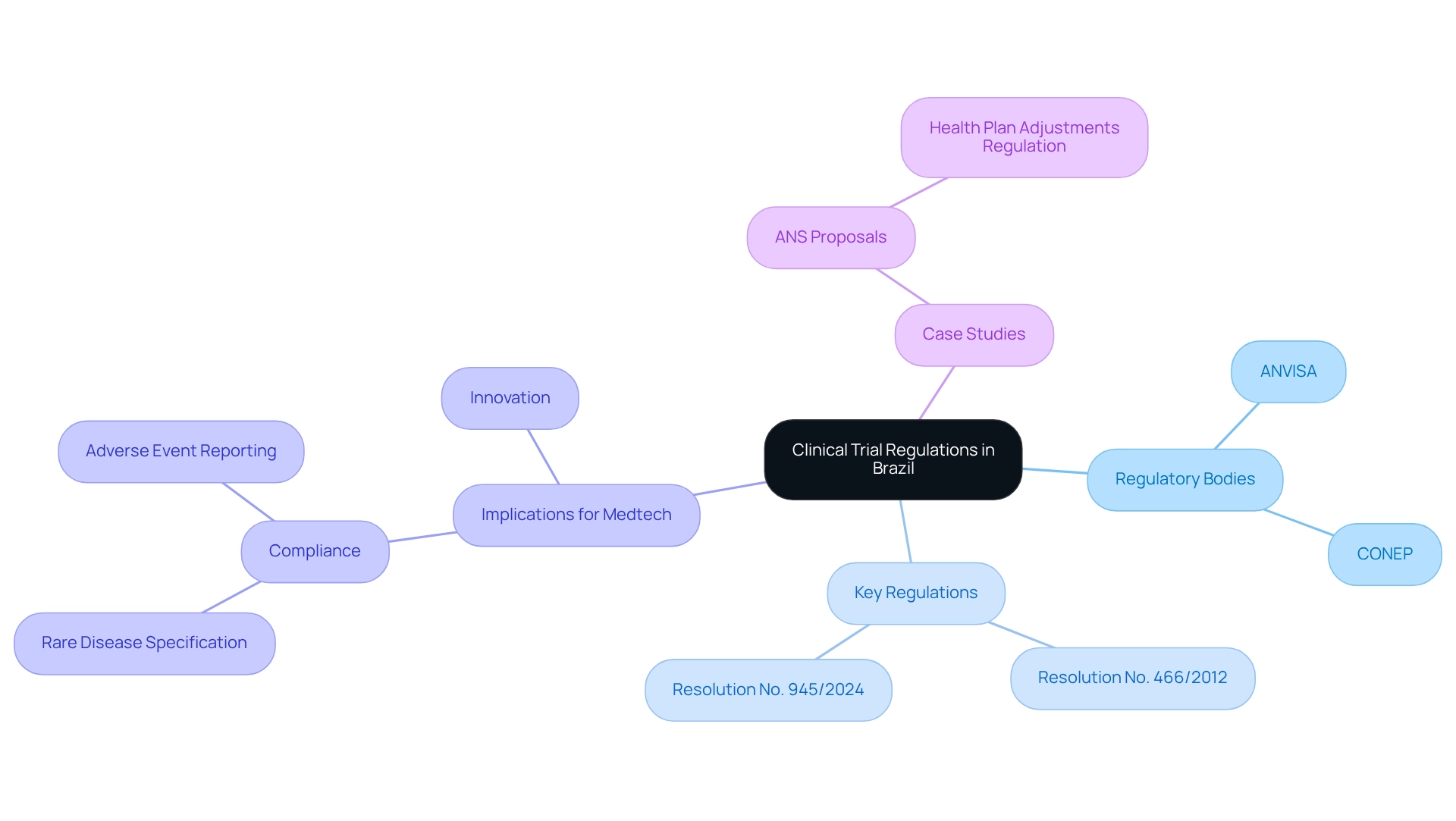
Overview of Clinical Trial Regulations in Brazil
Brazil's research regulations are primarily overseen by the National Health Surveillance Agency (ANVISA) and the National Research Ethics Commission (CONEP). These regulatory organizations play a crucial role in supervising the authorization and assessment of research studies, ensuring participant safety and the integrity of the data collected. A cornerstone of these regulations is Resolution No. 466/2012, which establishes ethical principles for research involving human subjects. Recent updates under Resolution No. 945/2024 have further streamlined the approval process, enhancing efficiency for Medtech firms initiating research studies.
In 2025, it is imperative for Medtech companies to stay informed about the clinical trial requirements in Brazil to ensure compliance and facilitate smoother testing operations. Notably, ANVISA has implemented an online system for reporting unexpected serious adverse events related to drugs and vaccines, significantly enhancing transparency and safety in medical research. Moreover, the approval procedure requires sponsors to clearly specify whether their drug development pertains to a rare disease, showcasing a tailored approach to oversight that is essential for regulatory adherence.
The evolving landscape of medical research regulations in the country is also illustrated by recent case studies, such as the proposed modifications by the National Supplementary Health Agency (ANS) aimed at refining health plan adjustments regulation. These changes are designed to mitigate conflicts and achieve financial sustainability for operators while protecting consumers, highlighting the dynamic nature of healthcare regulation in Brazil. The proposals seek to establish a clearer framework that benefits both consumers and healthcare providers.
Expert opinions underscore the vital roles of ANVISA and CONEP in ensuring that clinical trial requirements in Brazil for Medtech are met, while also supporting the advancement of medical technology. As Anderson Ribeiro from Souto Correa highlights, "Our Life Sciences & Healthcare team is available if you have any questions about these topics and their possible developments via email: lifesciences@soutocorrea.com.br." Successful medical studies conducted under the supervision of these agencies showcase Brazil's potential as a viable site for Medtech innovation and development, reinforcing the importance of clinical trial requirements for Medtech and solidifying its position in the international medical research arena.
At bioaccess®, we specialize in navigating these complex regulatory environments, offering comprehensive services that encompass feasibility studies, investigator selection, project management, and reporting study status and adverse events. Our commitment to data protection ensures that client concerns are addressed with compliance and transparency, fostering trust in our processes. As a prominent CRO in Latin America, bioaccess® is dedicated to expediting research services for medical devices by addressing the clinical trial requirements in Brazil for Medtech, assisting startups in overcoming regulatory challenges and achieving successful outcomes.
For any grievances or data protection inquiries, clients can reach out to our Grievance Officer at info@bioaccessla.com.
