Overview
The article emphasizes best practices for effective clinical trial design within the medtech industry, highlighting the critical nature of:
- Strategic planning
- Regulatory compliance
- Innovative technologies
A well-structured trial design, which incorporates elements such as:
- Randomized controlled studies
- Advanced data management practices
is essential for generating reliable evidence regarding medical devices and enhancing patient outcomes. In the evolving landscape of medtech, understanding these best practices is paramount for addressing the challenges faced in clinical research.
Collaboration among stakeholders is vital to navigate regulatory complexities and leverage technological advancements effectively. Ultimately, the integration of these strategies not only improves trial efficiency but also fosters innovation that can significantly benefit patient care.
Introduction
In the rapidly evolving Medtech landscape, the design and execution of clinical trials hold unprecedented significance. Organizations are tasked with validating the safety and efficacy of innovative medical devices while navigating a complex web of regulatory requirements, strategic planning, and patient engagement.
With randomized controlled trials (RCTs) recognized as the gold standard for research integrity, the emphasis on meticulous trial design has surged, propelled by technological advancements and an increasing focus on sustainability.
This article explores the fundamental aspects of clinical trial design, underscoring the critical importance of strategic planning, regulatory compliance, and innovative methodologies to address the myriad challenges inherent in clinical research management.
By examining these key elements, it becomes clear how Medtech companies can enhance trial efficiency and patient outcomes, ultimately shaping the future of healthcare.
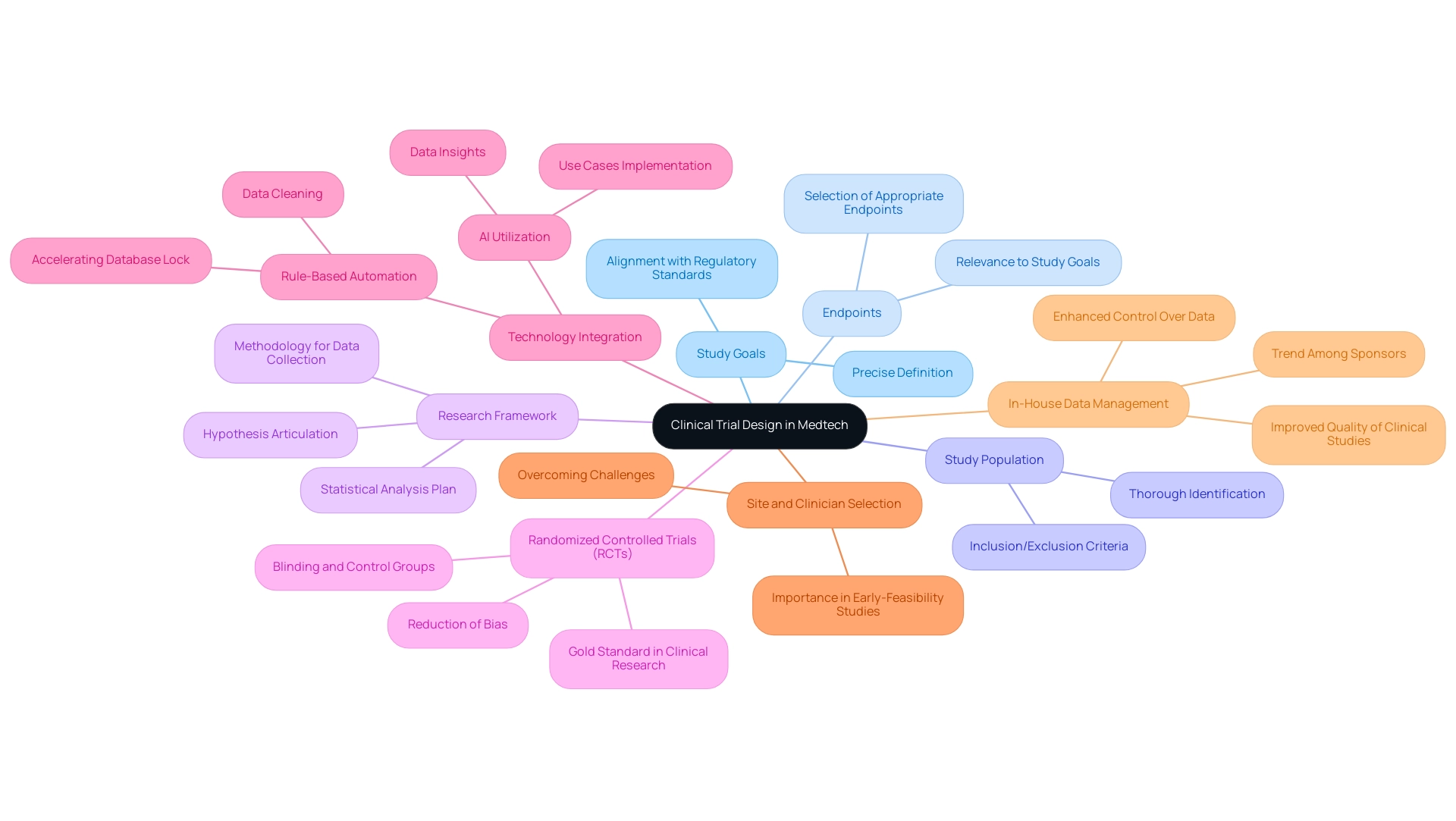
Fundamentals of Clinical Trial Design in Medtech
The effective clinical trial design for medtech hinges on several crucial elements, including the precise definition of study goals, the selection of appropriate endpoints, and the thorough identification of the study population. A robust research framework is essential, articulating the hypothesis, outlining the methodology for data collection, and detailing the statistical analysis plan. In 2025, effective clinical trial design for medtech, particularly through randomized controlled studies (RCTs), continues to represent the gold standard in clinical research, significantly reducing bias and enhancing the reliability of findings.
