Overview
The article examines the vital considerations and functions of ethics committees in Brazil, specifically their oversight of clinical research involving human subjects. These committees, established under national regulations, play an essential role in ensuring ethical compliance and protecting participant rights. They are also tasked with adapting to ongoing challenges, such as resource shortages and regulatory changes. This adaptability reinforces their critical role in maintaining the integrity of clinical studies in Brazil, highlighting the need for collaboration and proactive measures in the field.
Introduction
In Brazil, the landscape of clinical research is intricately woven with ethical considerations, primarily overseen by ethics committees known as Comitês de Ética em Pesquisa (CEPs). These committees, established to protect human subjects and ensure adherence to ethical standards, have become essential players in the research process.
With over 500 active CEPs committed to continuous improvement, they navigate the complexities of informed consent, participant rights, and the evolving regulatory environment.
As the medical research field grows, the collaboration between ethics committees and clinical research organizations emerges as a crucial element for fostering ethical practices and enhancing participant safety.
This article delves into the pivotal role of ethics committees in Brazil, exploring their structure, key guidelines, challenges faced, and the future trends that will shape their operations in an increasingly innovative landscape.
Overview of Ethics Committees in Brazil
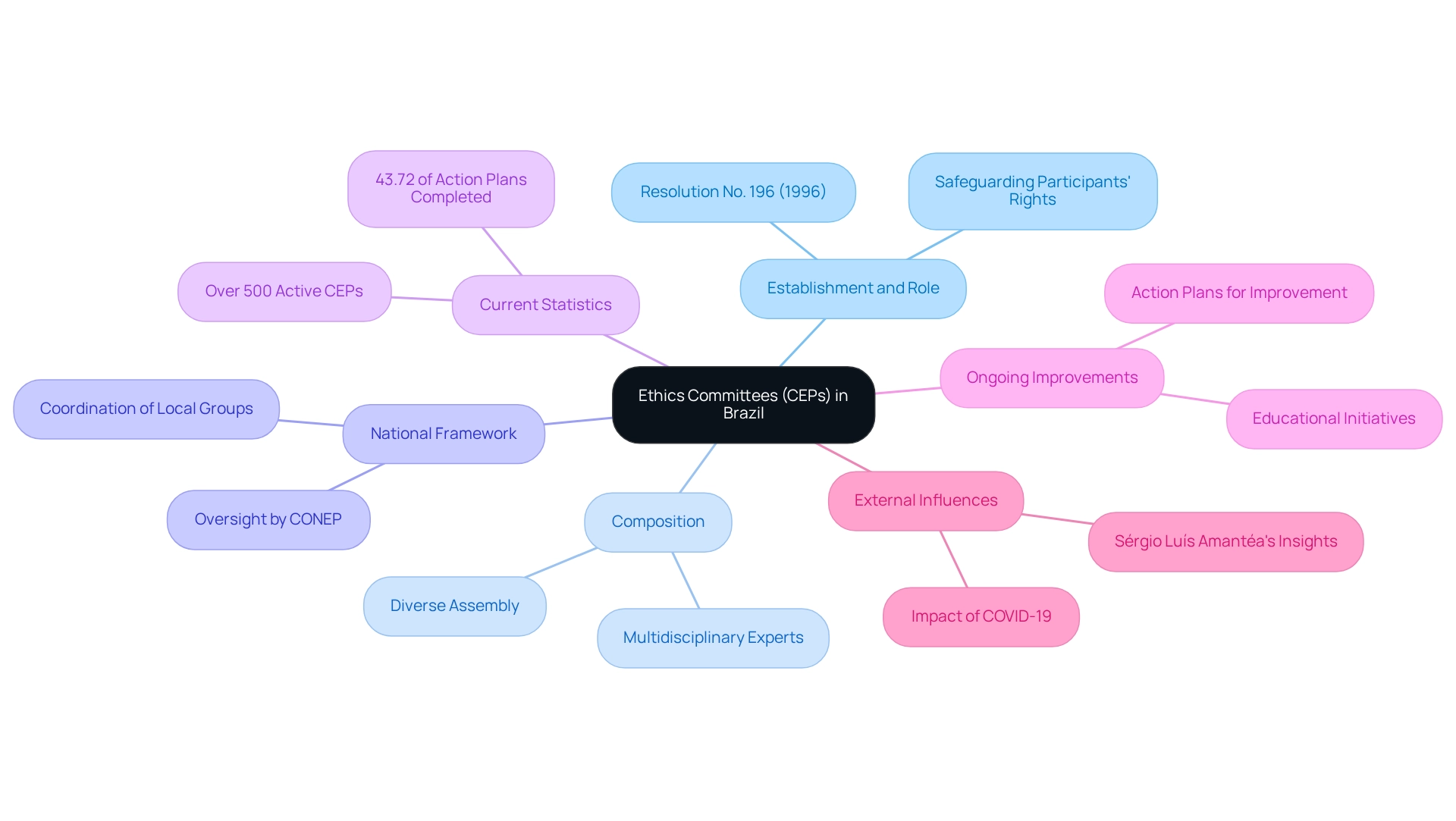
Ethics committee considerations in Brazil, known as Comitês de Ética em Pesquisa (CEPs), are pivotal in supervising studies involving human subjects. Established under the National Health Council's Resolution No. 196 in 1996, these committees are tasked with ensuring that study protocols comply with ethical standards and safeguard participants' rights.
Each CEP is composed of a diverse assembly of experts from various disciplines, fostering a multidisciplinary approach to review that enhances the quality and integrity of clinical research. Operating within a national framework overseen by the National Commission on Research Ethics (CONEP), the CEPs coordinate and supervise the actions of local groups across Brazil. This framework is essential for maintaining the integrity of clinical investigations, as it guarantees the consistent application of ethics committee considerations throughout studies.
As of 2025, Brazil boasts a robust network of review boards, with a significant increase in their numbers reflecting a growing emphasis on ethical oversight in clinical trials. Current statistics reveal that there are over 500 active CEPs nationwide, each contributing to the ethical framework of medical research. Recent evaluations indicate that these committees are committed to continuous improvement, with 43.72% of proposed action plans aimed at enhancing operational quality reported as completed or underway.
Following these assessments, each REC was encouraged to develop action plans addressing identified areas for improvement, demonstrating a proactive approach to refining their operations. The importance of CEPs, in the context of ethics committee considerations in Brazil, transcends mere compliance; they serve as a vital resource for researchers, providing guidance and support in navigating the ethical complexities of clinical trials. Ongoing educational initiatives, such as thematic discussions and participation in relevant events, are crucial for strengthening the role of CEPs as knowledge-producing entities.