Key Requirements for Conducting Clinical Trials in Brazil
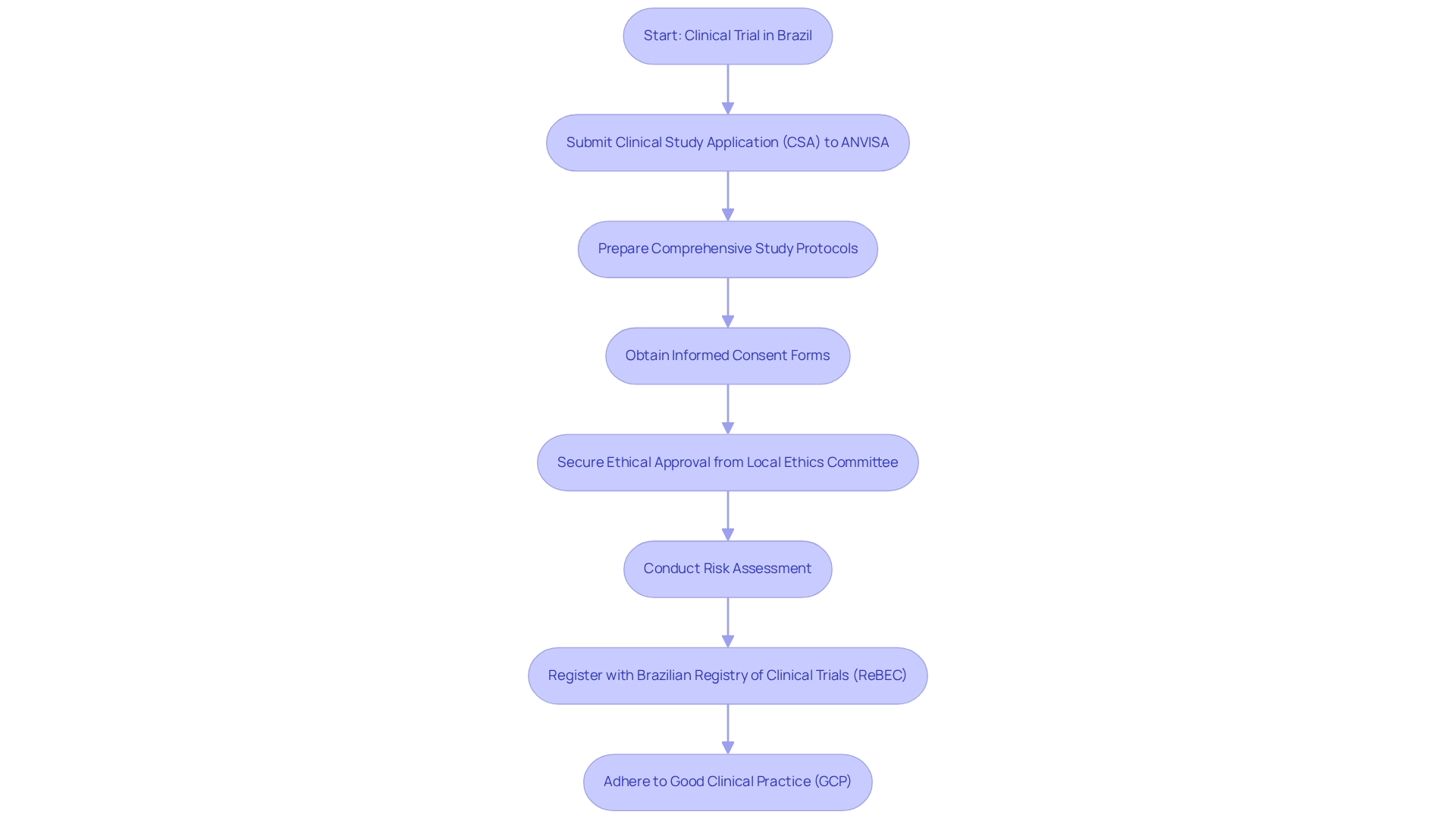
To commence clinical studies in Brazil, Medtech firms must adhere to the clinical trial requirements by presenting a Clinical Study Application (CSA) to ANVISA, the national health regulatory body. This application necessitates comprehensive study protocols, informed consent forms, and proof of ethical approval from a local ethics committee. Adhering to Good Clinical Practice (GCP) guidelines is essential, ensuring the integrity of the study and the safety of participants.
In Brazil, adherence to GCP is evidenced by a compliance rate exceeding 90%, in accordance with the ICH E6 R2 requirement that sponsors oversee risk to lessen the burden on sites, signifying a strong commitment to ethical practices.
A thorough risk assessment must accompany the CTA, addressing potential challenges and outlining mitigation strategies. Additionally, registration of the trial with the Brazilian Registry of Clinical Trials (ReBEC) is critical, promoting transparency and fostering public trust in the research process.
Recent case studies underscore distinct features of the CTA process in Brazil. For instance, a comparative analysis of dossier requirements in MIST countries, including Brazil, revealed that while Brazil aligns with ICH standards, it imposes additional stringent requirements, particularly in environmental impact assessments and risk management plans. This complexity can affect approval timelines, making it crucial for companies to be well-prepared.
Understanding the clinical trial requirements in Brazil for Medtech can significantly influence the efficiency of the approval process.
As we look to 2025, the regulatory landscape continues to evolve, with ANVISA emphasizing collaboration among stakeholders to ensure that new medical products benefit all patients, regardless of their background. Successful submissions to ANVISA often showcase meticulous attention to detail and a proactive approach to addressing the clinical trial requirements in Brazil for Medtech, setting a benchmark for future applications. As noted by Ibrahim Kamstrup-Akkaoui, Vice President of Data Systems Innovation, "We did a small AI initiative to see if we can generate meaningful test data for setting up and validating our systems. It turns out we can." This highlights the significance of utilizing technology in the Medtech industry to enhance the research process. Furthermore, the Common Technical Document (CTD) serves as a standardized format for regulatory applications, providing a structured approach that can facilitate the submission process to ANVISA.
At bioaccess®, we focus on extensive research study management services, including feasibility assessments, site selection, compliance evaluations, study setup, import permits, project oversight, and reporting. Our expertise in Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies establishes us as a prominent contract research organization enabling medical device studies in Latin America. By navigating the complexities of the regulatory landscape, we contribute to job creation, economic growth, healthcare improvement, and international collaboration, ultimately driving global health improvement through innovation in Medtech.
