Overview
The key factors in device trial success encompass:
- Effective study design
- Regulatory compliance
- Participant recruitment and retention
- Data management practices
- Stakeholder collaboration
These elements are not merely components of the process; they are critical for ensuring the safety and efficacy of medical technologies. By enhancing patient outcomes and streamlining the approval process, they leverage innovative strategies and thorough evaluations. Understanding and implementing these factors is essential for advancing clinical research and improving healthcare delivery.
Introduction
In the realm of medical technology, the significance of device trials is paramount. These trials form the backbone for validating the safety and efficacy of innovative medical devices prior to their market introduction, ultimately shaping patient care and outcomes. As the healthcare landscape evolves—driven by rapid advancements and shifting regulatory environments—the methodologies and strategies surrounding these trials are undergoing a transformation.
From adaptive trial designs that enhance flexibility to the integration of cutting-edge technologies like artificial intelligence, the future of device trials promises to be both dynamic and impactful. This article explores the critical elements of effective clinical trial design, navigates regulatory requirements, and examines the emerging trends redefining the landscape of medical device research, all aimed at ensuring that only the safest and most effective technologies are introduced into healthcare systems.
Understanding the Importance of Device Trials in Medical Technology
Device evaluations are pivotal in validating the safety and efficacy of medical technologies prior to market introduction. These experiments provide vital information that not only guides regulatory approvals but also supplies producers with an understanding of performance in real-world situations. The success of these experiments is closely linked to enhanced patient results, which are key factors in device trial success and highlight their essential role in medical equipment development.
Moreover, they play a crucial role in identifying potential risks and benefits associated with new technologies, ensuring that only safe and effective tools are integrated into healthcare systems. Recent progress in medical equipment tests has further highlighted their importance. The healthcare technology landscape is evolving rapidly, necessitating innovative approaches to study design that can adapt to changing regulatory environments and patient needs. As noted by Samruddhi Yardi, "the healthcare technology landscape is dynamic, requiring expertise and innovation to deliver impactful results."
In 2025, the significance of equipment evaluations remains crucial, as they not only confirm new technologies but also improve the overall quality of care provided to patients. Statistics show that key factors in device trial success can result in substantial enhancements in patient outcomes. For instance, a study suggested that instruments undergoing stringent clinical evaluations showed a 30% greater success rate in attaining desired health results compared to those that did not. This highlights the critical nature of thorough testing in the development process.
Furthermore, telemedicine consultations in the U.S. reached over 1.7 billion in 2020, demonstrating the increasing dependence on technology in healthcare, which further underscores the significance of equipment assessments. Real-world instances of successful medical equipment studies further demonstrate this point. A significant case involved a new cardiac instrument that underwent thorough research studies, resulting in a 40% decrease in adverse events among patients. Such outcomes not only confirm the effectiveness of the apparatus but also foster trust among healthcare providers and patients alike.
Moreover, bioaccess® has shown its ability in overseeing various studies, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF), ensuring thorough management services customized to fulfill regulatory requirements. Specialist views underscore the importance of clinical studies in the medical equipment field. Industry leaders emphasize that these assessments are key factors in device trial success, not merely regulatory hurdles but essential processes that ensure the delivery of safe and effective medical technologies. As the worldwide market for artificial intelligence in pharmaceutical discovery is anticipated to expand considerably, with a valuation expected to hit USD 12.8 billion by 2032, the incorporation of advanced methods in assessments will likely improve their efficiency and significance in the upcoming years.
In summary, the significance of equipment evaluations in medical technology cannot be overstated. They are essential for ensuring that innovative solutions are both safe and effective, ultimately leading to better patient outcomes and advancing the field of healthcare. Furthermore, comprehending the function of INVIMA as a Level 4 health authority in Colombia offers essential context for navigating the regulatory environment in which these studies operate.
