Overview
Best practices for data collection in medtech trials are essential for establishing clear protocols, leveraging technology, ensuring regulatory compliance, incorporating patient perspectives, and facilitating continuous monitoring. These strategies not only enhance data quality and integrity but also significantly improve the reliability of clinical trials. Ultimately, they play a crucial role in the successful advancement of medical devices, underscoring the importance of meticulous planning and execution in clinical research.
Introduction
In the rapidly evolving landscape of medical technology, the significance of effective data collection in clinical trials is paramount. As Medtech companies strive to innovate and introduce new devices to the market, the integrity and accuracy of data gathered during trials form the bedrock for regulatory approvals and successful product development. Given the challenges posed by diverse patient populations, stringent regulatory requirements, and the necessity for real-time data capture, organizations must implement robust data management strategies to navigate this intricate environment.
Advancements in technology, such as electronic data capture systems and mobile applications, present promising solutions to enhance data quality and streamline processes. As the industry continues to evolve, understanding and adopting best practices in data collection will be essential for Medtech companies seeking to excel in this competitive arena.
The Critical Role of Data Collection in Medtech Trials
Data collection in medtech trials serves as the cornerstone of Medtech studies, providing essential evidence that underpins regulatory submissions and product development. The precision and thoroughness of data collection in these trials are vital, ensuring that the results of research studies are both valid and reliable. This reliability is crucial in assessing the safety and efficacy of medical devices.
In an industry characterized by rapid innovation, effective data collection and analysis in medtech trials can significantly expedite the journey from concept to commercialization.
Since the implementation of GDPR in 2018, MedTech firms conducting clinical studies in the EU have faced new responsibilities that complicate data collection processes. Common pitfalls, such as reliance on paper-based systems or Excel spreadsheets, can lead to integrity issues and challenges in compliance with these regulations. However, recent advancements in data collection, particularly the transition to digital platforms, have mitigated these challenges, enhancing management processes and bolstering the overall integrity of experiments.
Effective information gathering techniques are crucial for data collection in medtech trials, enabling the early identification of potential risks in the testing process. This proactive approach not only boosts patient safety but also enhances the integrity of the study. Collaboration between medical researchers and statisticians is essential in this context, fostering a comprehensive understanding of information needs and improving the analysis process. As Chris Rush, a biomedical engineer with 13 years of experience in the medical device field, notes, "Data collection in medtech trials is essential for guaranteeing the success and regulatory compliance of research studies."
Moreover, the impact of robust data collection extends beyond execution; it significantly influences regulatory submissions. A comprehensive research management service, such as that offered by bioaccess, includes:
- feasibility studies
- site selection
- compliance evaluations
- setup
- import permits
- project oversight
- reporting
These components not only support compliance but also enhance the overall effectiveness of trials, facilitating the advancement of medical devices in the marketplace.
Additionally, the economic implications of Medtech clinical studies—ranging from job creation to healthcare improvement—underscore the necessity for international collaboration in this field. As the Medtech landscape continues to evolve, the critical role of data collection as an effective information gathering strategy cannot be overstated. It remains a focal point for organizations striving for success in this competitive arena.