Incorporating elements such as blinding and control groups further strengthens the study's design, ensuring that observed outcomes can be confidently attributed to the intervention rather than external influences.
Recent statistics highlight the increasing reliance on RCTs, with a notable uptick in their application across various Medtech studies. This trend is underscored by insights from industry experts, who assert that effective clinical trial design for medtech is vital for generating valid scientific evidence regarding the safety and effectiveness of medical devices. For instance, GSK is already leveraging rule-based automation for information cleansing to expedite the time to database lock, illustrating the integration of technology in trial design.
Moreover, the shift towards internal information management procedures among sponsors reflects a broader trend aimed at enhancing control over information quality and improving trial outcomes. By conducting studies internally, sponsors can ensure higher quality results for patients while maintaining direct access to real-time information, thereby enhancing overall research efficiency. The significance of proper site and practitioner selection cannot be overstated, particularly in early-feasibility studies (EFS), where challenges may arise if not adequately addressed.
As emphasized by Eileen Mihas from MDIC, effective site and clinician selection is crucial for overcoming challenges in the EFS program. Additionally, Danish Mairaj, principal engineer of medical device design at RESYCA, notes that this area can greatly benefit from AI's data utilization, but insights from this data can only be gleaned if specific use cases are implemented in relevant facilities. By utilizing the comprehensive research management services offered by bioaccess®, which include expertise in effective clinical trial design for medtech such as Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies—and backed by over 20 years of experience in the Medtech sector—bioaccess® provides the flexibility and specialized knowledge necessary to navigate the complexities of research and accelerate the advancement of innovative medical devices.