As noted by Sérgio Luís Amantéa, the Q-CEP visits were temporarily suspended in March 2020 due to the COVID-19 pandemic, underscoring the influence of external factors on the functioning of ethics committees. These activities ensure that CEPs remain at the forefront of principled practices in Brazil.
Key Ethical Guidelines for Research in Brazil
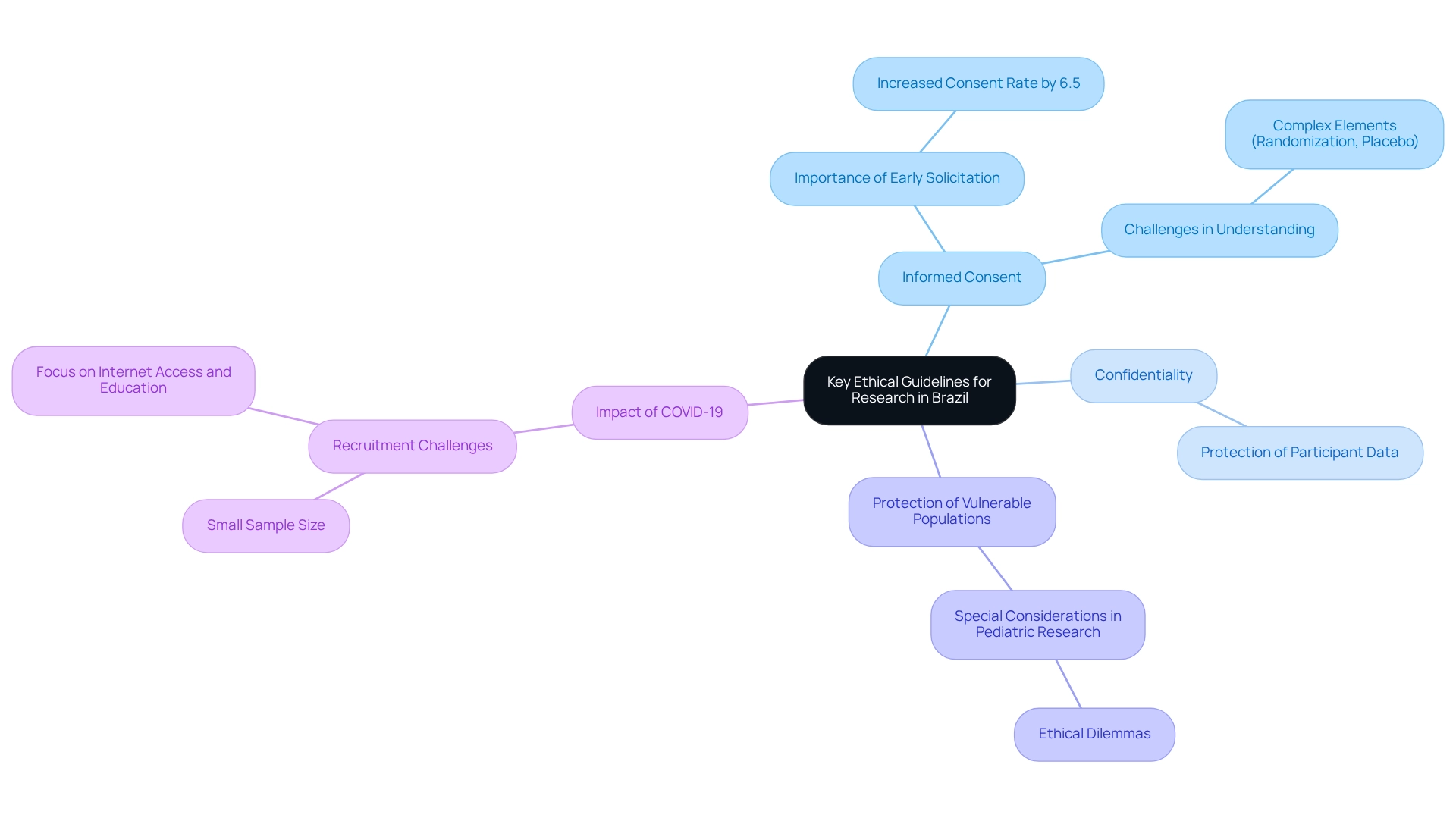
In Brazil, the moral guidelines governing studies are primarily outlined in Resolution No. 466/2012, which underscores the importance of respecting the dignity, rights, and welfare of study participants. This resolution delineates several key principles, including informed consent, confidentiality, and the requirement for review before initiating any study involving human subjects.
Researchers are mandated to ensure that participants are fully informed about the study's objectives, methodologies, potential risks, and benefits, thereby enabling them to make informed and voluntary decisions regarding their involvement. A significant aspect of these guidelines is the focus on informed consent practices. Recent studies reveal that soliciting consent early in the interview process can enhance consent rates by as much as 6.5 percentage points, thereby highlighting the critical role of clear communication. Despite these improvements, a meta-analysis indicates that approximately 75% of participants grasped the essential components of informed consent; however, challenges remain in understanding more complex elements such as randomization and placebo effects.
This situation emphasizes the necessity for continuous efforts to improve participants' comprehension of all facets of informed consent, safeguarding their rights and welfare. Investigators must adopt a proactive approach to ensure participants achieve complete understanding, making certain they are fully aware of their rights and the implications of their participation.
Furthermore, the guidelines dictate that special attention must be directed toward vulnerable populations to shield them from potential exploitation. As John Pearn notes, understanding the possible moral dilemmas in pediatric studies is crucial, as ethical challenges can arise in these contexts.
The landscape of clinical investigation has also encountered limitations due to the COVID-19 pandemic, which has impacted recruitment and the capacity to conduct studies effectively. As the field of clinical investigations continues to evolve, adherence to ethical standards and ethics committee considerations in Brazil remains essential for ensuring the integrity of studies and the protection of participants in this dynamic investigative environment.