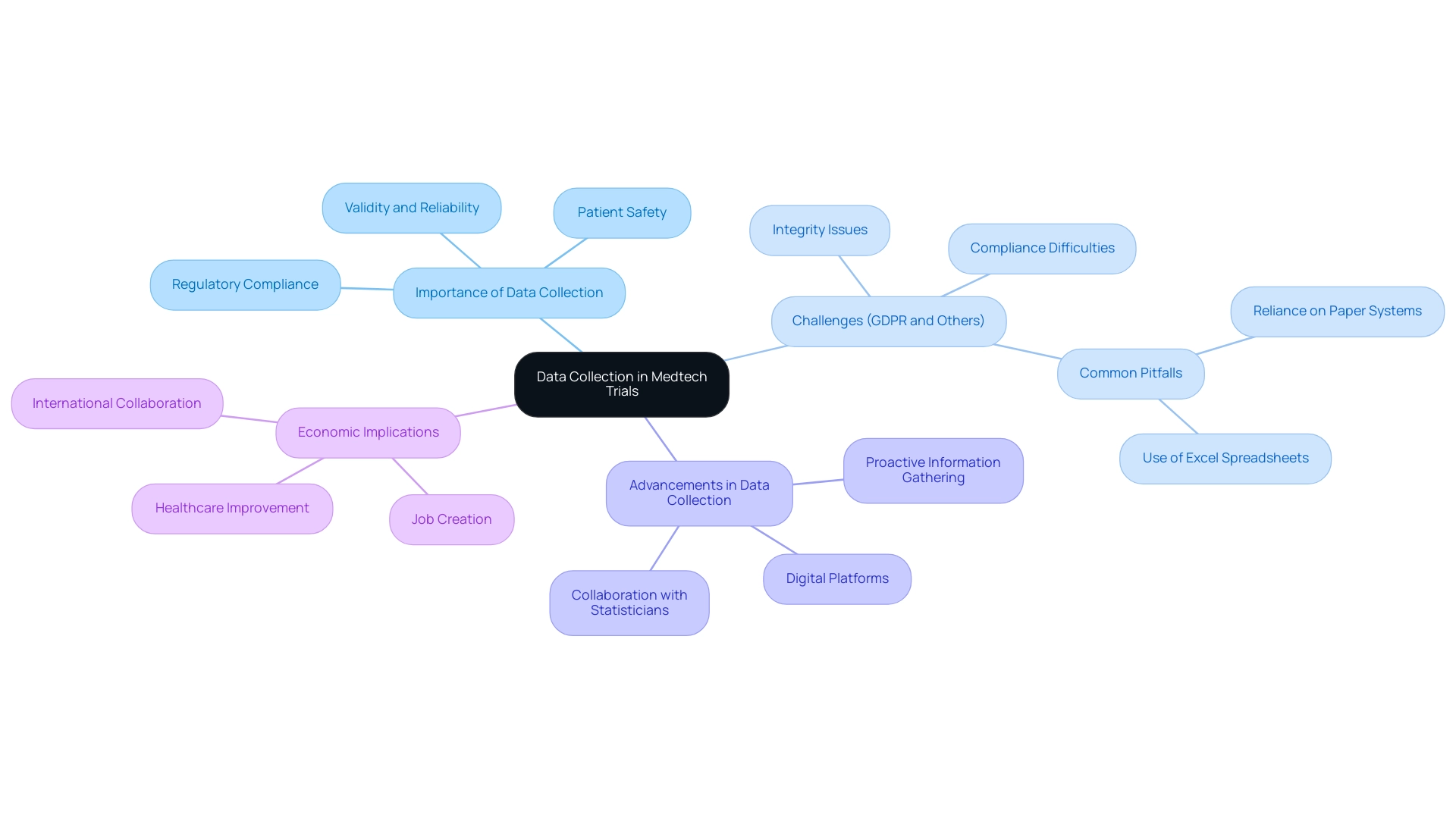
Challenges in Medtech Clinical Data Collection
Medtech studies encounter numerous obstacles in data collection during trials, primarily due to regulatory intricacies, diverse patient groups, and the increasing demand for real-time information capture. Regulatory requirements can vary greatly across regions, complicating the information-gathering process and necessitating a thorough understanding of local regulations to ensure compliance. This complexity is further exacerbated by the heterogeneity of patient demographics, introducing variability in information quality and complicating the analysis of trial outcomes.
Moreover, traditional methods of data collection in medtech trials, such as paper forms, are often fraught with errors and inefficiencies, leading to concerns about data integrity. A recent study highlighted that costs associated with securing and storing healthcare information remain disproportionately high compared to the expenses of information generation, underscoring the urgent need for more efficient management solutions. This statistic emphasizes the necessity for Medtech companies to adopt innovative approaches to information handling.
The Grand Challenges for Medtech Analytics case study illustrates these issues, revealing that obstacles such as heterogeneous information, small patient cohort sizes, and privacy concerns hinder data collection in medtech trials and analysis. The study emphasizes the necessity for robust methodologies and improved validation techniques to navigate these obstacles successfully.
As the landscape of Medtech evolves, the importance of data collection in medtech trials through electronic information capture platforms becomes increasingly clear. These platforms enhance data collection by enabling real-time information gathering and improving patient involvement and adherence, ultimately resulting in superior quality information. In 2025, addressing collection challenges will be paramount, as the demand for innovative medical devices and diagnostics continues to rise.
