Overview
Early feasibility studies (EFS) under Brazilian regulations play a crucial role in evaluating the practicality of new healthcare devices. These studies not only help identify potential issues but also ensure compliance with the local regulatory standards established by ANVISA. By conducting EFS, companies can accelerate product development and enhance their chances of successful market entry. Furthermore, this process aligns seamlessly with Brazil's evolving healthcare landscape, which increasingly prioritizes innovation and patient safety.
Introduction
In Brazil's evolving medical device landscape, early feasibility studies (EFS) have emerged as a vital tool for assessing the viability of innovative technologies in real-world settings. These preliminary investigations not only provide essential insights into device safety and functionality but also assist Medtech companies in refining their products before larger clinical trials.
As Brazil grapples with unique healthcare challenges and diverse patient demographics, the role of EFS becomes even more critical, aligning with regulatory expectations and fostering a culture of innovation. Recent trends indicate a growing acknowledgment of the importance of these studies, paving the way for enhanced market readiness and patient safety in the medical technology sector.
With the increasing integration of advanced methodologies and regulatory improvements, the Brazilian market is poised for significant growth, making it an exciting time for researchers and manufacturers alike.
Understanding Early Feasibility Studies in Brazil's Medical Device Landscape
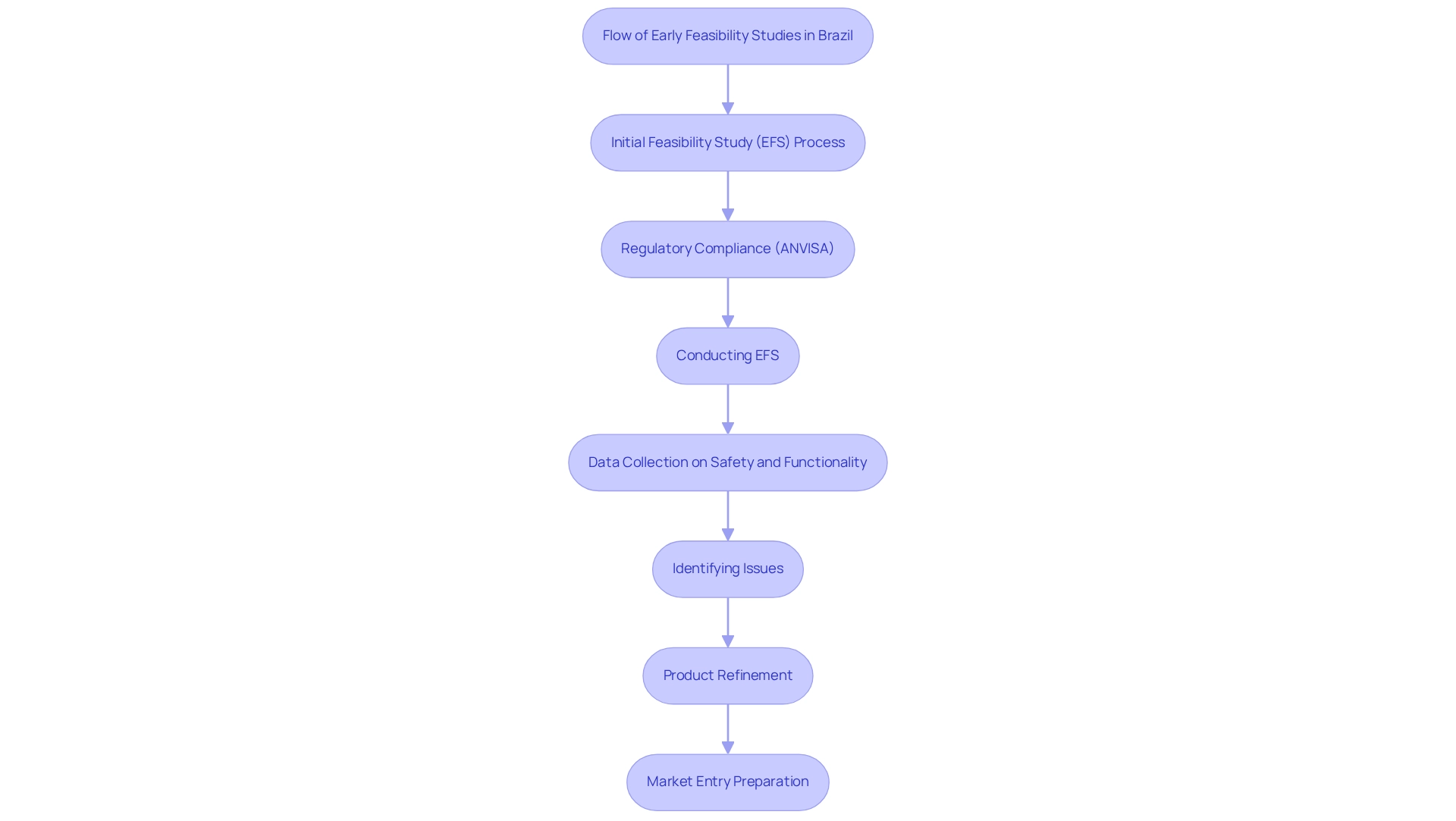
Initial feasibility evaluations (EFS) serve as critical preliminary assessments aimed at determining the practicality of new healthcare instruments in real-world environments. In Brazil, early feasibility studies conducted under Brazilian regulations are essential due to the country's diverse patient demographics and unique healthcare challenges. These analyses empower researchers to gather vital information regarding equipment safety, functionality, and usability before advancing to more extensive clinical trials.