Key Elements of Effective Clinical Trial Design
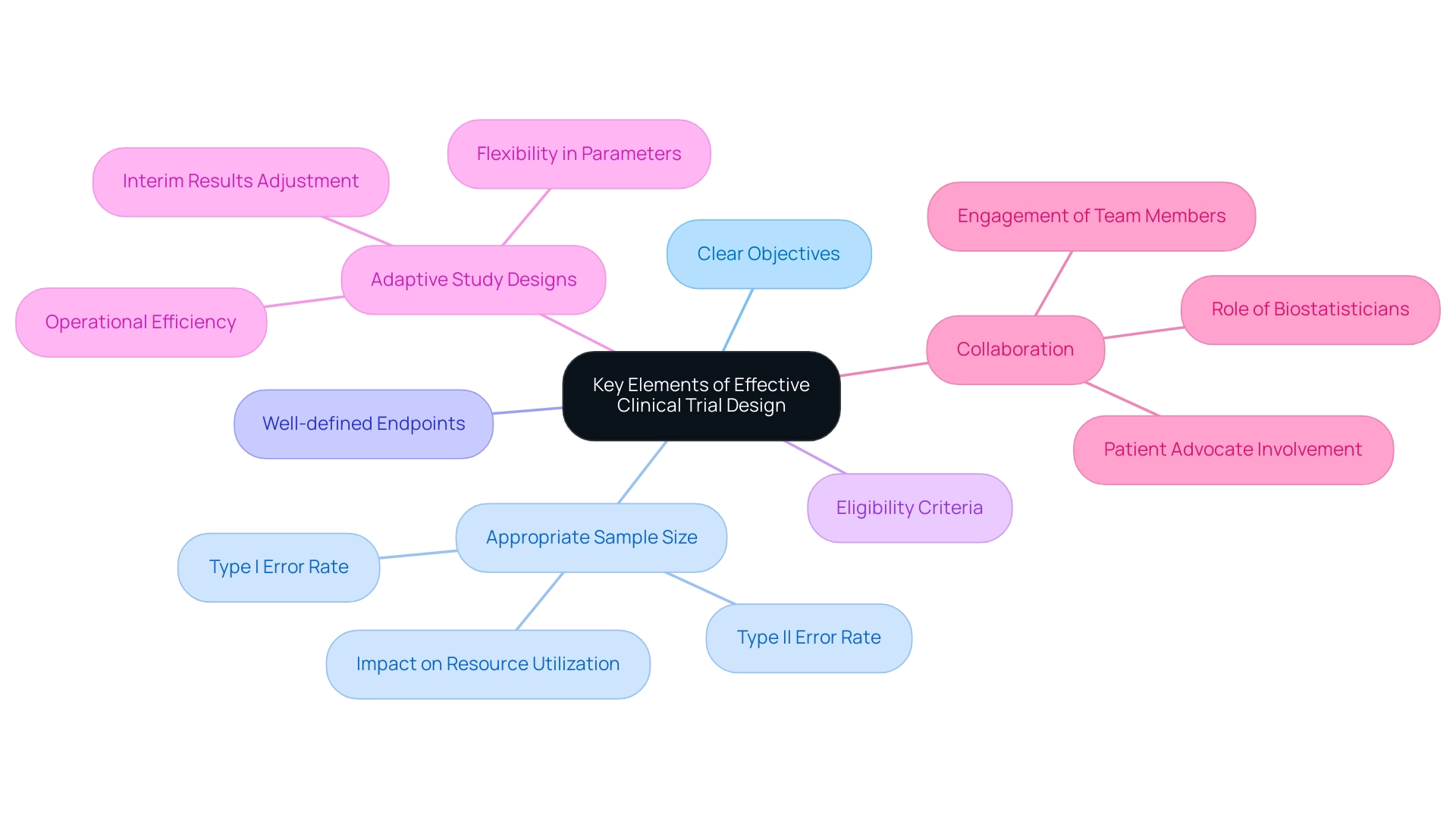
Effective research design is a pivotal factor in the success of device trials, encompassing essential elements such as clear objectives, appropriate sample size, and well-defined endpoints. Establishing eligibility criteria that accurately reflect the target population is vital, ensuring that the study can be completed within the allocated timeframe and budget. In 2025, adaptive study designs have gained significant traction in clinical research, highlighting their importance.