Strategic Planning: The Key to Successful Clinical Trials
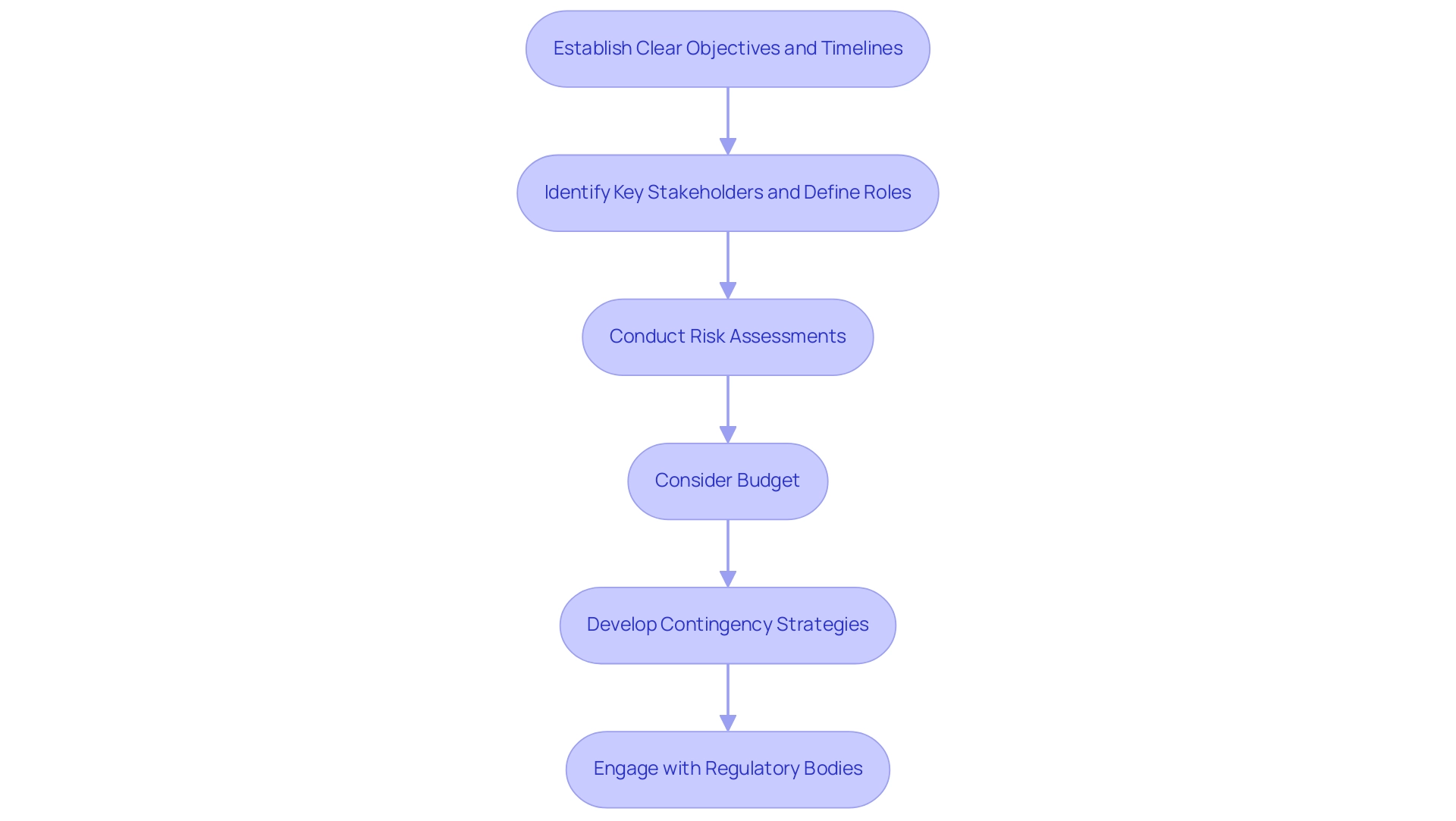
Strategic planning is essential for effective clinical trial design in medtech, serving as a roadmap that directs the entire process. It commences with the establishment of clear, measurable objectives and timelines, which are vital for tracking progress and ensuring accountability. Early identification of key stakeholders and the definition of their roles foster collaboration and enhance communication, both of which are crucial for success.
A robust strategic plan must encompass:
- Risk assessments
- Budget considerations
- Contingency strategies to address potential setbacks
Engaging with regulatory bodies from the outset clarifies requirements and facilitates smoother approval processes, ultimately saving time and resources. In fact, 77% of industry leaders anticipate a greater regulatory focus on sustainability in 2025, underscoring the necessity for proactive engagement with regulatory frameworks.