Recent Regulatory Changes Affecting Medtech Startups
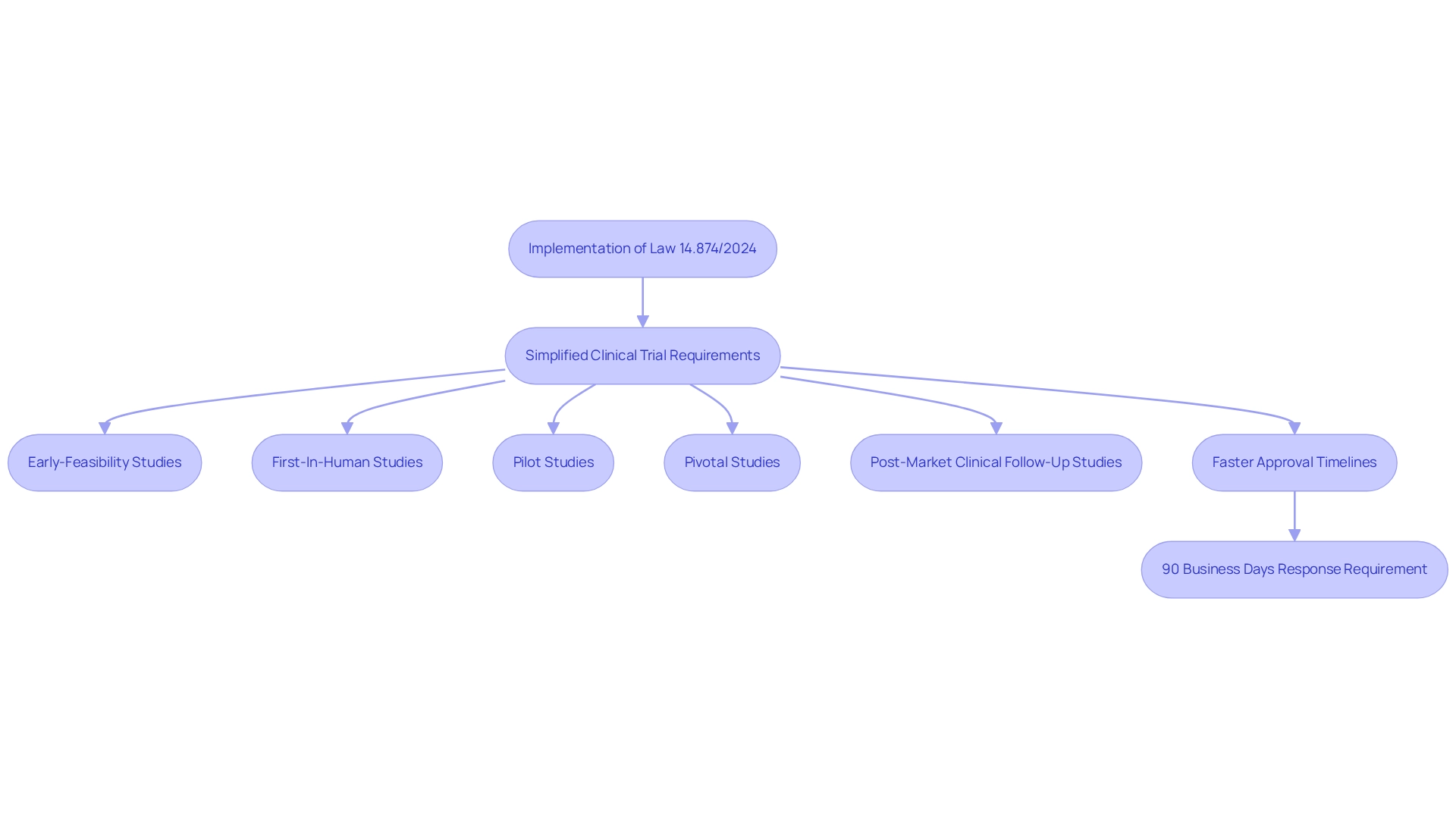
Recent regulatory modifications, particularly the implementation of Law 14.874/2024, have significantly simplified the clinical trial requirements in Brazil for medtech research studies. This crucial legislation reduces bureaucratic obstacles by allowing local ethics boards to authorize research protocols without the need for national committee review, thereby accelerating the timeline for study initiation. Consequently, Medtech startups can anticipate a more efficient pathway to market their innovations, particularly when collaborating with experts like bioaccess®, who specialize in managing:
- Early-Feasibility Studies
- First-In-Human Studies
- Pilot Studies
- Pivotal Studies
- Post-Market Clinical Follow-Up Studies
With over 20 years of expertise in Medtech, bioaccess® offers a tailored strategy that ensures effective management designed to meet each client's unique requirements.
The new regulations also promote enhanced collaboration between regulatory bodies and Medtech companies, fostering a more supportive environment for innovation. For instance, the Brazilian Regulatory Authority is now mandated to respond to research study applications within 90 business days, a change expected to attract both local and international pharmaceutical and biotech investments. This shift is particularly significant, given that of the 33 new molecular entities (NMEs) tested in Latin America, only 5 demonstrated therapeutic value for specific patient subsets.
The streamlined approval process under Law 14.874/2024 is anticipated to improve these outcomes by facilitating quicker access to trials for potentially valuable therapies.
Startups should strategically leverage these regulatory advancements to expedite their clinical development processes. Expert opinions highlight that establishing clear timelines for regulatory responsibilities is vital to avoid hindering innovation and development, especially for groundbreaking medications targeting serious and rare illnesses. As Hiran Goncalves noted, "Setting a time limit on this obligation is crucial to prevent the Government from further discouraging research and the development of innovative drugs, particularly those targeting serious and rare diseases."
The execution of Law 14.874/2024 is expected to enhance the country's attractiveness as a hub for medical studies, specifically addressing the clinical trial requirements in Brazil for medtech, ultimately promoting greater innovation within the sector.
Moreover, upholding the veto on indefinite supply obligations is a critical factor, as it may hinder the country's ability to attract more research studies. The case study titled 'The Effect Of the Brazilian Clinical Research Law 14.874/24' illustrates the anticipated outcomes of this law, underscoring its potential to enhance the country's appeal for conducting medical studies and highlighting the clinical trial requirements in Brazil for medtech, while encouraging both domestic and international investments in the sector, particularly through comprehensive research management services provided by bioaccess®. For further details on how bioaccess® can assist with your research requirements, please contact us.