These designs provide enhanced flexibility, enabling researchers to adjust study parameters based on interim results. This adaptability not only improves operational efficiency but also increases the likelihood of achieving meaningful outcomes. A recent analysis underscores that studies employing adaptive designs can lead to more robust conclusions while optimizing resource utilization.
Sample size considerations are particularly critical, as they directly impact the validity of study results. A well-calibrated sample size strikes a balance between the need for statistical power and resource constraints. Research indicates that careful consideration of Type I and Type II error rates during sample size calculations can significantly influence the overall success of medical studies.
The probability of failing to reject the null hypothesis when it should be rejected, known as Type II error, underscores the necessity of meticulous planning in this area. Studies have shown that inadequate sample sizes often result in inconclusive results, as outlined in the case study titled 'Sample Size Considerations in Clinical Trials,' which discusses how sample size impacts resource utilization and the ability to draw conclusive results.
Moreover, the collaborative nature of the design process is paramount. Engaging all team members—including statisticians, clinicians, and patient advocates—ensures a comprehensive approach that addresses various perspectives and areas of expertise. As noted by PharPoint Research, 'Your study’s biostatistician must be an active listener, working to understand pressure points adequately.' This joint endeavor is crucial for developing evaluations that not only comply with regulatory standards but also align with the needs of the patient population.
At bioaccess®, we leverage over 20 years of experience overseeing research across Latin America, focusing on Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies. Our extensive research management services encompass feasibility studies, site selection, compliance evaluations, setup, import permits, project management, and reporting. By prioritizing the key factors in device trial success, researchers can enhance the likelihood of successful outcomes and contribute to the advancement of medical technology.
Furthermore, the significance of research statistics in shaping treatment comprehension and medical choices emphasizes the broader effects of efficient study design.
Navigating Regulatory Requirements for Device Trials
Navigating regulatory requirements is a critical factor in the success of device trials, as each country enforces specific regulations that must be meticulously adhered to. In the United States, the FDA's Investigational Device Exemption (IDE) plays a pivotal role, while Europe follows the Medical Device Regulation (MDR). These regulations govern the design, execution, and reporting of research studies, making it essential for study sponsors to engage with regulatory authorities early in the process.
This proactive engagement clarifies expectations and ensures that all necessary documentation is prepared and submitted promptly, significantly streamlining the approval process and reducing potential delays. As we approach 2025, understanding the nuances of the FDA IDE process is more critical than ever. Recent updates underscore the importance of comprehensive submissions that demonstrate compliance with both safety and efficacy standards. Successful IDE submissions typically include robust medical information and a clear justification for the proposed study design, as these elements are vital for device trial success and can enhance the likelihood of approval.
As bioaccess® aims to expedite the enhancement of medical equipment through their expertise and tailored strategies, adherence to these regulatory standards is crucial for study sponsors. Furthermore, insights from regulatory specialists, such as Katherine Ruiz, an authority in regulatory affairs for medical instruments and in vitro diagnostics in Colombia, suggest that maintaining open channels of communication with the FDA can facilitate the review process. Case studies reveal that manufacturers who proactively address potential concerns during the IDE submission phase often encounter fewer obstacles during the review.
The DHHS conclusions and recommendations on device experiments registry highlight the complexities of the regulatory environment, indicating that while a data bank could improve access to research information, it should be limited to serious or life-threatening conditions without alternative therapies. As the landscape of medical device experiments continues to evolve, staying informed about the key factors in device trial success—including regulatory updates and best practices—is essential for experiment sponsors seeking to advance their devices efficiently and effectively. The urgency of this endeavor is underscored by the statistic that U.S. life expectancy fell by 0.2 years from 2019 to 2021, emphasizing the critical need for timely advancements in medical technology. Additionally, the U.S. Food and Drug Administration has stated that no human studies were conducted by the authors for this article, reinforcing the significance of regulatory compliance and the integrity of research.