Expert opinions highlight that navigating regulatory complexities is crucial for successful trial outcomes. As Prof. Dan Ariely aptly noted, "Big information is like teenage sex: everyone talks about it, nobody really knows how to do it, everyone thinks everyone else is doing it, so everyone claims they are doing it…" This quote underscores the prevalent challenges in effectively managing big information within the Medtech sector. With the right strategies in place, Medtech companies can effectively address these challenges, ensuring that data collection in trials is both compliant and efficient.
At bioaccess, our extensive research study management services encompass feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting. We employ specific methodologies such as risk-based monitoring and adaptive study designs to enhance the quality of our research. By leveraging technology and adopting a strategic approach to information management, organizations can improve the quality of their clinical trials through effective data collection, contributing to the advancement of medical devices that enhance patient outcomes.
Furthermore, our commitment to information protection ensures that client trust is maintained throughout the process, with clear grievance procedures in place to address any concerns. For any queries regarding information protection, clients can reach out to our Grievance Officer at bioaccess, ensuring transparency and adherence to applicable laws.
Additionally, the concept of a continuous learning healthcare system is relevant here, proposing a framework for bridging the gap between research and operational healthcare. By employing information effectively, this system aims to enhance quality and results in Medtech studies, further emphasizing the significance of data collection and efficient information management. Understanding the role of INVIMA and insights from experts like Katherine Ruiz can provide valuable context for navigating the regulatory landscape in Colombia.
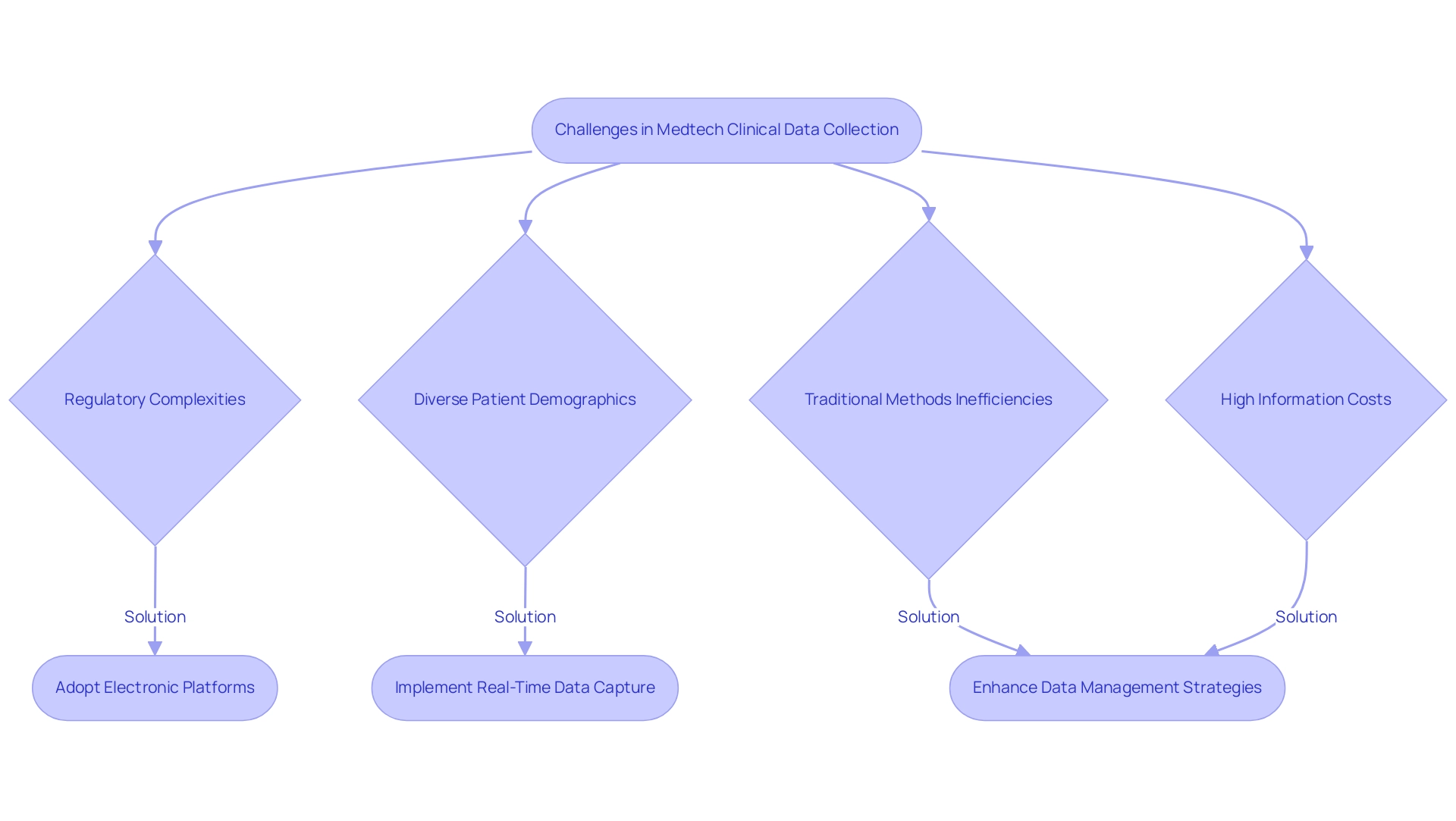
Fundamentals of Effective Data Management in Medtech Trials
Effective information management is paramount in medtech trials, relying heavily on data collection that hinges on several essential components. Establishing clear protocols for data collection is crucial, as it guarantees consistency and precision throughout the study. Employing standardized information entry methods streamlines the process and significantly reduces the risk of errors.