The Review Process of Research Proposals by Ethics Committees
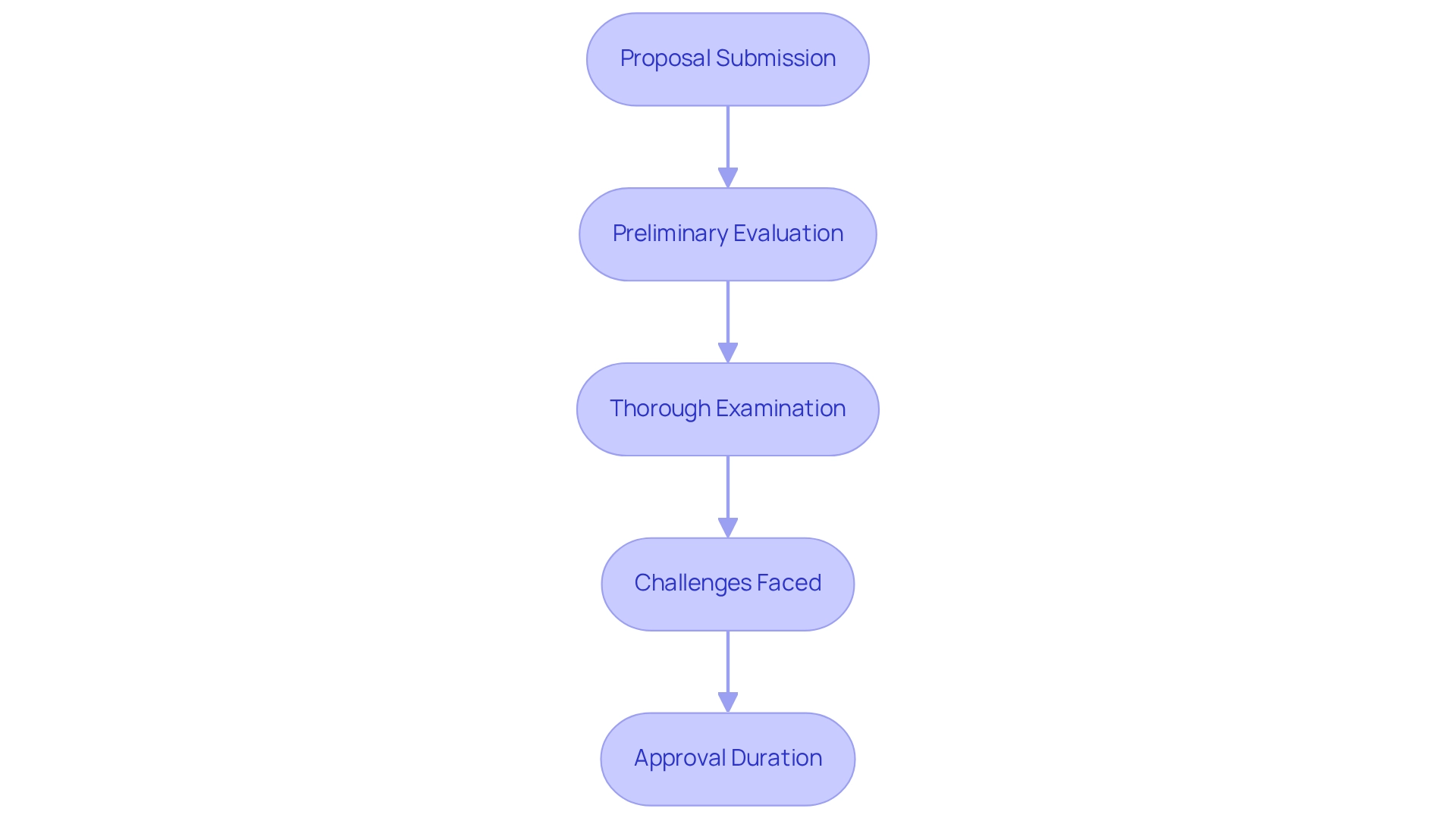
The evaluation procedure for research proposals by review boards in Brazil is organized and comprehensive, incorporating essential steps and ethics committee considerations. Initially, researchers must submit their proposals alongside extensive documentation, including informed consent forms and detailed study protocols. This submission is critical, as incomplete documentation is a primary reason for project returns.
Mariana Bueno emphasizes a frequent lack of information regarding financial assistance, complicating the submission process further.
Upon submission, the ethics group conducts a preliminary evaluation to ensure that all necessary information is included. Following this initial assessment, the proposal undergoes a thorough examination, wherein committee members analyze the moral implications, scientific validity, and potential risks to participants. This stage is vital, as it guarantees that the study adheres to ethical standards and protects participant rights.
The review process faces challenges. A study assessing projects submitted to the Research Ethics Committee at HCFMUSP found that 42.5% of the projects involved new drugs, vaccines, and diagnostic tests, while a significant 68.9% lacked sponsorship. Such findings highlight the necessity of understanding the reasons behind project resubmissions, which can streamline future submissions and expedite the review process.
Approval durations are influenced by the nature of the study and the complexity of the task, with primary studies requiring greater methodological attention from evaluators. Furthermore, the typical duration for review processes in Brazil has shown variability, with the pre-pandemic era averaging 121.41 days compared to 109.17 days during the pandemic. This fluctuation underscores the impact of external factors on the evaluation process's effectiveness.
In summary, the review procedure in Brazil is structured with ethics committee considerations to uphold strict moral standards while fostering research advancement. By ensuring meticulous evaluation of all proposals, ethics groups play a crucial role in safeguarding participants and promoting responsible research practices.