With bioaccess® leading the charge in Medtech research across Latin America, their focus on innovation and regulatory excellence, coupled with extensive services such as feasibility studies, site selection, compliance reviews, study setup, project management, and reporting, positions them as an indispensable ally in navigating these challenges.
Strategies for Successful Participant Recruitment and Retention
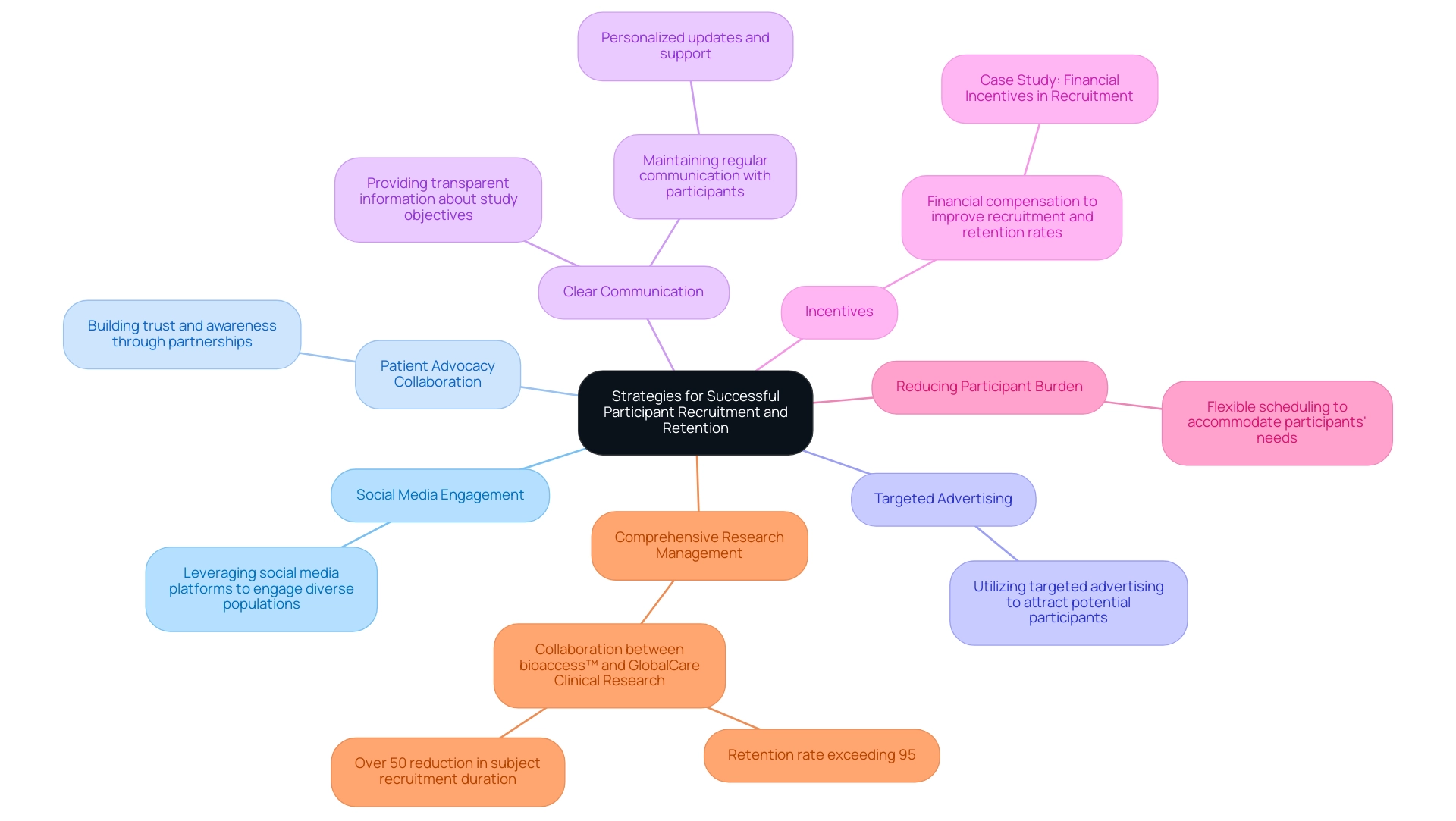
Key factors in device trial success encompass effective participant recruitment and retention, which are critical for preserving the integrity and validity of clinical studies. Recent statistics reveal that successful device trials often feature robust recruitment strategies capable of achieving enrollment targets within projected timelines. For instance, one study remarkably met its enrollment target within the initially estimated timeframe of 12 months, underscoring the effectiveness of well-planned approaches. Strategies that drive device trial success include:
- Leveraging social media platforms to engage diverse populations
- Collaborating with patient advocacy groups to build trust and awareness
- Utilizing targeted advertising to attract potential participants
Providing clear and transparent information about the study's objectives, procedures, and potential benefits is essential, as it alleviates participant concerns and fosters involvement. Once participants are enrolled, maintaining regular communication becomes vital. This can be enhanced through personalized updates and support, which not only keeps participants informed but also cultivates a sense of community.