Moreover, implementing robust information validation processes enhances the integrity of the information collected. A comprehensive management plan must delineate the roles and responsibilities of team members, specify the tools and technologies to be employed, and outline the timelines for collection and analysis. Regular training sessions for staff on information management practices are vital; they bolster compliance and significantly reduce the likelihood of errors. Statistics indicate that organizations with well-structured information management plans see a marked improvement in compliance rates, which is essential in the highly regulated Medtech environment.
The integration of advanced information management tools, such as Electronic Information Capture (EIC) systems and Clinical Trial Management Systems (CTMS), has become increasingly prevalent. Recent trends reveal that 73% of pharmaceutical firms now employ cloud-based research management systems, a notable rise from 45% five years prior, according to Sarah Lee. This shift underscores the growing recognition of the importance of efficient information management strategies in medical research.
A case study of bioaccess® illustrates this point effectively. As a leading contract research organization in Latin America, bioaccess® successfully bridges the gap between innovative Medtech companies and the potential for conducting clinical research. Their expertise in managing various study types—including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies—demonstrates their capability to navigate the complexities of the Latin American Medtech landscape.
By implementing robust information management practices, bioaccess® has facilitated the advancement of medical devices from initial studies to market entry. Notably, Datavant serves 75% of the 100 largest health systems, highlighting the widespread adoption of effective information management tools in the industry. By prioritizing these fundamentals, organizations can ensure that data collection in medtech trials is not only efficient but also compliant with regulatory standards, ultimately facilitating the successful advancement of medical devices from initial studies to market entry.
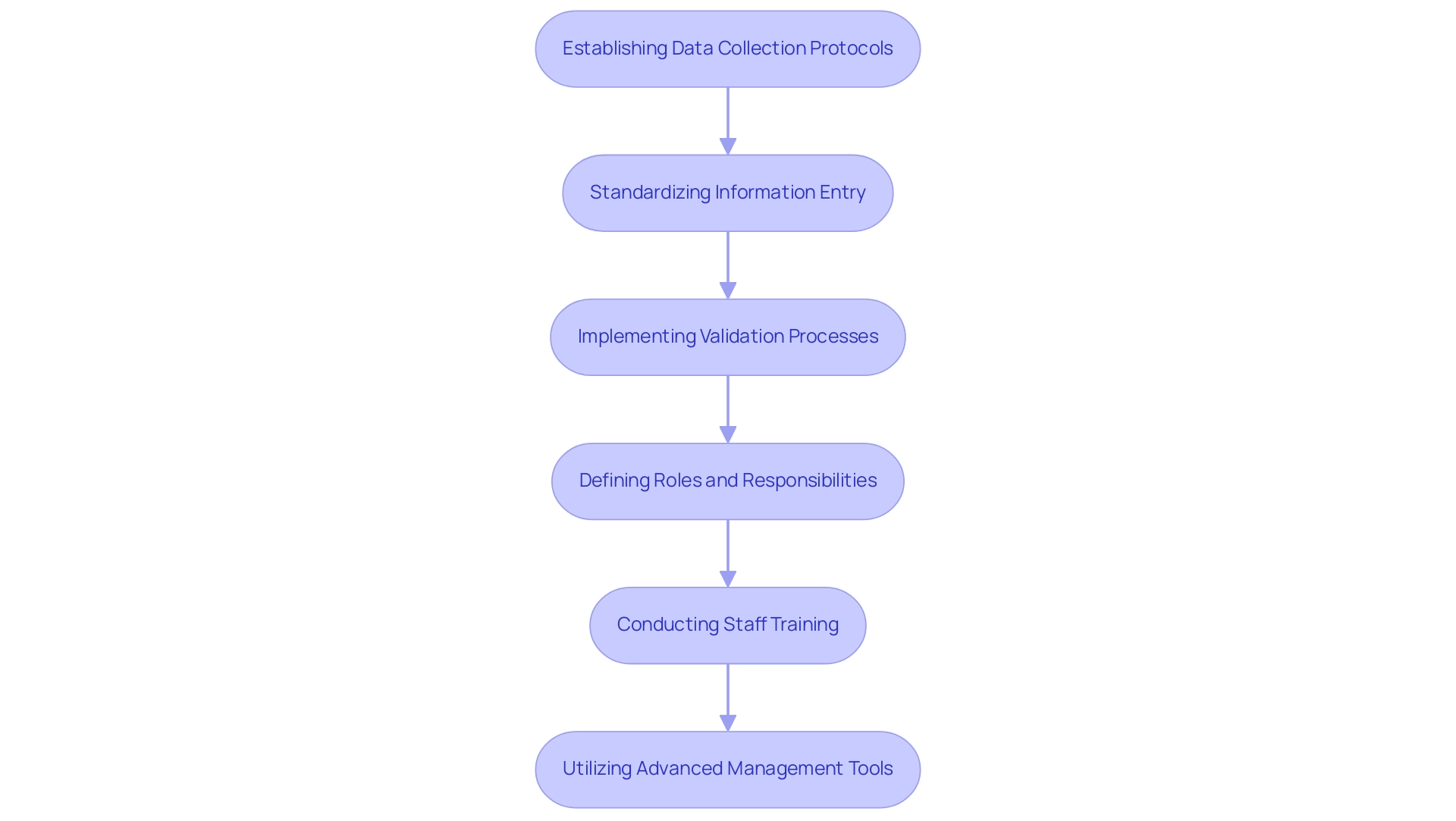
Proven Strategies for Optimizing Data Collection