Early feasibility studies under Brazilian regulations enable Medtech firms to proactively identify potential issues, refine their products, and significantly enhance their chances for successful market entry. This strategy not only accelerates the development timeline but also aligns with the regulatory expectations set forth by ANVISA, Brazil's health regulatory agency. Recent trends indicate a growing recognition of the importance of early feasibility studies within Brazil's healthcare industry.
Successful EFS have demonstrated the ability to streamline the path to commercialization, with companies reporting increased confidence in their products following these analyses. Furthermore, expert perspectives underscore that early feasibility studies under Brazilian regulations are vital for mitigating risks associated with new technologies, ultimately fostering innovation in the healthcare sector.
Data indicates that the Brazilian healthcare equipment market is increasingly embracing early feasibility studies, leading to a notable rise in applications for early-stage studies. In 2021, nearly 15,320 patent applications for healthcare technologies were submitted to the European Patent Office, with 41% originating from European countries. This trend reflects a global shift toward innovation, of which Brazil is an integral part, emphasizing a commitment to patient safety and device efficacy that aligns with global best practices.
As the regulatory landscape evolves, with new legislation proposed to facilitate and expedite regulatory approvals, early feasibility studies under Brazilian regulations are expected to receive heightened attention. This shift will create opportunities for stronger research frameworks that support the advancement of healthcare technologies in Brazil. Additionally, the challenges faced in Europe, where a unified procedural framework is lacking, highlight the importance of establishing a standardized approach for early feasibility studies under Brazilian regulations to enhance the efficiency of these evaluations. As noted by Hoffmann-La Roche Ltd, support for research in this domain is critical for promoting innovation and ensuring patient safety.
With bioaccess®'s expertise in managing EFS, First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), along with a tailored approach, companies can effectively navigate these complexities, ensuring compliance and successful outcomes.
Navigating Brazil's Regulatory Framework for Medical Devices
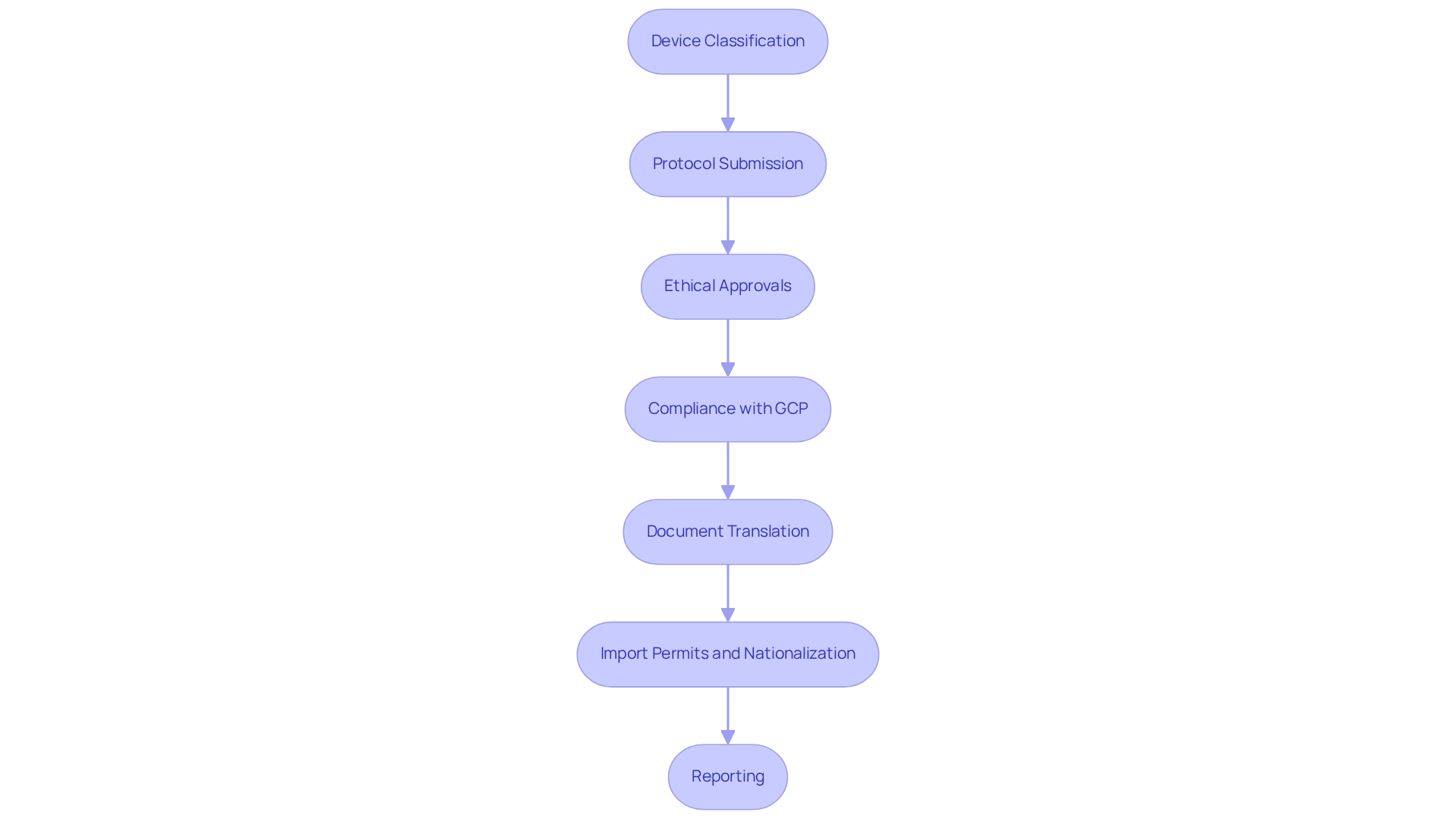
Brazil's regulatory framework for healthcare products is primarily overseen by ANVISA, the National Health Surveillance Agency, which plays a critical role in the approval and monitoring of clinical trials. To initiate a preliminary feasibility study, researchers must first categorize their health product according to ANVISA's established risk categories. This classification is pivotal, as it dictates the regulatory pathway and the specific documentation required for approval.