Incentives, such as financial compensation, play a significant role in device trial success, having been shown to substantially improve recruitment and retention rates. A case study titled "Financial Incentives in Recruitment" highlighted that offering considerable financial rewards upon completion of the study was a noteworthy factor in attracting and retaining participants, aligning with findings from prior research. Furthermore, key factors in device trial success include reducing participant burden through flexible scheduling and addressing their needs, which can further enhance retention rates and ensure ongoing engagement throughout the study.
The collaboration between bioaccess™ and GlobalCare Clinical Research exemplifies the impact of comprehensive research management services. By leveraging bioaccess's extensive presence in Colombia, GCCT achieved over a 50% reduction in subject recruitment duration and an impressive retention rate exceeding 95%. Bioaccess's services encompass feasibility studies, site selection, compliance reviews, experiment setup, and project management, all of which are crucial for successful execution.
As we look toward 2025, understanding the key factors in device trial success, particularly the implementation of innovative recruitment strategies, cannot be overstated. Future studies should prioritize:
- Investing in stakeholder motivation
- Reaching consensus on population characteristics for representativeness
- Including random samples to assess selection bias
As James R Hebert observed, "The instances of CBPR-focused initiatives outlined here demonstrate that effective recruitment of racial/ethnic minorities into cancer prevention research is achievable."
By applying these strategies, research studies can achieve higher retention rates and more reliable results, emphasizing the key factors in device trial success and ultimately promoting the advancement of medical devices that enhance lives.
Implementing Strong Data Management Practices
Establishing robust information management practices stands as a pivotal factor in the success of device trials. With over 20 years of experience in the Medtech sector, bioaccess® understands that implementing clear protocols for information collection, storage, and analysis is vital. The introduction of electronic data capture (EDC) systems not only streamlines information entry but significantly reduces the likelihood of errors, thereby enhancing overall study precision.
Statistics reveal that studies utilizing EDC systems experience a remarkable improvement in information integrity, with some research indicating a reduction in discrepancies by up to 30% when compared to traditional methods.
Regular evaluations and oversight are crucial for the swift identification of inconsistencies, ensuring that information remains reliable throughout the testing process. Adherence to Good Clinical Practice (GCP) guidelines in all data gathering is essential, as these standards safeguard the integrity of the study and affirm that results are both credible and reproducible.
bioaccess® excels in comprehensive research study management services, which encompass:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Study setup
- Import permits
- Project management
- Reporting
A recent analysis contrasting conventional monitoring techniques with modern approaches—such as risk-based and centralized monitoring—showed that while traditional methods like 100% source verification remain common, newer methodologies can achieve comparable compliance levels with reduced resource allocation. This evolution in oversight practices reflects the changing landscape of medical studies, where essential elements of device trial success, particularly effective information management techniques, are increasingly acknowledged as foundational for attaining successful outcomes.
As Lauren Houston noted, "This survey emphasizes the diversity of research quality management practices in Australia," underscoring the imperative for strong information management.