To enhance data collection in medtech trials, organizations must prioritize the adoption of electronic information capture (EDC) systems. These systems not only streamline the entry process but also significantly reduce the risk of human error, which is crucial for maintaining integrity. Statistics indicate that EDC systems can lower overall study expenses by expediting study initiation and substantially shortening the time to study closure, making them a worthy investment for clinical research.
Integrating mobile technology further elevates real-time information gathering, which is essential for increasing patient involvement and adherence. By allowing participants to submit information directly from their devices, researchers can gather more precise and timely insights, ultimately enhancing the quality of trial results.
Implementing a centralized information management platform is another critical strategy. This approach facilitates seamless integration and analysis, empowering researchers to make informed decisions swiftly. Regular audits and feedback loops are vital for pinpointing areas that require improvement in the information collection process.
Such practices ensure that information quality is consistently enhanced, aligning with the evolving standards of medical research.
A case study on the integration of Risk-Based Monitoring (RBM) within EDC systems illustrates the effectiveness of these strategies. By identifying risks early and implementing targeted monitoring strategies, the study demonstrated improved patient safety and compliance with study protocols. This integration led to more efficient monitoring processes and quicker responses to potential safety issues, showcasing the advantages of merging RBM with EDC technology in enhancing information collection.
Furthermore, as bioaccess® aims to connect innovative Medtech firms with opportunities for conducting research studies in Latin America, the implementation of EDC systems becomes increasingly essential. With over 20 years of experience in the Medtech sector, bioaccess® offers expedited medical device research services, including Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies, ensuring strong information security is paramount. Organizations should enforce stringent login ID and password requirements, including restrictions on reuse, minimum length, frequency of changes, and automatic log-off.