The process involves several key steps:
- Device Classification: Manufacturers must accurately determine the risk class of their device, as this influences the regulatory requirements and approval timeline. As Margret Seidenfaden, an expert in the field, states, "Manufacturers intending to market a medical device in Brazil must first determine the risk class of the device."
- Protocol Submission: A comprehensive research protocol must be submitted, detailing the objectives, methodology, and anticipated outcomes of the research.
- Ethical Approvals: Researchers must secure ethical approvals from local ethics committees, ensuring that the research complies with ethical standards and safeguards participant rights.
- Compliance with GCP: Following Good Clinical Practice (GCP) guidelines is crucial for preserving the integrity of the research and the validity of the results.
- Document Translation: It is also essential for manufacturers to translate all relevant documents into Portuguese, as this is a specific requirement for compliance in Brazil.
- Import Permits and Nationalization: Manufacturers must also secure import permits and nationalize investigational devices to comply with local regulations.
- Reporting: Continuous reporting on research status, including serious and non-serious adverse events, is essential for regulatory compliance and participant safety.
Understanding and navigating these regulations not only streamlines the execution of medical studies but also enhances the credibility of the research findings. Recent statistics indicate that the approval times for trials in Brazil have improved, reflecting ANVISA's commitment to enhancing efficiency while ensuring safety and quality standards. Furthermore, as regulatory frameworks evolve globally, Brazil is positioning itself as a competitive hub for healthcare innovation, attracting manufacturers looking to conduct clinical research in a supportive environment.
This transformation is underscored by the increasing incorporation of artificial intelligence and automation in Regulatory Affairs, which promises to further simplify processes for product approvals. Additionally, countries in the APAC, LATAM, and MENA regions are updating their regulations to attract innovation while maintaining safety and quality standards in healthcare, illustrating a broader trend that Brazil is part of.
In conclusion, knowledge of ANVISA's regulatory criteria and the processes for categorizing health products is crucial for researchers looking to conduct early feasibility studies under Brazilian regulations. By adhering to these guidelines, researchers can facilitate smoother research execution and contribute to the advancement of health technology. With bioaccess®'s expertise in clinical trial administration services—including feasibility assessments, site selection, compliance evaluations, import permits, reporting, and project oversight—researchers can effectively tackle these challenges and enhance their prospects for success in the competitive arena of medical equipment trials.
To learn more about how bioaccess® can assist you, BOOK A MEETING today.
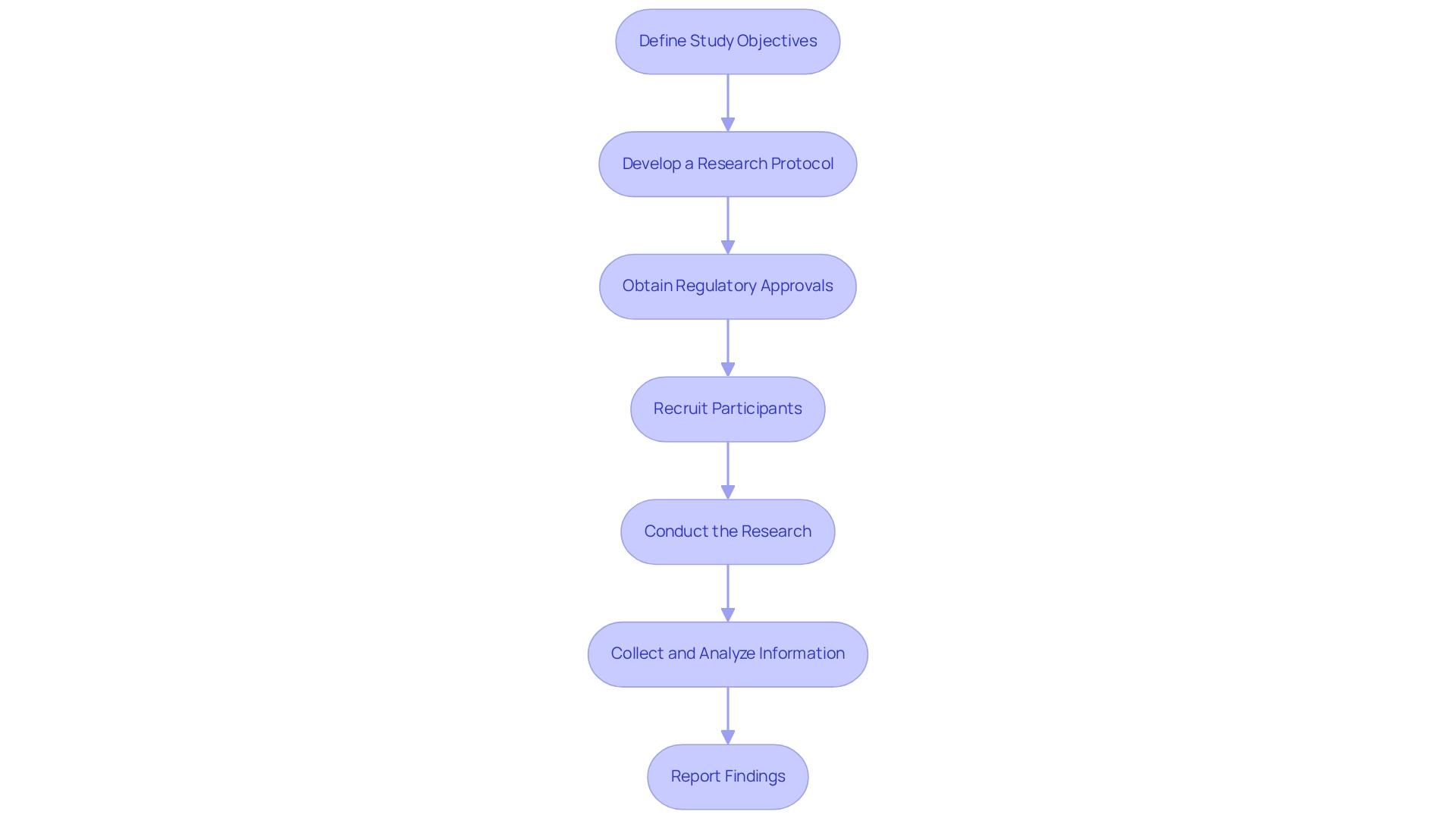
Step-by-Step Process for Conducting Early Feasibility Studies
-
Define Study Objectives: Clearly articulate the goals of the early feasibility studies under Brazilian regulations. This involves creating specific hypotheses that will direct the research and assist in assessing the potential effect on patient care. Consider the importance of accuracy and speed in skills, as highlighted by the Ignite Challenge, which can be crucial for assessing the device's effectiveness in real-world applications.