Advantages of Conducting Clinical Trials in Brazil
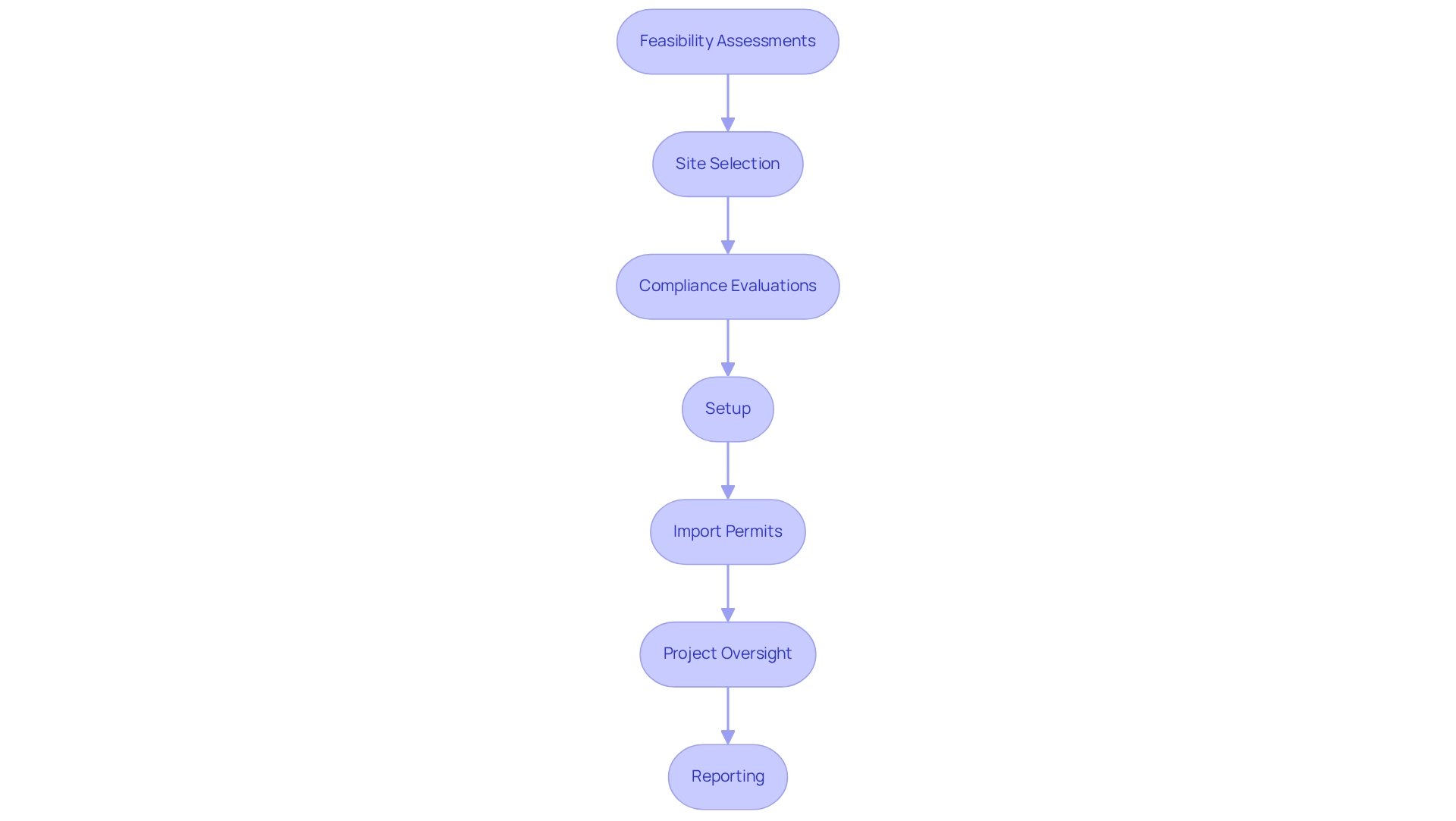
Brazil presents an attractive environment for conducting research, particularly in fulfilling the clinical trial requirements in Brazil for medtech. Its extensive and diverse patient base significantly enhances the applicability of study findings. With a literacy rate of 90.4%, the population demonstrates an increasing awareness of healthcare options, which is critical for patient recruitment and engagement in health studies. The nation boasts a robust healthcare system, supported by a growing network of research organizations that streamline study management processes, including:
- feasibility assessments
- site selection
- compliance evaluations
- setup
- import permits
- project oversight
- reporting
Recent regulatory changes have further refined the study approval process, addressing the clinical trial requirements in Brazil for medtech and improving efficiency, thereby reducing the time to market for new Medtech innovations. These adjustments are particularly beneficial for global Medtech companies seeking to navigate the clinical trial requirements in Brazil for medtech and leverage the unique advantages offered by the country. For instance, a recent survey of Brazilian oncologists revealed that heightened public awareness about research studies could significantly bolster recruitment rates, addressing a major barrier to participation.
As highlighted by Hoffmann-La Roche Ltd, increasing public awareness is essential for fostering involvement in medical research. Moreover, the patient population in Brazil is characterized by its diversity, which is crucial for studies aiming to encompass a wide range of demographics and health conditions. This diversity not only enriches the data collected but also ensures that the findings are relevant to various populations.
Nonetheless, challenges such as linguistic, cultural, and socio-economic barriers must be tackled to enhance patient protection and research quality. With bioaccess®'s expertise in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies, along with over 20 years of experience in the Medtech field, Brazil stands out as an optimal location for executing research studies. The combination of a supportive regulatory framework, a diverse patient population, and a well-established healthcare system collectively fulfills the clinical trial requirements in Brazil for medtech, facilitating successful trial outcomes while contributing to job creation, economic growth, and healthcare improvement in the region.