As noted by Angelica Neisa, larger studies are more likely to adopt EDC, reinforcing the argument for its integration in Medtech studies. Overall, leveraging EDC systems and mobile technology, along with a robust information management strategy, positions Medtech organizations to achieve enhanced quality and operational efficiency in their clinical studies. This is vital for effective data collection in medtech trials, especially with the expertise and tailored approach of bioaccess® guiding the process.
Ensuring Regulatory Compliance in Data Collection
Regulatory adherence stands as a fundamental aspect of successful Medtech studies, with data collection in medtech trials ensuring that information gathering techniques align with established guidelines and standards. Organizations must be well-versed in pertinent regulations, including Good Clinical Practice (GCP) and local regulatory requirements, to maintain compliance throughout the study process. Adhering to GCP guidelines not only bolsters the reliability of clinical information but also cultivates trust among stakeholders.
To achieve this, implementing standardized operating procedures (SOPs) for data collection in medtech trials is crucial. SOPs help maintain consistency and accuracy across all trial phases, reducing variability that could compromise study outcomes. Furthermore, regular training sessions for staff on compliance matters and information handling practices are essential. Such training fosters a culture of accountability and integrity within research teams, ensuring that all personnel are equipped to uphold the highest standards of information management.
A notable example of GCP adherence can be seen in the NESIC study, which evaluated the effectiveness of neuromuscular electrical stimulation for patients with intermittent claudication. The idea for conducting the systematic review emerged while developing the Statistical Analysis Plan for the NESIC study, which was funded by the National Institute for Health Research. This study emphasized the significance of adherence in clinical studies and the necessity for robust statistical methods to analyze treatment effects.
The insights gained from this study underscore the value of meticulous data management practices, especially in the context of data collection in medtech trials, to achieve reliable results. Looking ahead to 2025, as regulatory landscapes evolve, it is imperative for Medtech organizations to stay updated on compliance requirements. This includes understanding the latest FDA guidance, which clarifies evaluation criteria for specific device types. Additionally, bioaccess® highlights the significance of feasibility and choice of research locations, principal investigator (PI) selection, and adherence to country-specific regulations to ensure smooth study operations.
Reporting on study status, inventory, and significant and non-significant adverse events is also essential for upholding transparency and accountability. By emphasizing regulatory compliance and GCP adherence, organizations can improve the quality of their research studies, ultimately speeding up the journey to market for innovative medical devices. Moreover, the negative entry order gradient for device approvals suggests that delays primarily affect the first entrant in a device product code, underscoring the need for timely compliance to navigate the competitive landscape. bioaccess® is dedicated to tackling client issues related to information protection and compliance, guaranteeing transparency and trust during the process.
If you have any queries or concerns about the processing of your information, you may contact our Grievance Officer at IMH ASSETS CORP (doing business as 'bioaccess®'), 1200 Brickell Avenue, Suite 1950 #1034, email: info@bioaccessla.com. We will address your concerns in accordance with applicable law.
Leveraging Technology for Enhanced Data Collection
The incorporation of technology into data collection in medtech trials is revolutionizing the efficiency and precision of these processes, particularly within the expedited medical device clinical study services provided by bioaccess® in Latin America. With over 20 years of experience in Medtech, bioaccess® employs electronic information capture (EDC) systems, mobile applications, and wearable devices to streamline data collection in medtech trials, significantly enhancing patient engagement. These advanced technologies enable real-time information entry and monitoring, effectively reducing reliance on manual processes that are often prone to errors.
Moreover, utilizing analytics tools for data collection in medtech trials allows researchers to uncover trends and insights from the gathered information, facilitating more informed decision-making throughout the process. A recent study highlighted that 30 promising molecules were selected for further drug development evaluation, underscoring the critical role of robust information management in fostering medical innovations.
The numerous advantages of EDC systems become particularly evident in data collection for medtech trials. They not only enhance information accuracy but also improve experimental efficiency by automating routine tasks and minimizing the time spent on data entry. As Ken Getz, Deputy Director and Professor, notes, 'The variety and amount of medical information is also increasing rapidly, and the tools for managing these must enhance to keep up.' This statement emphasizes the challenges faced in research studies, especially as the complexity of data escalates.
Mobile applications have emerged as essential tools in clinical research, facilitating data collection in medtech trials by bolstering participant engagement and retention. For instance, AI-driven platforms utilize linguistic pattern recognition to analyze patient feedback, predicting behaviors such as the likelihood of early dropout. This predictive capability empowers study designers to proactively adjust protocols, ultimately enhancing the patient experience and retention rates.