Challenges and Barriers for Ethics Committees in Brazil
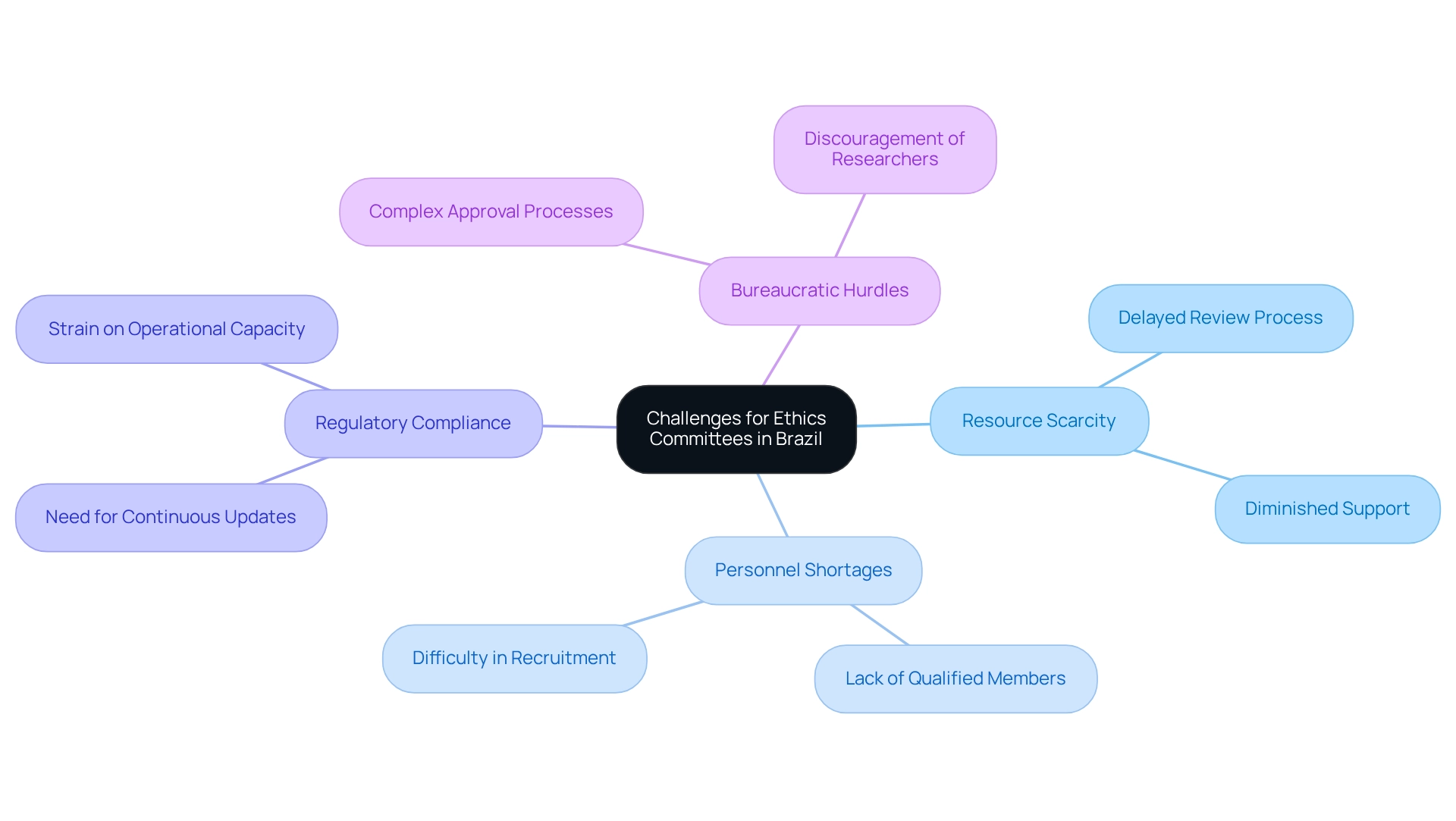
The effectiveness of ethics panels in Brazil is significantly compromised by various challenges related to ethics committee considerations. A primary obstacle is the persistent scarcity of resources, which not only delays the review process but also diminishes the support available for group members. This lack of resources is exacerbated by a notable shortage of qualified personnel, rendering it increasingly difficult to form diverse and knowledgeable committees capable of navigating the complexities of clinical research.
Furthermore, the need to comply with rapidly changing regulations presents an ongoing challenge. Committees must continuously update their knowledge of new laws and ethical standards, which can strain their operational capacity. The bureaucratic nature of the approval process complicates matters further, often discouraging researchers from submitting proposals. This reluctance can impede the advancement of clinical studies in Brazil, ultimately affecting the development and commercialization of innovative medical devices.
A recent study involving 84 Research Ethics Committee (REC) Chairs across Brazil underscored these issues, revealing that challenges in member recruitment and participation are prevalent nationwide. Although many chairs believe their groups are adequately structured for accreditation, operational difficulties and a lack of interest from potential members were identified as significant hurdles. This highlights the urgent need for increased awareness and support for RECs to ensure they can fulfill their crucial role in clinical investigation, particularly regarding ethics committee considerations in Brazil.
As Daniel Ribeiro Paes de Castro noted, 'it strengthens the communication network framework, which is essential for the qualification of evaluation related to ethical review.' This statement underscores the necessity for efficient communication among review groups to manage the intricacies of clinical research.
The Federal Constitution of Brazil recognizes health as a fundamental right, emphasizing the importance of effective oversight teams in safeguarding this right. However, the current landscape indicates that without addressing these challenges—such as resource allocation and personnel shortages—ethics committee considerations in Brazil may struggle to meet the demands of modern clinical studies effectively.
In this context, bioaccess® aims to expedite the advancement of medical devices through its comprehensive clinical trial management services, which include Early-Feasibility Studies, First-In-Human Studies, and Post-Market Clinical Follow-Up Studies.
With over 20 years of experience in Medtech, bioaccess® leverages its expertise and tailored strategies to enhance the operational capacity of review boards. By providing adaptable solutions designed for the specific challenges faced by these groups, bioaccess® supports the responsible oversight essential for successful clinical trials, ultimately contributing to the improvement of clinical studies in Brazil.