-
Develop a Research Protocol: Create a detailed research protocol that describes the research design, methodology, and statistical analysis plan. This document should outline the rationale behind the selected methods and how they correspond with regulatory requirements, including early feasibility studies under Brazilian regulations to ensure a robust framework for the research. Be mindful of potential limitations, such as non-probabilistic sampling and low retention rates, which may affect the generalizability of your findings. Bioaccess® can assist in developing this protocol to ensure compliance with local regulations.
-
Obtain Regulatory Approvals: Submit the research protocol to ANVISA and pertinent local ethics committees for approval. It is essential to verify that all documentation complies with regulatory standards in early feasibility studies under Brazilian regulations, as this step is important for the credibility and acceptance of the research. The regulatory landscape in Colombia, overseen by INVIMA, emphasizes the importance of compliance, classified as a Level 4 health authority by PAHO/WHO. Note that the lack of specific tools for assessing cognitive limitations during screening may pose challenges in participant recruitment and research design. Bioaccess®'s expertise in navigating early feasibility studies under Brazilian regulations can streamline this phase.
-
Recruit Participants: Identify and recruit a suitable cohort of participants that accurately reflects the target patient population. Effective recruitment strategies are essential to ensure that the sample size is adequate for statistical analysis and that the findings are generalizable. Consider the challenges faced in implementing new assessment tools, as noted by Nadia Maria Giaretta, which may resonate with your experiences in clinical settings. Bioaccess® can offer assistance in participant recruitment to improve the project's success.
-
Conduct the Research: Implement the research in strict accordance with the approved protocol, maintaining adherence to Good Clinical Practice (GCP) guidelines. This involves ensuring that all team members are educated and that the research environment is favorable for gathering dependable data. Incorporate best practices from case studies, such as the randomization and blinding methods used in the ASD study, to enhance the credibility of your study design. Bioaccess® offers comprehensive clinical trial management services to facilitate this process.
-
Collect and Analyze Information: Systematically gather data on equipment performance, safety, and usability. Utilize suitable statistical techniques to evaluate the outcomes, offering insights into the practicality of the apparatus and guiding future development. Bioaccess® can assist in data analysis to ensure accurate interpretation of results.
-
Report Findings: Prepare a detailed report summarizing the study outcomes. This report should encompass an examination of the collected data, insights acquired, and suggestions for additional development or changes to the apparatus. Sharing these findings with stakeholders is crucial for advancing the device towards commercialization. Utilizing the knowledge of Bioaccess® can greatly improve the effectiveness and achievements of your clinical trials in Latin America, as demonstrated by their history of successful projects.