A case study titled "How AI Helps Patient Engagement in Clinical Trials" illustrates this point, demonstrating how AI can bolster patient engagement through a better understanding of participant feedback.
As we advance into 2025, the impact of technology on data collection in medtech trials will continue to evolve, with electronic data capture systems playing a pivotal role in improving study outcomes. It is crucial to recognize that research studies comprise four phases:
- Phase I assesses safety on a small group
- Phase II evaluates efficacy and safety on larger groups
- Phase III continues to evaluate efficacy and safety
- Phase IV monitors post-approval
The ongoing integration of these technologies not only simplifies processes but also enhances data collection in medtech trials, promoting a more patient-focused approach and ensuring that studies are both efficient and effective.
However, it is vital to acknowledge the limitations of AI in medical studies, which necessitate collaborative efforts to address ethical, regulatory, and practical challenges. With bioaccess®'s expertise and tailored approach in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies, the company is well-positioned to navigate these complexities and drive successful outcomes in the ever-evolving landscape of Medtech clinical trials.
Incorporating Patient Perspectives in Data Collection
Incorporating patient viewpoints during information gathering is essential for ensuring that the assessed results align with what truly matters to patients. Engaging patients in the design of data collection tools for medtech trials—such as surveys and questionnaires—significantly enhances the relevance and clarity of the gathered information. By actively involving patients, researchers can tailor these tools to more accurately capture the nuances of patient experiences and treatment effectiveness.
Utilizing patient-reported outcomes (PROs) as primary measures can yield invaluable insights, especially for conditions that lack objective metrics. PROs can serve as primary outcome measures for such conditions, highlighting their importance in research. A recent initiative involving patient organizations, including WECAN, has aimed to establish international standards for PROs in cancer research.
This project seeks to create uniform standards for the design, analysis, interpretation, and presentation of PRO information, ultimately enhancing the quality of research in oncology.
The impact of PROs on study results is profound; studies indicate that prioritizing patient perspectives in data collection during medtech trials improves data quality, leading to findings that are more applicable to real-world settings. Furthermore, statistics demonstrate that including patient viewpoints can enhance patient involvement and satisfaction, both of which are critical for the success of research studies. As Paul Robinson noted, "What is evident is that chances to enhance PE in research studies have improved—but more needs to be accomplished."
By fostering a collaborative environment where patients are viewed as partners in the research process, researchers can ensure that the outcomes of clinical studies are not only scientifically robust but also resonate with the needs and expectations of the patient community.
Key Takeaways and Best Practices for Successful Data Collection
To achieve successful data collection in Medtech trials, organizations should prioritize the following best practices:
- Establish Clear Protocols and Standardized Methods: Developing well-defined information collection protocols is crucial. Utilizing standardized methods not only enhances consistency but also improves the reliability of the information gathered. This approach minimizes variability and ensures that all participants are assessed uniformly, which is essential for valid results.
- Leverage Technology for Efficiency: Implementing advanced health information management systems (CDMS) can significantly streamline information entry processes. These specialized software applications enable the effective gathering, storage, and management of clinical research information, ultimately improving patient involvement and decreasing the chances of mistakes.
- Ensure Regulatory Compliance: Regular training and strict adherence to regulatory guidelines are vital for maintaining compliance throughout the trial. Organizations should conduct periodic reviews and updates of their training programs to reflect the latest regulations and best practices, ensuring that all team members are well-informed. This includes understanding the requirements set forth by INVIMA, the Colombia National Food and Drug Surveillance Institute, which plays a critical role in medical device oversight and compliance as a Level 4 health authority by PAHO/WHO.
- Include Patient Viewpoints: Involving patients in the information gathering process can result in more pertinent and high-quality insights. By understanding patient experiences and perspectives, organizations can customize their information gathering strategies to better capture the nuances of patient responses, thereby enhancing the overall quality of the information.
- Continuous Monitoring and Auditing: Regularly monitoring and auditing collection processes is essential for identifying areas that require improvement. This proactive method enables organizations to tackle potential problems before they worsen, ensuring the integrity and quality of the information gathered. Comprehensive project management and reporting practices are crucial in this regard, as they provide insights into study status, inventory, and adverse events.