Protecting Human Subjects: The Role of Ethics Committees
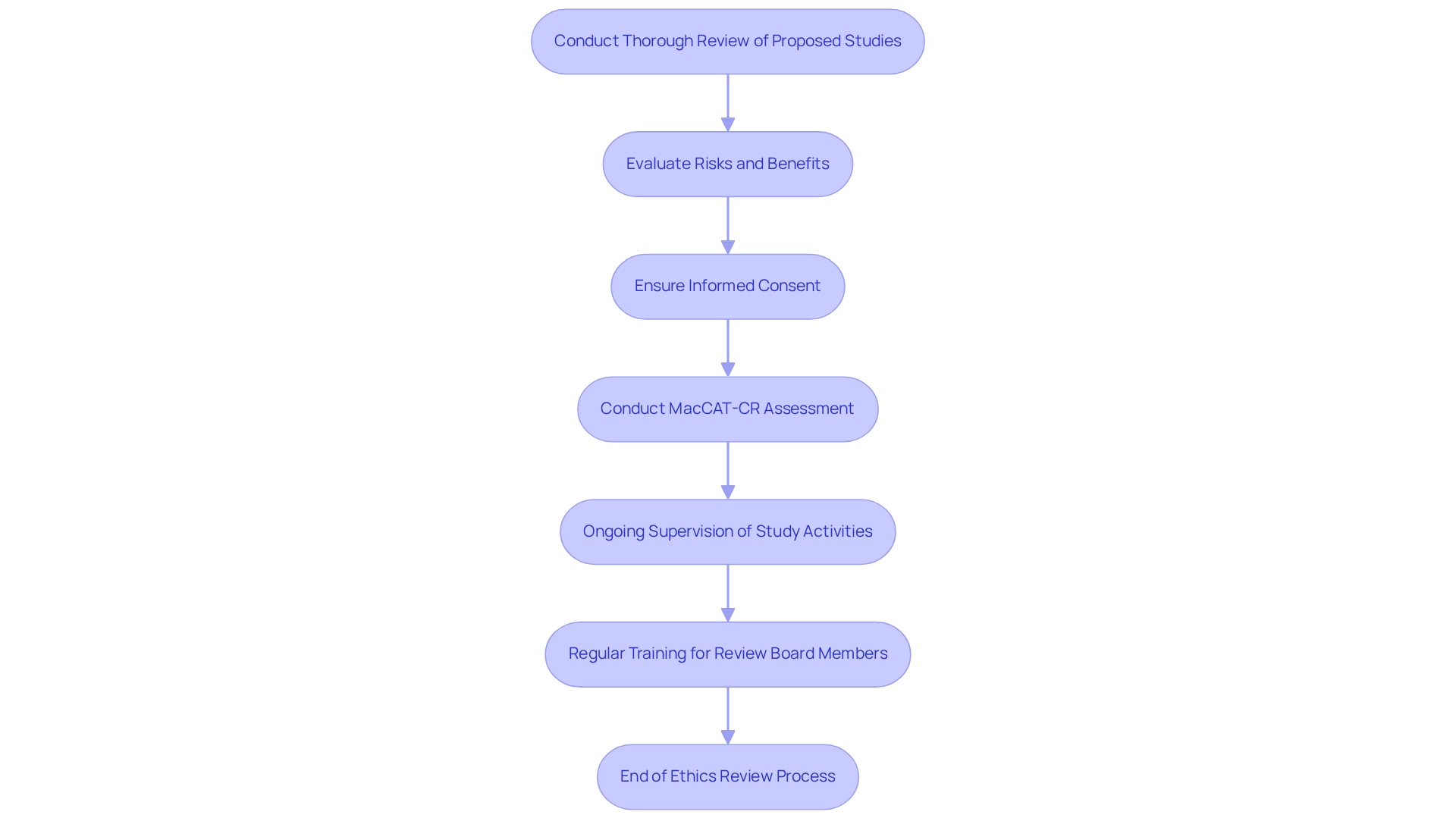
In Brazil, the considerations of ethics committees are paramount for safeguarding the rights and welfare of human participants involved in clinical studies. Their primary function is to conduct thorough reviews of proposed studies, meticulously evaluating potential risks and benefits while integrating the ethics committee considerations in Brazil. This process is crucial for ensuring that informed consent is not only acquired but that participants are fully educated about their rights, including the ability to withdraw from the study at any point without penalty.
Statistics reveal that adherence to informed consent protocols is a significant concern in Brazilian clinical trials, with studies indicating that a considerable percentage of participants may not fully grasp the consent process.
The MacCAT-CR assessment is particularly pertinent in this context, as it assesses whether participants comprehended the explanation and possessed the competence to decide on their participation. To address these issues, oversight groups are tasked with ensuring that the informed consent procedure is clear, comprehensive, and accessible, thereby enhancing participant understanding and involvement. Moreover, these groups are responsible for the ongoing supervision of study activities, ensuring that ethics committee considerations in Brazil are upheld throughout the duration of the investigation.
This continuous oversight is vital for maintaining public confidence in the investigative process and ensuring the safety and well-being of participants. The administration of these groups is supported by the CNESS, composed of 37 individuals, including representatives from the scientific community and scholars, who are charged with formulating standard operating procedures for its operations.
A significant case study underscores the importance of training and communication within review boards. Regular training sessions for group members on integrity in studies, coupled with efficient communication with the National Commission for Integrity in Studies (CONEP), have demonstrated improvements in the quality of moral supervision in clinical trials. This proactive strategy guarantees that group members remain well-informed about evolving moral standards and practices, ultimately contributing to the integrity of clinical studies in Brazil.
Additionally, the DPM's review of clinical trial applications hinges on obtaining written proof of review board approvals, highlighting the regulatory framework surrounding these groups and their significance in the clinical trial process.
In summary, the role of review boards in Brazil, including ethics committee considerations, extends beyond initial approvals; they are integral to cultivating a principled study environment that prioritizes participant rights and informed consent, thereby reinforcing the moral foundation of clinical trials. As Sidney Leal, a doctoral student in natural language processing, noted, effective communication and training within these groups are essential for upholding high standards of integrity in scholarly work.
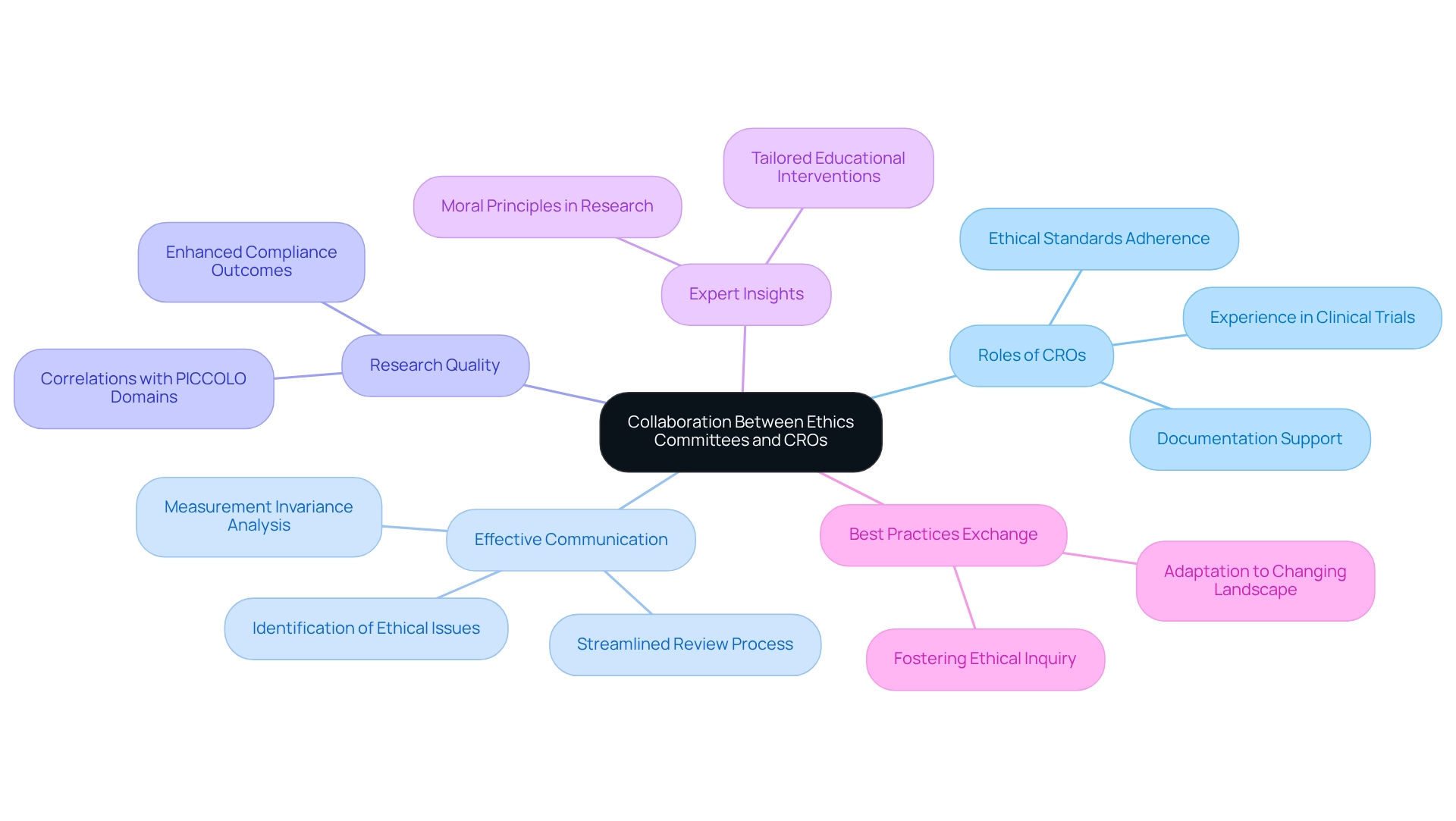
Collaboration Between Ethics Committees and Clinical Research Organizations