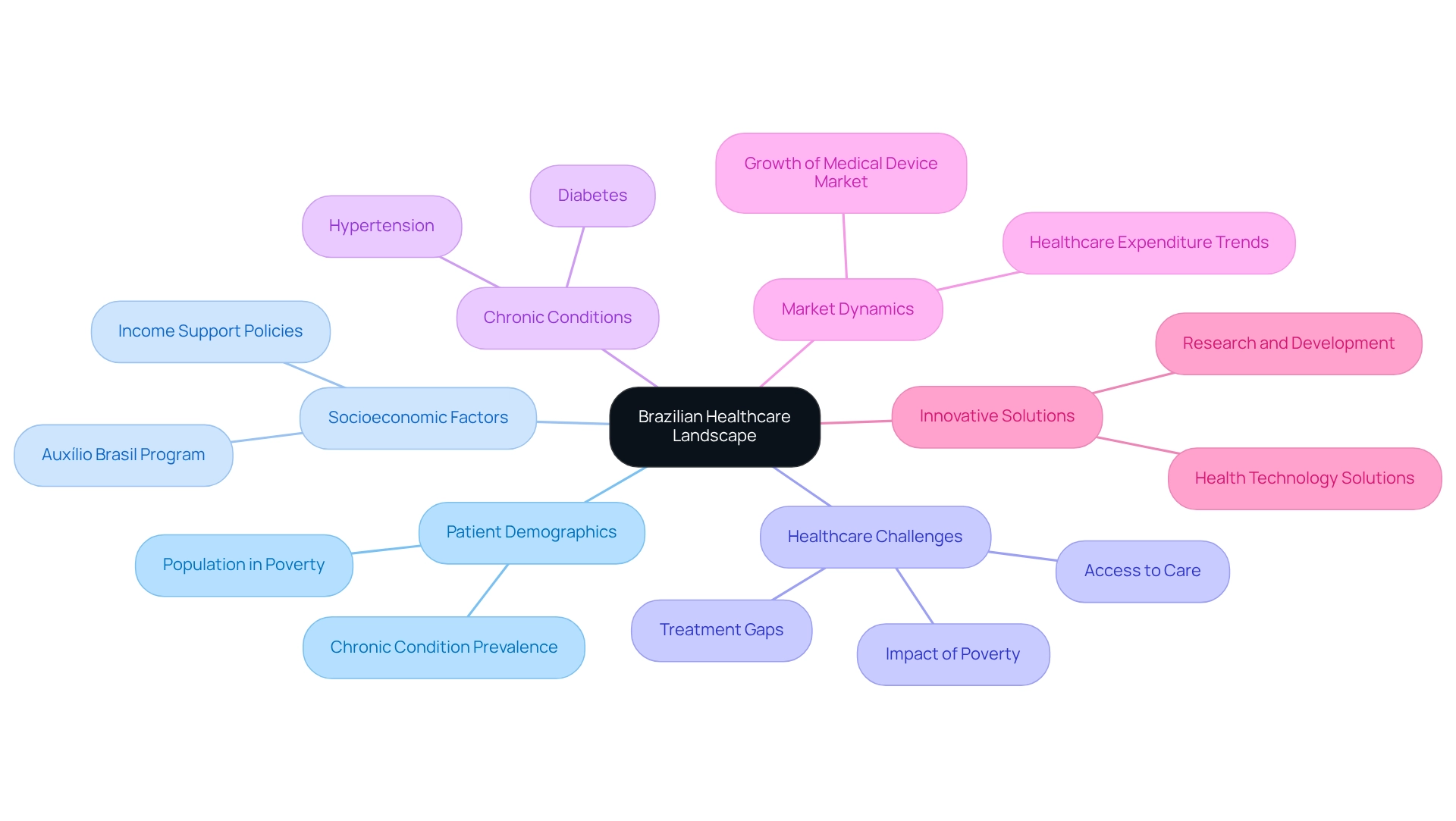
Assessing Market Dynamics and Patient Needs in Brazil
A comprehensive understanding of the local healthcare landscape and patient demographics is crucial for executing early feasibility studies in compliance with Brazilian regulations. Researchers must scrutinize critical factors such as disease prevalence, treatment gaps, and patient preferences to effectively inform their study design. For instance, Brazil's healthcare system faces significant challenges, with approximately 25% of the population living in poverty, which directly impacts access to healthcare and the utilization of medical equipment.
The Auxílio Brasil Program, designed to support families in poverty, plays an essential role in addressing these socioeconomic challenges, underscoring the need for innovative health technology solutions.
Engaging with healthcare professionals and potential users is vital for gaining insights into the practical challenges encountered by patients and clinicians. This interaction can reveal treatment gaps, particularly the high incidence of chronic conditions like hypertension and diabetes, which affect millions of Brazilians and create an urgent demand for innovative healthcare solutions. Recent statistics indicate that chronic illnesses account for a substantial portion of healthcare expenditure in Brazil, highlighting the importance of aligning healthcare equipment development with real-world needs.
Moreover, assessing the competitive landscape is critical for researchers to strategically position their products within the market. The Brazilian medical device market is anticipated to experience significant growth, fueled by increasing healthcare expenditures and a rising demand for advanced medical technologies. As noted by Otavio Berwanger, MD, PhD, the expertise available in Brazil, particularly in fields such as cardiology, enhances the quality of medical research and the understanding of patient needs.