This regulatory emphasis highlights the importance of strategic planning aligned with evolving industry standards. Furthermore, the incorporation of project management tools is increasingly recognized as a best practice in research study design. These tools streamline communication among team members and enhance collaboration, ensuring that all participants are aligned with the project's objectives. For instance, bioaccess provides extensive research management services, including:
- Feasibility studies
- Site selection
- Compliance reviews
- Study setup
- Import permits
- Project management
- Reporting on study status
- Serious and non-serious adverse events
This holistic approach aids in mitigating challenges faced by medical device startups, such as regulatory hurdles and recruitment issues. As Vivienne van der Walle, Founder and Medical Director, aptly stated, "Anything that takes away time from patients is a pain point for a site, and anyone who resolves that is helping patient care." This perspective reinforces the necessity of strategic planning that prioritizes efficiency and patient outcomes.
As the life sciences sector undergoes transformation propelled by digital progress and scientific breakthroughs, the significance of strategic planning in research studies cannot be overstated. With 83% of non-US respondents expecting the EU's Corporate Sustainability Reporting Directive to significantly influence their strategies in 2025, Medtech companies must adapt their planning processes accordingly. By prioritizing strategic planning and stakeholder involvement, Medtech organizations can enhance their chances of conducting successful studies, which rely on effective clinical trial design, ultimately advancing medical devices that improve patient outcomes.
To learn more about how bioaccess can assist you, BOOK A MEETING today.
Navigating Regulatory Compliance in Clinical Trials
Navigating regulatory compliance is essential for effective clinical trial design in medtech research within the industry. Companies must thoroughly understand the guidelines established by regulatory authorities such as the FDA and EMA, which dictate the conduct of research studies. This encompasses a comprehensive grasp of Good Clinical Practice (GCP) standards, informed consent processes, and the reporting requirements for adverse events.
For instance, thorough documentation and adherence to protocols are essential in reducing the risks linked to non-compliance.
In 2025, a significant 77% of respondents in the U.S. expect an increased regulatory focus on sustainability, reflecting a broader trend towards responsible medical practices. Regular training for research staff on the latest regulatory updates is vital to keep pace with these evolving standards. Moreover, innovations focused on enhancing site experiences in research trials, such as streamlined information entry processes and platform-based methods, have been demonstrated to improve site efficiency, ultimately aiding patient care and trial results.
As highlighted in a case study, clinical information leaders are prioritizing the site experience by streamlining entry processes, which has resulted in quicker queries and enhanced overall efficiency.
Leianne Ebert, Head of Clinical Information Operations at Alcon, emphasized the significance of timely information management, stating, "This is something we monitor regularly, and last week, our records indicated that 45% of our information is entered on the same day as the visit date." This underscores the critical role of efficient data practices in maintaining compliance and enhancing integrity.
Moreover, the integration of technologies like generative AI is anticipated to boost operational efficiencies and drive breakthrough innovations in the life sciences industry. By prioritizing regulatory compliance, Medtech companies not only enhance the integrity of their studies but also ensure effective clinical trial design for medtech, fostering trust with stakeholders and paving the way for successful product development and market entry. The optimism among global life sciences executives regarding personalized medicine and operational efficiencies further emphasizes the relevance of these compliance efforts in shaping the future of medical studies.
At bioaccess, we provide extensive clinical study management services, including:
- Feasibility studies
- Site selection
- Compliance reviews
- Setup
- Import permits
- Project management
- Reporting
Our methodologies involve a systematic approach to each phase of the process, ensuring that all regulatory requirements are met and that procedures are conducted efficiently. For instance, our compliance reviews are designed to identify potential regulatory pitfalls early in the process, allowing for timely adjustments that enhance the integrity of the study.
Our project management services ensure that all aspects of the process are coordinated effectively, leading to improved outcomes and adherence to timelines.
Our expertise, led by professionals like Katherine Ruiz, an expert in regulatory affairs for medical devices and in vitro diagnostics in Colombia, ensures that our clients navigate the complexities of regulatory compliance effectively. This dedication to regulatory excellence establishes bioaccess as a leader in Medtech research in Latin America, emphasizing innovation and quality.