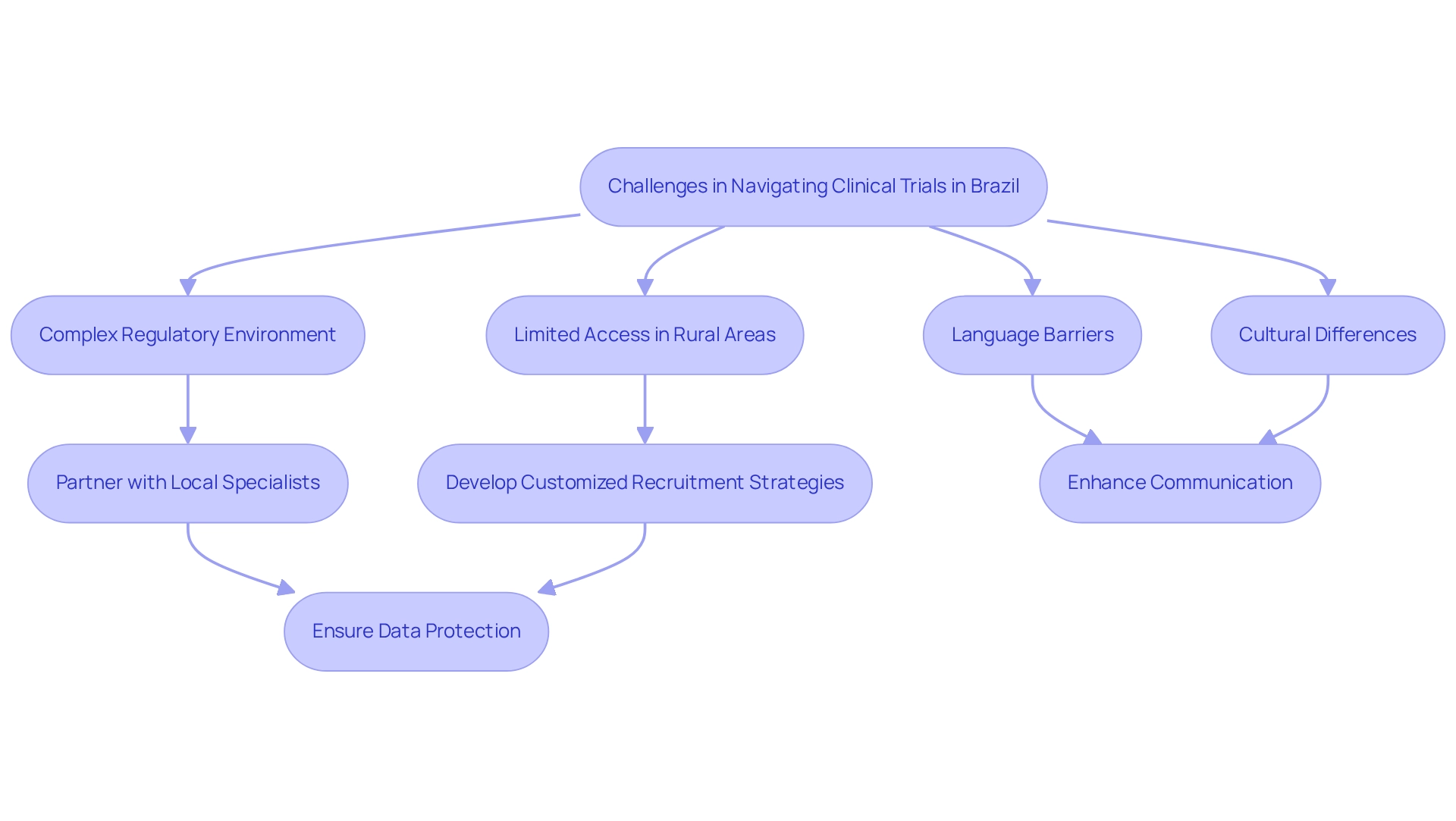
Challenges in Navigating Brazil's Clinical Trial Landscape
Carrying out medical studies in Brazil presents considerable benefits due to the area's diverse patient demographic and increasing healthcare funding. However, it also poses notable challenges. Navigating the complex regulatory environment, particularly the clinical trial requirements for medtech, is paramount. Regulations can differ significantly across states and municipalities, creating a patchwork of requirements that can be daunting for Medtech companies.
As we approach 2025, these challenges are compounded by the need for compliance with evolving regulations aimed at enhancing patient safety and data integrity. Language barriers and cultural differences further complicate communication with local stakeholders, which can hinder the recruitment process. As Carlos H Barrios notes, addressing these language barriers is crucial for building trust with participants and stakeholders alike. Effective patient recruitment strategies are essential, particularly in rural areas where access to healthcare may be limited.
Statistics indicate that investments in lower-middle-income nations, including Brazil, have risen by 4% to 16% from 2005 to 2012. This emphasizes the potential for expansion in medical research while highlighting the necessity for customized recruitment strategies.
To tackle these challenges, Medtech companies should consider partnering with local specialists and contract research organizations (CROs) like bioaccess®, which is dedicated to supporting medical device research in Latin America. bioaccess® aims to advance medical devices sooner through its expertise and customized approach. Involving these specialists can promote smoother testing operations and improve patient recruitment efforts.
Successful case studies, such as the partnership between bioaccess™ and Caribbean Health Group to establish Barranquilla as a prominent research location, illustrate how utilizing local expertise can significantly enhance recruitment rates and retention, ultimately resulting in more effective study timelines. This initiative, supported by Colombia's Minister of Health, underscores the importance of strategic partnerships in navigating the Medtech landscape.
Additionally, ensuring information security and client trust is paramount. bioaccess® is committed to data protection in medical device studies, with established grievance and data protection procedures to address client concerns regarding compliance and transparency.
By prioritizing these strategies and remaining informed about the clinical trial requirements for medtech in Brazil, firms can navigate the intricate regulatory environment more effectively, ensuring that their research studies are not only compliant but also successful in achieving their goals.