By leveraging insights from case analyses and expert perspectives, researchers can navigate the complexities of the Brazilian healthcare system more effectively, ultimately improving the outcomes of early feasibility studies under Brazilian regulations. Furthermore, bioaccess® offers extensive trial management services—including feasibility assessments, site selection, compliance reviews, trial setup, import permits, project management, and reporting—enabling researchers to address these challenges efficiently. Public policy initiatives aimed at mitigating health disparities, such as the National Health Plan 2020–2023, provide a supportive framework for conducting clinical research in Brazil, further emphasizing the necessity of tackling healthcare challenges through innovative medical devices.
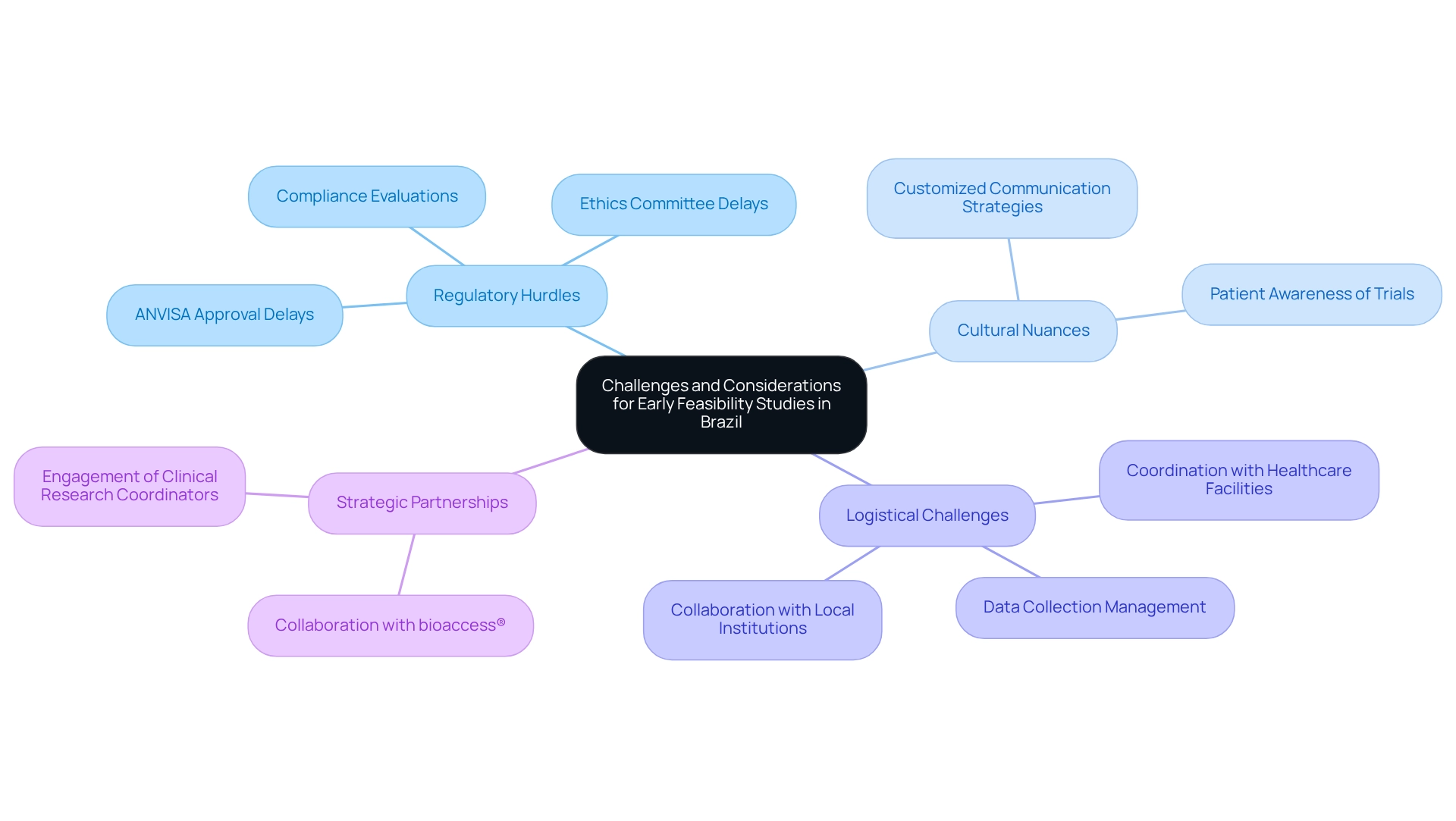
Challenges and Considerations for Early Feasibility Studies in Brazil
Early feasibility studies under Brazilian regulations require navigating a complex landscape characterized by regulatory hurdles, cultural nuances, and logistical challenges. A significant obstacle is the delay in obtaining approvals from ANVISA and ethics committees, which can greatly extend research timelines. Recent statistics indicate that the average wait for ANVISA approvals can stretch to several months, adversely affecting the overall efficiency of research trials.
In this context, partnering with bioaccess® offers a strategic advantage. With over 20 years of experience in Medtech, bioaccess® specializes in managing early feasibility studies under Brazilian regulations, as well as First-In-Human Trials, Pilot Trials, and Post-Market Clinical Follow-Up Assessments. Their expertise ensures compliance with local regulations while streamlining the approval process. Their extensive trial management services encompass:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Trial setup
- Import permits
- Project management
- Reporting
All tailored to address the specific challenges of the Latin American market.