Overcoming Challenges in Clinical Research Management
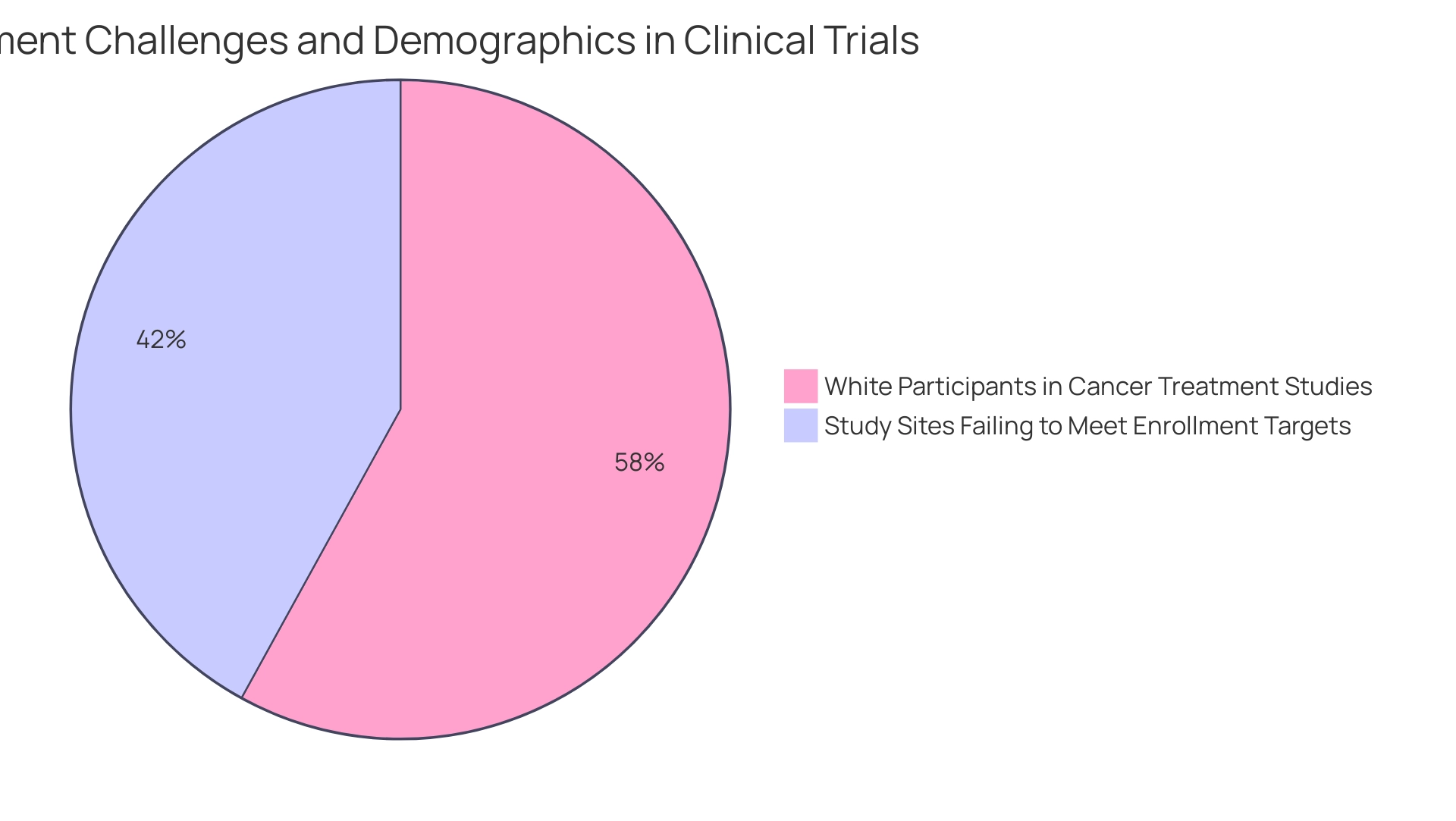
In the Medtech sector, effective clinical trial design is paramount due to the complexities of clinical research management, particularly in patient recruitment and retention, as well as the challenges of managing multiple projects concurrently. A significant concern is that approximately 60% of study sites fail to meet enrollment targets, with 11% not enrolling any patients at all, primarily due to ineffective recruitment strategies. This underscores the necessity of developing more effective outreach efforts to ensure adequate patient enrollment in clinical trials.