Ethical Considerations in Brazilian Clinical Trials
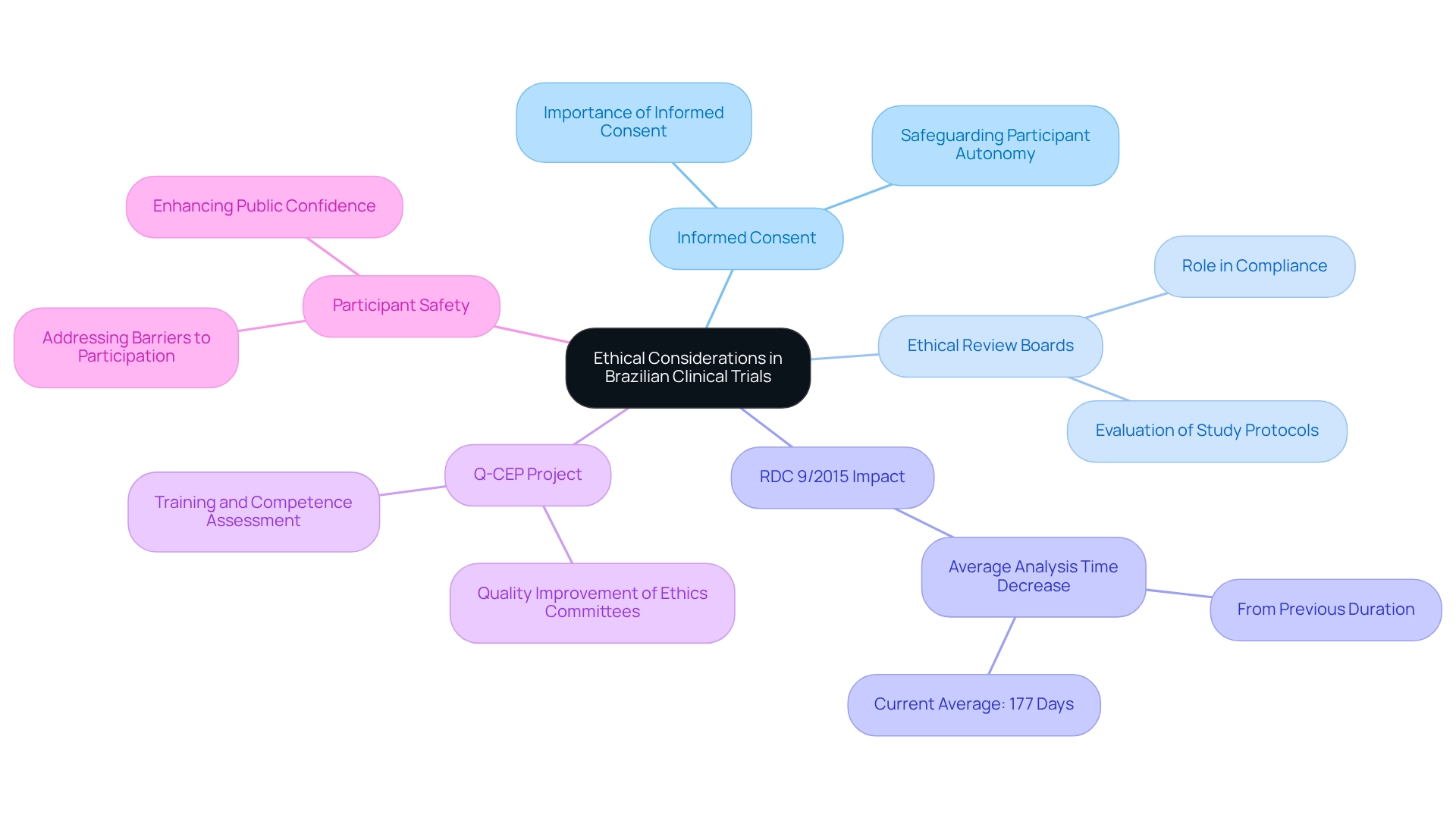
Ethical considerations are paramount in the execution of research studies in Brazil, deeply influenced by the principles outlined in the WMA Declaration of Helsinki and reinforced by local regulations. Informed consent is a cornerstone of this ethical framework, requiring that participants are comprehensively informed about the potential risks and benefits associated with their involvement in a study. This process not only safeguards participant autonomy but also enhances the trustworthiness of the study.
The role of ethical review boards is crucial, as they are responsible for scrutinizing study protocols to ensure compliance with ethical standards. These boards evaluate the adequacy of informed consent processes, ensuring that participants are not only informed but also fully understand the implications of their participation. Recent statistics reveal that following the implementation of RDC 9/2015, the average analysis time by Anvisa has significantly decreased to 177 days, indicating a more efficient regulatory environment that continues to prioritize ethical oversight.
Moreover, the ethical landscape in Brazilian medical studies is evolving, particularly in light of recent investigations that underscore obstacles to participation in trials. Addressing these obstacles is essential for enhancing participant safety and fostering public confidence in clinical studies. The Q-CEP Project exemplifies efforts to elevate the quality of ethics committees in the country, providing training and establishing a framework for continuous education.
This initiative has effectively assessed and improved the competencies of all 836 active ethics committees, thereby reinforcing the ethical review process and ultimately contributing to better public health outcomes.
Informed consent practices in Brazil are not merely regulatory requirements but also reflect a commitment to ethical research. Organizations involved in studies, such as bioaccess®, must prioritize these ethical practices to ensure participant safety and uphold the integrity of their work, which is critical for regulatory compliance and sustaining public trust. Furthermore, bioaccess® is dedicated to ensuring information security and addressing client concerns through its grievance and data protection procedures.
If you have any queries or concerns regarding the processing of your information, you may contact our Grievance Officer at IMH ASSETS CORP (doing business as "bioaccess®"), located at 1200 Brickell Avenue, Suite 1950 #1034, or via email at info@bioaccessla.com. This commitment to transparency and adherence is vital in tackling the ongoing ethical crisis in Brazilian medical studies, necessitating prompt regulatory measures to enhance participant safety and maintain national sovereignty.
Collaborative Opportunities with Local Institutions
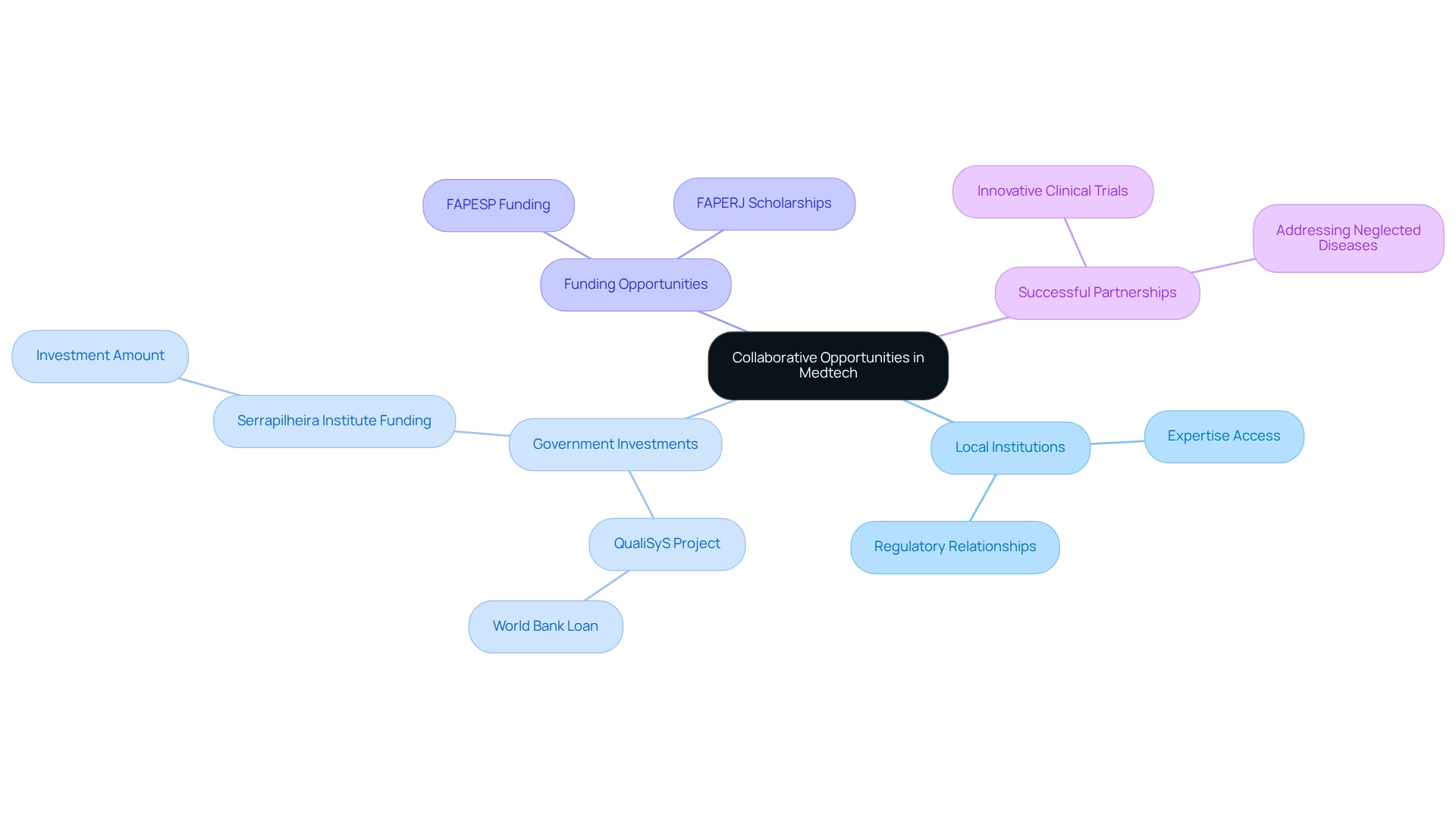
Collaborating with regional organizations, including universities and research hospitals, is paramount for the success of medical studies in Brazil. Such partnerships not only provide access to local expertise and diverse patient populations but also offer valuable resources that can optimize operations. For example, local institutions often have established relationships with regulatory bodies, significantly expediting the approval process for clinical trials.