Cooperation between review boards and clinical study organizations (CROs) is essential for upholding ethical standards and facilitating the study process in Brazil. Effective communication among these entities streamlines the review process, allowing for the swift identification and resolution of ethical issues. CROs, such as bioaccess®, play a crucial role by providing necessary documentation and support to review boards, ensuring that project proposals adhere to established ethical standards while leveraging over 20 years of experience in managing clinical trials.
Recent statistics indicate a growing trend in the collaboration between review boards and CROs, with studies showing that such partnerships significantly enhance the quality of research conducted. For instance, a measurement invariance analysis highlighted the effectiveness of communication strategies employed during the review process, revealing that well-structured interactions lead to improved compliance outcomes. Notably, the adapted RIFLE-P demonstrated significant associations with all four Brazilian Portuguese PICCOLO domains, with correlations ranging from 0.32 to 0.47, all with p < 0.001, emphasizing the critical role of effective communication.
Expert insights underscore the significance of these collaborations, as they foster a culture of ethical inquiry and protect participant rights. As Luiz Paulo Carvalho astutely noted, "Officially, it should be acknowledged that the concept of 'human values' originates from moral principles, and not the other way around," which underscores the fundamental role of morality in academic practices. A case study examining gender differences in patient safety attitudes revealed that tailored educational interventions, informed by collaborative insights, can effectively address both professional and gender-based variations in attitudes towards patient safety.
This illustrates how the partnership between advisory groups and CROs, like bioaccess®, can yield more knowledgeable and efficient study practices.
Moreover, ongoing collaboration facilitates the exchange of best practices and insights, which is vital for adapting to the changing landscape of clinical research in Brazil. By working together, ethics committees and CROs can ensure that ethical considerations in Brazil remain paramount in clinical trials, ultimately advancing the field of medical technology and enhancing participant safety. Bioaccess® aims to expedite medical devices through their expertise and tailored strategies, highlighting the importance of these partnerships in relation to clinical trial considerations.
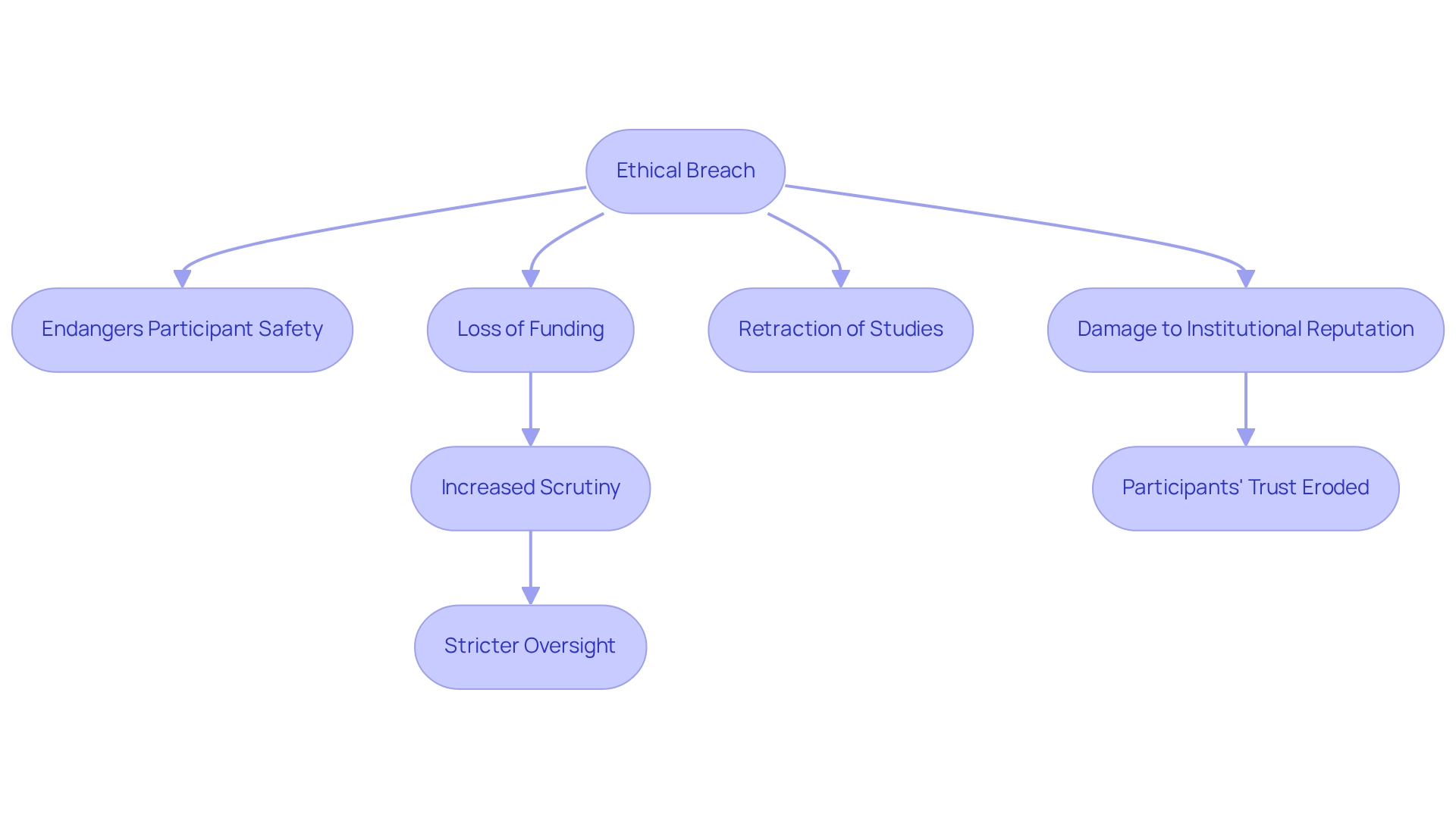
Consequences of Ethical Breaches in Research
Ethical violations in studies can have profound consequences for both investigators and participants. Breaches of moral standards not only endanger the safety and welfare of participants but also diminish public confidence and damage the standing of academic institutions. In Brazil, ethics committee considerations have led to a substantial number of disciplinary measures against researchers for misconduct, with many facing serious repercussions such as loss of funding, retraction of published studies, and potential legal consequences.