Cultural factors significantly influence patient recruitment and retention. Researchers must recognize that communication strategies need to be customized to effectively engage diverse patient groups, ensuring that potential participants are well-informed about the benefits and procedures of research trials. This awareness is crucial, as research has shown that many patients in Latin America express a willingness to participate in trials when adequately informed.
Moreover, the socio-economic landscape, marked by income inequality, can impact patient engagement, evidenced by a strong correlation between income inequality and homicide rates in regions such as Rio de Janeiro.
Logistical challenges further complicate the study process, particularly when coordinating with multiple healthcare facilities and managing data collection across various sites. To effectively navigate these issues, researchers should prioritize establishing robust partnerships with local institutions and engaging experienced clinical research coordinators who are familiar with the regional landscape. Collaborating with bioaccess® can enhance these efforts, as their expertise in navigating the regulatory environment and their role as a specialist medical Importer of Record (IOR) can streamline the import process and ensure compliance with regulations, which is essential for overcoming regulatory hurdles.
Developing contingency plans to adapt to unexpected situations can bolster the robustness of the research design.
By implementing these strategies and leveraging the offerings of bioaccess®, researchers can adeptly navigate the regulatory environment and improve the feasibility of conducting early feasibility studies under Brazilian regulations. As Dr. Colin Scott, a senior pharmaceutical industry executive, observes, "For biotech and pharma companies seeking strategic, operational, and financial advantages, Brazil is no longer just an alternative... it’s a leading choice for successful early-phase trials." Ultimately, these efforts contribute to the advancement of medical technologies in the region, underscoring the importance of raising awareness of clinical trial opportunities to enhance participation rates.
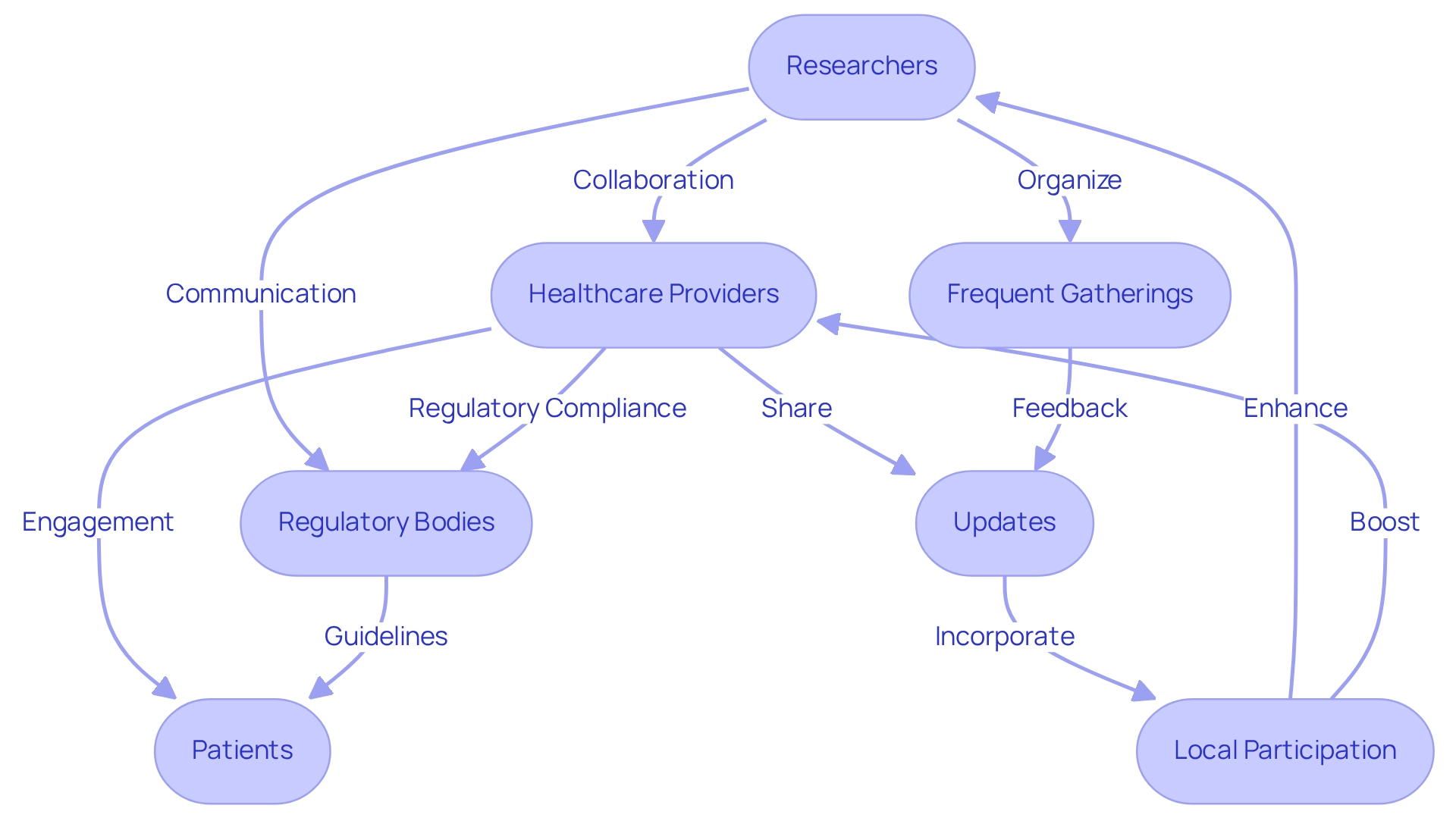
Collaboration and Communication: Key to Successful Feasibility Studies
Successful early feasibility studies under Brazilian regulations depend on strong cooperation among a diverse group of stakeholders, including researchers, healthcare providers, regulatory bodies, and patients. Clear communication pathways are essential to ensure that all parties align on research objectives and expectations. Frequent gatherings and updates serve as crucial tools for addressing issues and fostering problem-solving, ultimately enhancing the effectiveness of the research.