The Brazilian government has made substantial investments in innovation and development, particularly within the Medtech sector, through initiatives designed to enhance drug development and medical research capabilities. This commitment is exemplified by the $235 million loan approved by the World Bank for the QualiSyS project, aimed at improving healthcare services within the Unified Health System (SUS). Additionally, the Serrapilheira Institute has allocated R$ 51,868,416.42 to various projects, fostering an environment conducive to collaborative studies among Medtech companies.
Successful partnerships have emerged between Medtech firms and Brazilian universities, resulting in innovative clinical trials that capitalize on local knowledge and infrastructure. These collaborations have been instrumental in addressing neglected diseases and advancing biotechnology. As Cristiano Mendes Gomes, MD, notes, "Collaborations, involving both local and international scholars, not only enhance funding opportunities but also increase the potential outcomes and significance of the study."
Statistics reveal a rising trend in collaboration between Medtech companies and local institutions, with numerous projects benefiting from support from key Brazilian research agencies such as FAPESP and FAPERJ. These organizations offer various funding opportunities and scholarships, further incentivizing collaborations that can lead to successful research outcomes.
In conclusion, Medtech firms like bioaccess® should proactively pursue partnerships with local organizations to leverage these advantages, thereby enhancing their chances of achieving successful testing results and advancing medical technologies that benefit patients. By employing comprehensive research study management services—including feasibility assessments, site selection, compliance evaluations, study setup, import permits, project management, and reporting—bioaccess® is strategically positioned to foster global health improvement through international collaboration and innovation in Medtech.
Future Trends in Clinical Trials for Medtech in Brazil
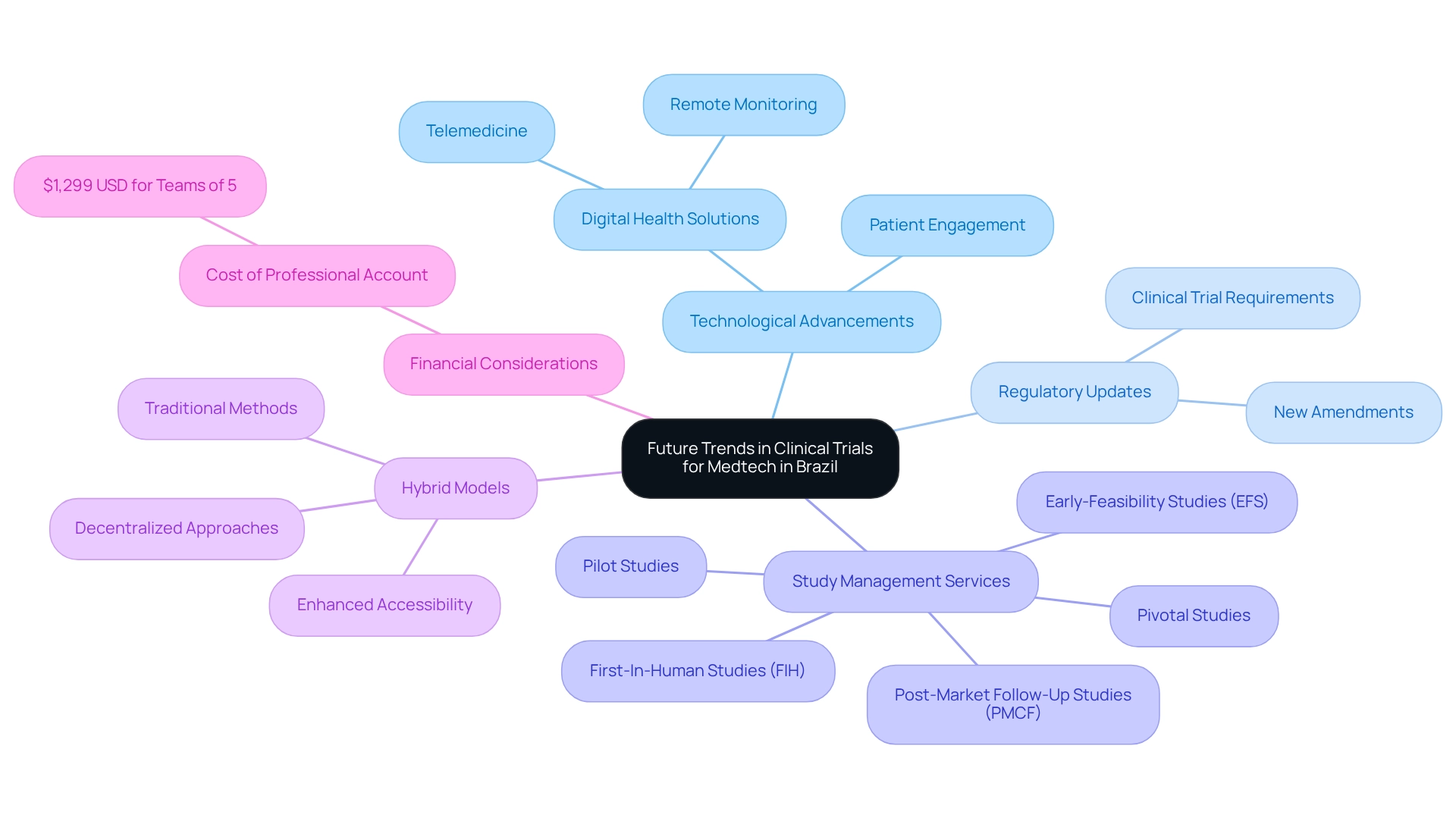
The landscape of research studies in Brazil is poised for a significant transformation, propelled by technological advancements and proactive regulatory updates addressing clinical trial requirements for medtech. The increasing integration of digital health solutions, such as telemedicine and remote monitoring, is expected to substantially enhance patient engagement and streamline data collection processes. These innovations not only improve the efficacy of experiments but also foster a more patient-centered approach, which is crucial in today's medical research environment.