Furthermore, expert opinions highlight the importance of transparent quality monitoring processes to enhance information integrity in research studies. As we approach 2025, the emphasis on effective data management practices will intensify, necessitating that research teams adapt and innovate in their methodologies. With bioaccess® as a trusted CRO and consulting partner, U.S. medical device companies can confidently navigate the complexities of research studies in Latin America.
Fostering Collaboration and Communication Among Stakeholders
Encouraging collaboration and communication among stakeholders stands as a pivotal factor in the success of device trials. This includes not only the research team but also sponsors, regulatory bodies, and patient representatives. Regular meetings and updates, alongside clear communication channels, ensure that all parties remain aligned on objectives and progress.
Effective communication strategies, including the use of collaborative tools and platforms, are essential as they significantly enhance information sharing and teamwork. Statistics reveal that streamlined communication can lead to a 30% increase in efficiency, underscoring the critical nature of these practices. Additionally, understanding patient involvement is vital; for instance, adults typically prefer to travel no more than 30 minutes and under 22 miles for urgent care, which can inform recruitment strategies in clinical studies. A case study investigating the influence of financial incentives on participant recruitment illustrates that while the effectiveness of remuneration is complex, fostering open dialogue about participant expectations and experiences can enhance recruitment and retention rates.
The mixed results surrounding financial incentives emphasize that key factors in device trial success involve engaging stakeholders in meaningful conversations to address their concerns and motivations. Furthermore, successful Medtech studies have demonstrated that essential elements in device trial success encompass implementing structured communication strategies—such as regular stakeholder feedback sessions and collaborative decision-making processes—which can lead to improved study outcomes. As bioaccess® aims to connect innovative Medtech firms with opportunities for conducting research studies in Latin America, prioritizing cooperation among research participants is crucial. The partnership between bioaccess™ and Caribbean Health Group to position Barranquilla as a leading destination for research trials in Latin America, supported by Colombia's Minister of Health, exemplifies this commitment.
By navigating challenges more effectively and driving the advancement of innovative medical devices, organizations can ensure that all voices are heard. As E. Daly-DeJoy aptly noted, "collaboration is essential to unlocking the full potential of medical research." Moreover, collaborations with entities such as IDx Technologies and GlobalCare Clinical Trials enhance the capabilities of bioaccess® in identifying opportunities and improving efficiency, achieving over a 50% reduction in recruitment time and 95% retention rates.
Acknowledging the role of service partnerships, such as those offered by Thermo Fisher Scientific in biostatistics, provides additional insights into effective collaboration strategies. By prioritizing cooperation among research participants, organizations can navigate challenges more effectively and promote the advancement of innovative medical tools.
Emerging Trends and Innovations in Device Trials
Emerging trends and innovations in device testing are pivotal to the success of device trials, fundamentally transforming the clinical research landscape. The integration of technologies such as artificial intelligence (AI) and machine learning is increasingly prevalent, optimizing study design and enhancing participant recruitment strategies. For instance, AI-driven analytics streamline information collection processes, resulting in improved quality and expedited decision-making.
Importantly, a total of 921 device studies were submitted as 'delayed posting' under the FDAAA, underscoring the urgent need for more efficient study management solutions. Furthermore, the resurgence of Medical Device Regulation (MDR) solutions aims to seamlessly integrate study design, data collection, analysis, and submission, thereby enhancing research efficiency. The emergence of decentralized research studies (DCTs) is revolutionizing participant involvement, allowing individuals to engage from the comfort of their homes.
This flexibility not only broadens the participant pool but also enhances the overall experience, making studies more accessible and less burdensome. Innovations in clinical trial methodologies focus on simplifying processes for sites and alleviating the burden on patients, which is essential in today's fast-paced healthcare environment.
Additionally, proactive issue management is gaining traction as a key strategy among sponsors. By leveraging historical trend information, sponsors can define thresholds and monitor evolving trends, thereby enhancing risk management. For instance, some sponsors utilize historical trend information to manage issues proactively, enabling teams to identify tolerable risks and implement effective mitigation plans. Centralized data reviews contribute to higher data quality, greater resource efficiency, and shorter study timelines, ultimately expediting the approval process.