Involving local healthcare professionals in the research design and execution not only elevates the significance of the investigation but also boosts participant engagement. For instance, Dr. John B. Simpson, CEO of Avinger, highlighted the positive experience of conducting OCT-guided atherectomy research in Cali, Colombia, underscoring the importance of collaboration with LATAM CRO Experts and bioaccess. Similarly, ReGelTec's early feasibility studies under Brazilian regulations on HYDRAFIL™ for addressing chronic low back pain in Barranquilla, Colombia, illustrated how effective communication and local participation can lead to successful outcomes, with all eleven patients treated effectively.
A case study on communication as a success factor revealed that effective collaboration between sponsors and researchers significantly influences trial success. This emphasizes the necessity for regional coherence among sponsors and collaborators, which can result in improved outcomes. Furthermore, expert insights suggest that cultivating trust and maintaining open dialogue among stakeholders are vital components that contribute to the overall advancement of medical technology in Colombia.
Johnson noted that approximately 80% of medical trials fail to meet their original enrollment targets and schedules, resulting in substantial financial losses of $8 million each day for drug development firms. Consequently, prioritizing effective communication strategies is crucial for mitigating these risks and ensuring the success of research initiatives. Additionally, Global Care Clinical Trials partners with bioaccess to enhance clinical trial ambulatory services in Colombia, achieving over a 50% reduction in recruitment time and 95% retention rates.
Conclusion
Early feasibility studies (EFS) are increasingly vital in Brazil's medical device sector, delivering critical insights that foster innovation and enhance patient safety. By evaluating device viability in real-world scenarios, EFS empower Medtech companies to identify potential challenges early on, ensuring alignment with ANVISA's regulatory expectations and promoting continuous improvement in response to Brazil's diverse healthcare demands.
Navigating Brazil's regulatory landscape is essential for effective EFS implementation. A thorough understanding of device classification, securing ethical approvals, and adhering to Good Clinical Practice are crucial steps in streamlining the process. As approval timelines improve and regulations adapt, Brazil is emerging as a competitive hub for healthcare innovation, necessitating that researchers remain informed and compliant.
Additionally, a focus on patient demographics and the engagement of local healthcare professionals are pivotal for EFS success. Customizing strategies based on insights from the healthcare landscape enables researchers to effectively address significant needs. Collaborating with organizations like bioaccess® can further enhance the efficiency of these studies, ensuring compliance and favorable outcomes.
In conclusion, the growing recognition of EFS in Brazil marks a significant shift towards prioritizing safety, efficacy, and market readiness within the medical technology sphere. As the landscape evolves, collaboration among stakeholders and a commitment to effective communication will be essential for advancing medical innovations that fulfill the needs of both patients and healthcare providers.
Frequently Asked Questions
What are initial feasibility evaluations (EFS) in healthcare?
Initial feasibility evaluations (EFS) are preliminary assessments that determine the practicality of new healthcare instruments in real-world environments.
Why are early feasibility studies important in Brazil?
Early feasibility studies are essential in Brazil due to its diverse patient demographics and unique healthcare challenges, allowing researchers to gather vital information regarding equipment safety, functionality, and usability before proceeding to larger clinical trials.
How do early feasibility studies benefit Medtech firms in Brazil?
These studies help Medtech firms identify potential issues early, refine their products, and enhance their chances for successful market entry, while also aligning with the regulatory expectations of ANVISA.
What trends are emerging regarding early feasibility studies in Brazil?
There is a growing recognition of the importance of early feasibility studies within Brazil's healthcare industry, leading to an increase in applications for such studies and a commitment to patient safety and device efficacy.
What role does ANVISA play in Brazil's healthcare product regulations?
ANVISA, Brazil's health regulatory agency, oversees the approval and monitoring of clinical trials, ensuring compliance with established safety and quality standards.
What are the key steps to initiate a preliminary feasibility study in Brazil?
The key steps include device classification, protocol submission, ethical approvals, compliance with Good Clinical Practice (GCP), document translation into Portuguese, securing import permits, and continuous reporting on research status.
How has the approval process for trials in Brazil changed recently?
Approval times for trials in Brazil have improved, reflecting ANVISA's commitment to enhancing efficiency while ensuring safety and quality standards.
What is the significance of understanding ANVISA's regulatory criteria?
Understanding ANVISA's regulatory criteria and the processes for categorizing health products is crucial for researchers to facilitate smoother research execution and contribute to health technology advancement.
How can bioaccess® assist researchers in navigating these regulations?
Bioaccess® provides expertise in clinical trial administration services, including feasibility assessments, compliance evaluations, import permits, and project oversight, helping researchers tackle challenges and enhance their prospects for success.