Moreover, the Brazilian government is actively working to refine regulatory frameworks, including clinical trial requirements for medtech, thereby enhancing the nation’s attractiveness for global research initiatives. This trend is anticipated to accelerate as new regulatory modifications are introduced, aimed at enhancing research service offerings. Medtech firms, including bioaccess®, must remain vigilant and adapt to these evolving dynamics by incorporating innovative approaches into their study designs.
With over 20 years of experience, bioaccess® specializes in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF). They ensure comprehensive management services that encompass feasibility studies, site selection, compliance reviews, study setup, import permits, project management, and reporting.
As we approach 2025, the emphasis on hybrid models that blend decentralized and traditional methods is likely to gain traction. These models are predicted to enhance accessibility and participation, ultimately leading to more efficient and effective studies. A case study on hybrid trial models for improved accessibility illustrates this trend, demonstrating how such approaches can bolster patient engagement and outcomes.
Furthermore, the biotech sector faces significant business model challenges as medical markets become saturated and the introduction of new revolutionary drugs dwindles. In this context, the cost of a Professional Account for teams of up to five people, priced at $1,299 USD, underscores the financial considerations that medtech companies must navigate. Additionally, as noted by Peng Lu, chief medical officer of Dutch biotech Pharvaris, "Standardizing the use of specific outcomes and outcome measures for research studies will aid in the development of medical guidelines and future indirect comparisons among interventions."
Staying informed about these trends and leveraging digital health solutions will be critical for medtech companies, like bioaccess®, striving to navigate the clinical trial requirements in Brazil while ensuring information security and client trust through robust data protection measures. Should you have any queries or concerns regarding the processing of your information, please contact our Grievance Officer at IMH ASSETS CORP (doing business as "bioaccess®"), 1200 Brickell Avenue, Suite 1950 #1034, email: info@bioaccessla.com. We will address your concerns in accordance with applicable law.
Conclusion
The clinical trial landscape in Brazil presents a distinctive amalgamation of regulatory advancements, ethical considerations, and collaborative opportunities, establishing the country as a pivotal player in the global Medtech sector. Recent regulatory reforms, notably the enactment of Law 14.874/2024, have streamlined the approval process, allowing Medtech startups to navigate the complexities of clinical trials with increased efficiency. Regulatory bodies such as ANVISA and CONEP are committed to ensuring participant safety and data integrity, empowering companies to engage in clinical research with enhanced confidence.
Brazil's diverse patient population, along with a robust healthcare infrastructure, significantly enhances the generalizability of clinical trial results. Nevertheless, challenges such as navigating the intricate regulatory environment, addressing cultural differences, and ensuring ethical compliance must be confronted directly. By collaborating with local institutions and leveraging the expertise of experienced contract research organizations like bioaccess®, Medtech companies can surmount these obstacles and achieve successful trial outcomes.
Looking ahead, the integration of digital health solutions and hybrid trial models is poised to revolutionize clinical research in Brazil. These advancements will not only elevate patient engagement but also cultivate a more patient-centric approach to clinical trials. As the regulatory landscape continues to evolve, it will be essential for Medtech companies to remain informed and adaptable in order to thrive within this dynamic market.
In conclusion, Brazil presents a compelling landscape for conducting clinical trials, characterized by regulatory support, a diverse patient base, and abundant opportunities for innovation. By prioritizing ethical practices, engaging in strategic partnerships, and embracing emerging technologies, Medtech companies can leverage Brazil's potential to advance medical research and enhance patient outcomes on a global scale.
Frequently Asked Questions
What organizations oversee research regulations in Brazil?
Research regulations in Brazil are primarily overseen by the National Health Surveillance Agency (ANVISA) and the National Research Ethics Commission (CONEP).
What is the significance of Resolution No. 466/2012?
Resolution No. 466/2012 establishes ethical principles for research involving human subjects, ensuring participant safety and the integrity of the data collected.
What recent updates have been made to Brazil's research regulations?
Recent updates under Resolution No. 945/2024 have streamlined the approval process for research studies, enhancing efficiency for Medtech firms.
What must Medtech companies do to comply with clinical trial requirements in Brazil?
Medtech companies must present a Clinical Study Application (CSA) to ANVISA, including comprehensive study protocols, informed consent forms, and proof of ethical approval from a local ethics committee.
What is the compliance rate for Good Clinical Practice (GCP) in Brazil?
Brazil has a compliance rate exceeding 90% for Good Clinical Practice (GCP), demonstrating a strong commitment to ethical practices in clinical trials.
What additional requirements does Brazil impose on clinical trial applications?
Brazil imposes additional stringent requirements, particularly in environmental impact assessments and risk management plans, compared to ICH standards.
How does ANVISA enhance transparency in medical research?
ANVISA has implemented an online system for reporting unexpected serious adverse events related to drugs and vaccines, significantly enhancing transparency and safety.
What role do case studies play in understanding Brazil's clinical trial requirements?
Recent case studies illustrate distinct features of the Clinical Trial Application (CTA) process in Brazil and highlight the evolving regulatory landscape.
What is the Common Technical Document (CTD)?
The Common Technical Document (CTD) is a standardized format for regulatory applications that provides a structured approach to facilitate submissions to ANVISA.
How can bioaccess® assist Medtech companies in Brazil?
bioaccess® offers comprehensive services including feasibility studies, investigator selection, project management, and reporting, helping Medtech companies navigate regulatory challenges and achieve successful outcomes.