To address these challenges successfully, leveraging social media platforms and community outreach initiatives can significantly enhance awareness about research trials. Engaging with diverse communities is essential, especially considering the historical underrepresentation of certain demographics in research. For example, between 2001 and 2010, an alarming 82.9% of participants in cancer treatment studies were white, highlighting the urgent need for more inclusive recruitment practices.
Moreover, a quote from Antidote reveals that home visits were more appealing to Hispanic and non-white individuals compared to their non-Hispanic and white counterparts, emphasizing the importance of tailored recruitment strategies. A notable example of effective recruitment and retention strategies is the partnership between GlobalCare Clinical Trials and bioaccess™. This collaboration has successfully enhanced ambulatory services for research in Colombia, resulting in over a 50% reduction in recruitment duration and an impressive retention rate exceeding 95%. Such outcomes illustrate the potential of strategic partnerships in achieving effective clinical trial design, thereby improving research efficiency and effectiveness.
The extensive presence of bioaccess™ in Colombia has been instrumental in this success, providing localized support and outreach. Building strong relationships with healthcare providers is another critical strategy, as these connections can facilitate referrals and enhance patient engagement. By fostering trust and dialogue with local practitioners, Medtech companies can access a broader patient base, which is essential for clinical trial designs that genuinely reflect the populations they aim to support. Furthermore, adopting efficient project management methods, such as centralized data management systems, can streamline operations and enhance communication among team members.
This approach not only boosts efficiency but also ensures that all stakeholders are aligned in their objectives, ultimately leading to more successful outcomes. Comprehensive management services for research studies offered by bioaccess™, including feasibility assessments, site selection, compliance reviews, study setup, import permits, project management, and reporting, are vital for reliable, safe, and effective medical research, ensuring the integrity of the scientific process and prioritizing safety.