By implementing these strategies, organizations can significantly enhance the quality and integrity of their clinical trials, leading to more valid and generalizable results. For instance, an expert panel agreed on 63 items to be included in the guidance for best practices, underscoring the importance of comprehensive protocols. Additionally, a case study highlighted the consequences of neglecting post-trial information management, which resulted in the loss of valuable information and hindered future research efforts.
To combat this, robust post-trial information management procedures, including archiving and secure storage, were recommended to ensure long-term accessibility while complying with regulatory requirements. As Meri Beckwith, Co-Founder of bioaccess, stated, "An adequately powered study ensures that the results are not only valid but also generalizable." Ultimately, these best practices not only enhance data quality but also contribute to the overall success of Medtech studies, especially regarding data collection in medtech trials.
For tailored support in your clinical trial needs, contact bioaccess to book a meeting and explore how our comprehensive services, including trial setup, import permits, and project management, can enhance your research outcomes.
Conclusion
Effective data collection is paramount for the success of Medtech clinical trials, serving as the cornerstone for regulatory compliance and research integrity. The transition from traditional methods to advanced electronic data capture systems and mobile technologies has significantly enhanced data accuracy, reduced costs, and improved patient engagement, leading to more reliable trial outcomes.
Incorporating patient perspectives into the data collection process is essential. Engaging patients in the design of data collection tools and emphasizing patient-reported outcomes provide insights that enrich the relevance and applicability of trial results.
As the Medtech landscape evolves, strict adherence to regulatory standards and best practices is critical. Ongoing training, vigilant monitoring, and robust data management strategies are necessary to navigate the complexities of clinical trials. By prioritizing these elements, Medtech organizations can effectively advance innovative medical devices while enhancing patient outcomes.
In conclusion, prioritizing effective data collection strategies is crucial for Medtech companies aiming to excel in a competitive market. Embracing technology, fostering patient collaboration, and ensuring regulatory compliance will drive successful clinical trials, placing patient safety and innovation at the core of medical device development.
Frequently Asked Questions
What is the importance of data collection in medtech trials?
Data collection in medtech trials is essential as it provides the evidence necessary for regulatory submissions and product development. It ensures the validity and reliability of research study results, which is crucial for assessing the safety and efficacy of medical devices.
How has GDPR affected data collection in medtech studies?
Since the implementation of GDPR in 2018, MedTech firms conducting clinical studies in the EU have faced new responsibilities that complicate data collection processes, necessitating compliance with stricter regulations.
What are common pitfalls in data collection for medtech trials?
Common pitfalls include reliance on paper-based systems or Excel spreadsheets, which can lead to integrity issues and challenges in compliance with regulations.
How have advancements in technology improved data collection in medtech trials?
Recent advancements, particularly the transition to digital platforms, have improved data collection by enhancing management processes and bolstering the overall integrity of experiments.
Why is effective information gathering crucial in medtech trials?
Effective information gathering techniques enable early identification of potential risks in the testing process, which boosts patient safety and enhances the integrity of the study.
What role do collaboration and expertise play in data collection for medtech trials?
Collaboration between medical researchers and statisticians is essential for understanding information needs and improving the analysis process, ensuring successful and compliant research studies.
What components are included in a comprehensive research management service for medtech trials?
A comprehensive research management service includes feasibility studies, site selection, compliance evaluations, setup, import permits, project oversight, and reporting.
What are the economic implications of medtech clinical studies?
Medtech clinical studies have significant economic implications, including job creation and healthcare improvement, highlighting the need for international collaboration in the field.
What obstacles do medtech studies face in data collection?
Medtech studies encounter obstacles such as regulatory intricacies, diverse patient groups, and the demand for real-time information capture, which complicate the data collection process.
How do traditional data collection methods impact data integrity in medtech trials?
Traditional methods, such as paper forms, are often inefficient and error-prone, leading to concerns about data integrity and the need for more efficient management solutions.
What strategies can medtech companies adopt to improve data collection?
Medtech companies can adopt innovative approaches, such as electronic information capture platforms, to enhance real-time data gathering, improve patient involvement, and ensure superior quality information.
How does bioaccess support medtech trials?
Bioaccess provides extensive research study management services, employing methodologies like risk-based monitoring and adaptive study designs to enhance research quality and ensure compliance throughout the trial process.