Significantly, the majority of article withdrawals in health and life sciences by Brazilian institutions were linked to misconduct, highlighting the pressing necessity for ethics committee considerations in Brazil.
For instance, a case study analyzing the Ethics Committee at the State University of Montes Claros revealed a high approval rate of 95.8% for projects submitted between 2000 and 2009. Nonetheless, informed consent concerns were the primary cause for disapproval, underscoring the crucial significance of adhering to moral standards in safeguarding study participants. As noted by Patricia Alves Paiva, "The article reports the experience of the Research Ethics Committee at the State University of Montes Claros, MG, Brazil, in the period from 2000 to 2009."
Recent data indicates that in 2025, the consequences of moral violations in Brazilian research remain significant, emphasizing the importance of ethics committee considerations in Brazil, with increased scrutiny from regulatory bodies leading to more stringent oversight of future studies. Furthermore, it is noteworthy that 85.4% of REC members were women, reflecting the ethics committee considerations in Brazil regarding diversity and representation.
Expert insights reveal that moral misconduct not only impacts the researchers involved but also has lasting consequences on participants, who may experience compromised safety and trust. The integrity of clinical studies relies on a commitment to moral principles, ensuring the safety of human participants and the trustworthiness of the scientific community. At bioaccess®, we strive to expedite the development of medical devices earlier through our expertise and tailored approach, emphasizing the significance of ethical standards in clinical studies.
We implement rigorous compliance methodologies and maintain transparency in our operations to uphold these standards. Should you have any queries or concerns about the processing of your information, please email our Grievance Officer at IMH ASSETS CORP (doing business as "bioaccess®"). We are dedicated to addressing your concerns in accordance with applicable law, ensuring transparency and trust in our operations.
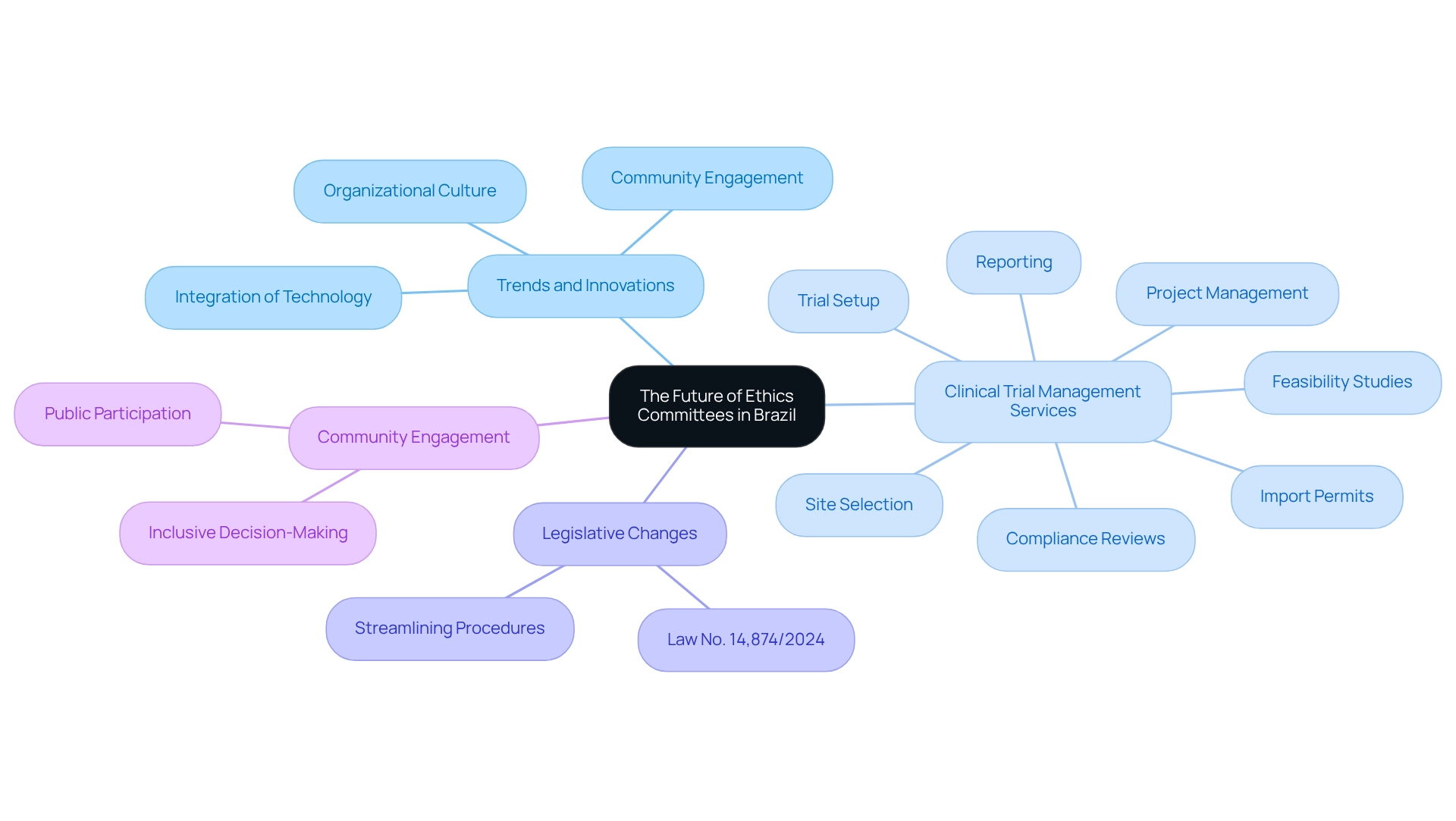
The Future of Ethics Committees in Brazil: Trends and Innovations
Substantial change is on the horizon for ethics committee considerations in Brazil, driven by new trends and technological advancements. As technology continues to reshape the landscape, ethics committee considerations in Brazil are increasingly likely to incorporate sophisticated tools for monitoring and reviewing research proposals. This shift not only enhances efficiency but also promotes greater transparency in the review process.