In summary, by proactively addressing recruitment and retention challenges through innovative strategies and strong partnerships—such as personalized communication, flexible monitoring options, and offering incentives—Medtech companies can significantly enhance the efficiency and effectiveness of their research studies. This, in turn, contributes to effective clinical trial design and drives global health improvement through international collaboration and innovation.
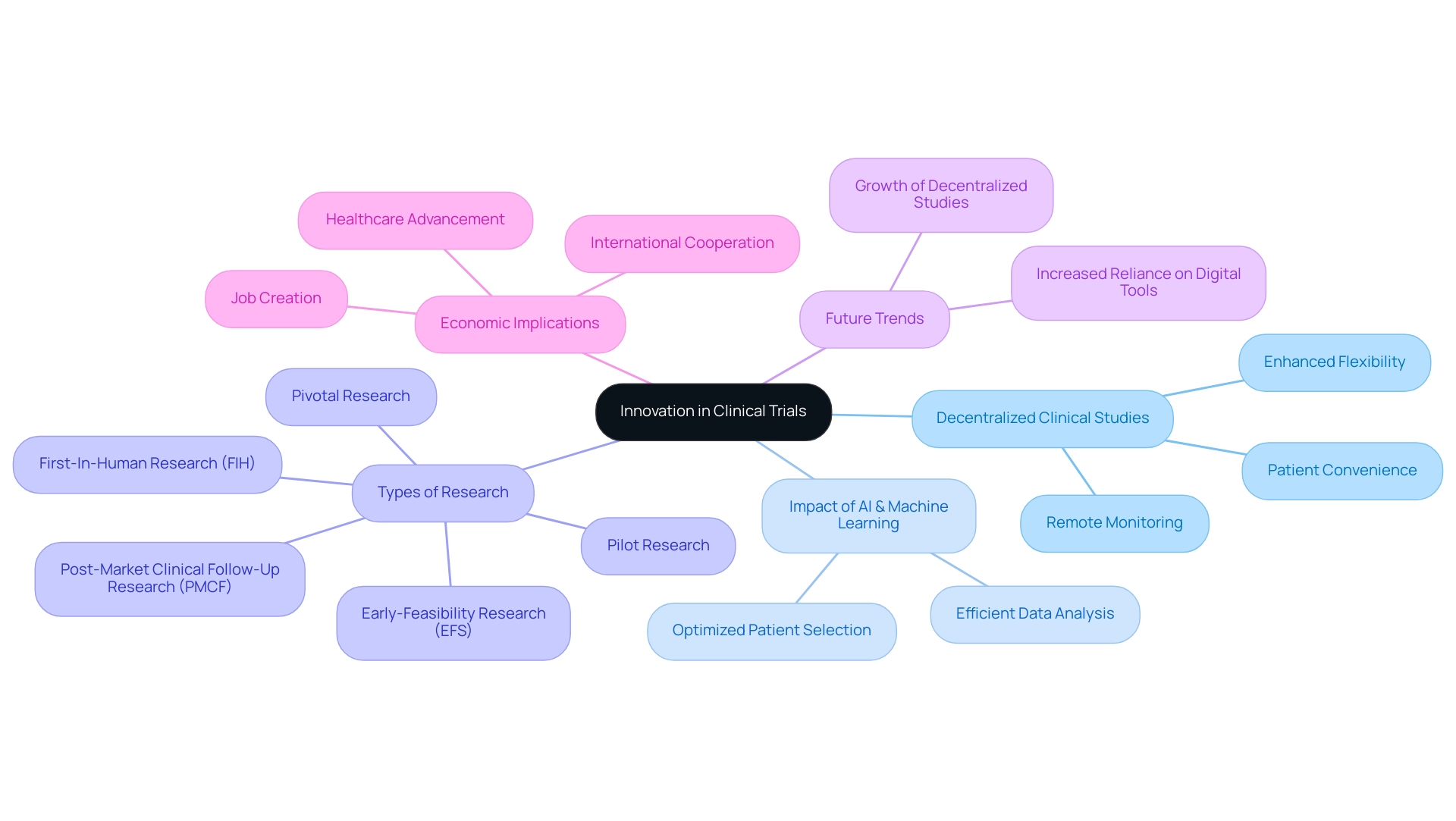
Embracing Innovation: The Future of Clinical Trials in Medtech
Innovation and technology are increasingly propelling the future of clinical studies in the Medtech sector. Effective clinical trial design for medtech is leading the way with decentralized clinical studies, which utilize digital tools and remote monitoring. This approach offers enhanced flexibility and convenience for patients. For instance, wearable devices play a crucial role in facilitating real-time information collection, significantly improving participant engagement and adherence.
Statistics indicate that the integration of such technologies can lead to a more streamlined information collection process. Companies like Alcon report that 45% of their records are entered on the same day as patient visits, enhancing the accuracy and timeliness of information. Leianne Ebert, Head of Clinical Data Operations at Alcon, noted, "This is something we monitor regularly, and last week, our records indicated that 45% of our information is entered on the same day as the visit date."
Furthermore, the integration of artificial intelligence (AI) and machine learning into study design is transforming patient selection and information analysis. These technologies enable Medtech companies to optimize their processes, ensuring that the right patients are enrolled and that data is analyzed efficiently. Consequently, adopting these innovations not only improves the efficiency of medical studies but also supports effective clinical trial design for medtech, thereby enhancing the overall patient experience.
This shift is crucial for achieving better outcomes and accelerating the time-to-market for new medical devices. In this context, bioaccess® provides extensive clinical management services that are crucial for navigating the complexities of clinical research. Their expertise encompasses various types of research, including:
- Early-Feasibility Research (EFS)
- First-In-Human Research (FIH)
- Pilot Research
- Pivotal Research
- Post-Market Clinical Follow-Up Research (PMCF)
This breadth of experience ensures that effective clinical trial design for medtech is utilized, allowing experiments to be conducted efficiently and effectively while addressing the unique challenges of each study type.