As the FDA has indicated, stakeholder feedback is crucial to inform future regulatory activities, emphasizing the importance of staying informed about these trends for stakeholders seeking to remain competitive and responsive to the evolving healthcare landscape. For Directors of Clinical Research, comprehending the key factors in device trial success is essential for the success of device studies, ensuring they can navigate the intricacies of contemporary medical research effectively.
Notably, partnerships like that of bioaccess® with GlobalCare Clinical Trials exemplify the potential for significant improvements in recruitment times and retention rates, highlighting the value of collaboration in enhancing clinical trial outcomes.
Conclusion
Device trials are indispensable for validating the safety and efficacy of medical technologies prior to their market introduction. They provide essential data for regulatory approval and significantly enhance patient outcomes by ensuring that only the safest and most effective devices are integrated into healthcare systems. As the medical landscape evolves, innovative methodologies—such as adaptive trial designs and the incorporation of artificial intelligence—are elevating trial effectiveness.
Successful clinical trial design hinges on clear objectives, appropriate sample sizes, and robust collaboration among stakeholders. Navigating the regulatory landscape, including compliance with the FDA's Investigational Device Exemption, is crucial for conducting ethical and efficient trials. Moreover, effective participant recruitment and retention strategies are vital for upholding trial integrity and fostering trust.
Emerging trends, including decentralized clinical trials and proactive issue management, are reshaping the clinical research landscape. Partnerships, such as those between bioaccess® and GlobalCare Clinical Trials, illustrate the potential for enhancing trial efficiency and participant engagement.
In conclusion, the future of device trials is characterized by a commitment to innovation, collaboration, and regulatory adherence. This evolution not only streamlines the development process but also enhances patient care, paving the way for safer and more effective medical technologies that ultimately benefit healthcare systems and patients alike.
Frequently Asked Questions
Why are device evaluations important in medical technology?
Device evaluations are crucial for validating the safety and efficacy of medical technologies before they enter the market. They provide essential information for regulatory approvals and help manufacturers understand performance in real-world scenarios, ultimately leading to improved patient outcomes.
How do device evaluations impact patient outcomes?
The success of device evaluations is linked to enhanced patient results, with studies suggesting that devices undergoing rigorous clinical evaluations have a 30% higher success rate in achieving desired health outcomes compared to those that do not.
What role do device evaluations play in identifying risks and benefits?
Device evaluations are instrumental in identifying potential risks and benefits associated with new medical technologies, ensuring that only safe and effective tools are incorporated into healthcare systems.
What recent trends highlight the importance of equipment evaluations?
The evolving healthcare technology landscape necessitates innovative study designs that can adapt to changing regulatory environments and patient needs, emphasizing the ongoing relevance of equipment evaluations.
Can you provide an example of successful medical equipment studies?
A notable case involved a new cardiac instrument that underwent extensive research, resulting in a 40% reduction in adverse events among patients, confirming the device's effectiveness and building trust among healthcare providers and patients.
What types of studies does bioaccess® manage?
Bioaccess® manages various studies, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF), ensuring compliance with regulatory requirements.
How does effective research design contribute to device trial success?
Effective research design includes clear objectives, appropriate sample sizes, and well-defined endpoints, which are essential for completing studies within budget and timeframe, thereby enhancing the likelihood of meaningful outcomes.
What is the significance of adaptive study designs in clinical research?
Adaptive study designs allow researchers to modify study parameters based on interim results, improving operational efficiency and increasing the chances of achieving significant outcomes.
Why is sample size important in clinical trials?
Sample size directly impacts the validity of study results. A well-calibrated sample size balances the need for statistical power with resource constraints, as inadequate sizes can lead to inconclusive results.
What collaborative approaches are essential in the design process of clinical trials?
Engaging all team members, including statisticians, clinicians, and patient advocates, is vital for a comprehensive approach that addresses various perspectives and ensures that evaluations meet regulatory standards while aligning with patient needs.