In this context, bioaccess offers comprehensive clinical trial management services that encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
These services are essential, especially with the recent enactment of Law No. 14,874/2024, which represents a pivotal moment in the evolution of ethical reviews, aiming to streamline procedures and reduce bureaucratic obstacles.
This legislative change is expected to expedite the approval process, factoring in ethics committee considerations in Brazil, thus enabling a quicker initiation of critical projects. Statistics suggest that the impact of this law could lead to a significant increase in the number of studies approved annually, thereby hastening innovation in the medical technology sector. For context, the average yearly attendances across the 21 hospitals is 384,479, underscoring the volume of studies that could benefit from these changes.
Moreover, there is a growing recognition of the importance of community engagement and public participation in the ethics committee considerations in Brazil throughout the review process. This trend is poised to foster more inclusive and representative decision-making, ensuring that the perspectives of diverse stakeholders are taken into account. As oversight groups adapt to these changes, they will play a vital role in upholding moral standards while aligning ethics committee considerations in Brazil with societal values.
Looking ahead to 2025, the integration of technology within advisory groups is anticipated to continue evolving. Innovations such as digital platforms for monitoring studies and data management will enhance the oversight capabilities of these groups, ensuring that ethics committee considerations in Brazil remain central to clinical investigations. As G6 noted, "The development and implementation time, that’s all I look at..." This statement underscores the challenges encountered in adopting new systems.
Additionally, the case study on the Internet of Things (IoT) in Brazil exemplifies the application of technology in healthcare, highlighting the country's potential for innovation despite previous missed opportunities. Furthermore, the significance of organizational culture and effective change management strategies will be crucial for oversight groups as they navigate the complexities of contemporary research principles. As these developments unfold, ethics committee considerations in Brazil will be essential in ensuring that ethical standards are upheld while embracing the advantages of technological advancements, ultimately contributing to job creation, economic growth, and healthcare improvement in the region.
Conclusion
The role of ethics committees in Brazil is fundamental to ensuring the integrity and ethical conduct of clinical research. These committees, known as Comitês de Ética em Pesquisa (CEPs), are tasked not only with the initial ethical review of research proposals but also with a continuous responsibility to safeguard participant rights and welfare. They adeptly navigate the complexities of informed consent, uphold confidentiality, and monitor ongoing research activities, thereby reinforcing the importance of ethical standards in clinical trials.
Despite their critical functions, ethics committees encounter numerous challenges, including resource limitations and the necessity to adapt to evolving regulations. Such barriers can impede their effectiveness and the overall advancement of clinical research in Brazil. However, a proactive approach that fosters collaboration between ethics committees and clinical research organizations holds promise for enhancing the ethical landscape of medical research. These partnerships facilitate improved communication, streamline the review process, and ultimately contribute to participant safety.
Looking ahead, the future of ethics committees in Brazil is poised for evolution through the integration of technology and legislative changes designed to enhance efficiency. As these committees embrace innovation and engage with the community, they will be better positioned to uphold ethical standards while adapting to the dynamic environment of clinical research. The commitment to ethical oversight remains crucial, not only for the protection of human subjects but also for maintaining public trust in the research process. As Brazil progresses in medical technology, the collaboration between ethics committees and research organizations will be vital in fostering an ethical research culture that prioritizes participant rights and well-being.
Frequently Asked Questions
What are Comitês de Ética em Pesquisa (CEPs) in Brazil?
CEPs are ethics committees in Brazil responsible for supervising studies involving human subjects. They ensure that study protocols comply with ethical standards and protect participants' rights.
When were CEPs established and under what regulation?
CEPs were established in 1996 under the National Health Council's Resolution No. 196.
What is the composition of each CEP?
Each CEP is composed of a diverse group of experts from various disciplines, promoting a multidisciplinary approach to enhance the quality and integrity of clinical research.
How do CEPs operate within Brazil?
CEPs operate within a national framework overseen by the National Commission on Research Ethics (CONEP), coordinating and supervising local groups across Brazil to maintain consistent ethical oversight in studies.
How many active CEPs are there in Brazil as of 2025?
As of 2025, there are over 500 active CEPs in Brazil.
What recent trends have been observed regarding CEPs?
There has been a significant increase in the number of CEPs, reflecting a growing emphasis on ethical oversight in clinical trials. Additionally, many CEPs are committed to continuous improvement.
What is the role of CEPs in supporting researchers?
CEPs serve as vital resources for researchers, providing guidance and support in navigating the ethical complexities of clinical trials.
What educational initiatives are important for CEPs?
Ongoing educational initiatives, such as thematic discussions and participation in relevant events, are crucial for strengthening the role of CEPs as knowledge-producing entities.
What impact did the COVID-19 pandemic have on CEP activities?
The COVID-19 pandemic temporarily suspended Q-CEP visits in March 2020, highlighting the influence of external factors on the functioning of ethics committees.
What are the key principles outlined in Resolution No. 466/2012?
The key principles include informed consent, confidentiality, and the necessity for review before initiating any study involving human subjects, emphasizing the dignity, rights, and welfare of participants.
Why is informed consent important in clinical studies?
Informed consent is crucial as it ensures participants are fully aware of the study's objectives, methodologies, potential risks, and benefits, allowing them to make informed and voluntary decisions about their involvement.
What challenges exist regarding participants' understanding of informed consent?
Despite improvements, approximately 75% of participants understand the essential components of informed consent, but challenges remain in comprehending more complex elements like randomization and placebo effects.
How should researchers address the needs of vulnerable populations?
Researchers must direct special attention to vulnerable populations to protect them from potential exploitation and ensure they fully understand their rights and the implications of their participation.
How has the COVID-19 pandemic affected clinical investigations?
The COVID-19 pandemic has posed limitations on recruitment and the capacity to conduct studies effectively, highlighting the need for adherence to ethical standards in this evolving landscape.