Looking ahead to 2025, the focus on decentralized clinical studies is expected to grow, driven by the need for patient-centric approaches and the increasing reliance on digital tools. The effect of these studies on patient convenience cannot be overstated, as they enable participation from the comfort of home, reducing barriers to entry and enhancing recruitment rates. However, challenges persist, especially concerning the dependence on spreadsheets for metadata management, which has proven challenging for companies striving to expand their collection processes.
As the Medtech industry continues to evolve, the integration of advanced technologies, alongside the comprehensive services provided by bioaccess®, will be essential for staying competitive and meeting the demands of a rapidly changing healthcare landscape. Furthermore, the positive effect of Medtech clinical research on local economies, including job creation and healthcare advancement, highlights the significance of these trials in promoting international cooperation and economic development. The case study on Alcon's evaluation of site experiences illustrates the practical implications of improving data entry processes and enhancing patient care.
Conclusion
Effective clinical trial design in the Medtech sector is crucial for ensuring patient safety and data integrity. The emphasis on randomized controlled trials (RCTs) as the gold standard underscores the necessity for strategic planning, which encompasses:
- Setting clear objectives
- Selecting appropriate endpoints
- Establishing a robust data management framework
With advancements in technology, Medtech companies increasingly leverage tools like artificial intelligence and automation to enhance trial efficiency and outcomes.
Navigating regulatory compliance is equally vital. Engaging with regulatory bodies early can streamline approval processes, while ongoing training ensures adherence to evolving guidelines. Collaborating with stakeholders and utilizing comprehensive clinical trial management services can effectively address challenges such as:
- Patient recruitment
- Retention
Ultimately improving trial quality.
The shift towards decentralized clinical trials signifies a major evolution in the industry. By harnessing digital tools and remote monitoring, companies can enhance patient engagement and expedite the development of new medical devices.
In conclusion, prioritizing meticulous planning, regulatory compliance, and innovative methodologies is essential for the success of clinical trials in the Medtech sector. By focusing on these key elements, organizations can enhance trial efficiency and patient outcomes, shaping the future of healthcare for improved global health.
Frequently Asked Questions
What are the key elements of effective clinical trial design in medtech?
Key elements include precise definition of study goals, selection of appropriate endpoints, thorough identification of the study population, a robust research framework, and a clear statistical analysis plan.
Why are randomized controlled studies (RCTs) considered the gold standard in clinical research for medtech?
RCTs significantly reduce bias and enhance the reliability of findings, making them essential for generating valid scientific evidence regarding the safety and effectiveness of medical devices.
What role does blinding and control groups play in clinical trial design?
Incorporating blinding and control groups strengthens the study's design by ensuring that observed outcomes can be confidently attributed to the intervention rather than external influences.
How is technology being integrated into clinical trial design?
Companies like GSK are leveraging rule-based automation for information cleansing to expedite the time to database lock, illustrating the integration of technology in trial design.
What is the significance of internal information management procedures among sponsors?
They enhance control over information quality and improve trial outcomes, allowing sponsors to ensure higher quality results for patients and maintain direct access to real-time information.
Why is site and practitioner selection important in early-feasibility studies (EFS)?
Proper site and clinician selection is crucial for overcoming challenges in EFS programs, as highlighted by industry experts.
How can AI benefit clinical trial design in medtech?
AI can utilize data effectively, providing insights that can enhance clinical trial design, but these insights depend on implementing specific use cases in relevant facilities.
What components should a robust strategic plan for clinical trial design include?
A robust strategic plan should encompass risk assessments, budget considerations, contingency strategies, and engagement with regulatory bodies.
What is the expected regulatory focus in 2025 according to industry leaders?
There is an anticipated greater regulatory focus on sustainability, emphasizing the need for proactive engagement with regulatory frameworks.
How can project management tools improve research study design?
These tools streamline communication among team members and enhance collaboration, ensuring that all participants are aligned with the project's objectives.
What services does bioaccess provide to assist in clinical trial design?
Bioaccess offers services including feasibility studies, site selection, compliance reviews, study setup, import permits, project management, and reporting on study status.
Why is strategic planning essential for medtech organizations?
Strategic planning enhances the chances of conducting successful studies, which are critical for advancing medical devices that improve patient outcomes.

