Overview
The article presents a detailed step-by-step guide on designing medtech clinical trials, underscoring the critical need to align study goals with regulatory compliance and effective recruitment strategies. It elaborates on the essential phases of clinical trials—Pilot, Pivotal, and Post-Market—while showcasing best practices, technological integration, and real-world examples that exemplify successful trial design and execution within the dynamic Medtech landscape.
Introduction
In the rapidly evolving realm of medical technology, the design of clinical trials stands as a critical factor in the successful development and commercialization of innovative medical devices. Understanding the foundational elements of trial design—including study objectives, endpoints, and regulatory compliance—profoundly impacts the outcomes of these investigations.
As the industry embraces technological advancements and prioritizes diverse patient populations, integrating strategies such as real-time data management and innovative recruitment methods becomes essential.
This article delves into the intricacies of Medtech clinical trial design, exploring:
- Best practices
- Stages of trials
- The pivotal role of regulatory adherence in ensuring the safety and efficacy of new medical devices.
Understanding the Basics of Medtech Clinical Trial Design
Understanding how to design medtech clinical trials begins with a clear grasp of essential concepts, particularly study goals and endpoints, which are crucial for the success of any medical investigation. The primary objective must closely align with the intended use of the medical device, ensuring that the focus remains relevant and impactful. Identifying specific endpoints is equally crucial; these measurable outcomes will assess the device's safety and efficacy.
Frequently utilized endpoints include:
- Health outcomes
- Quality of life assessments
- Device performance metrics
All of which provide valuable insights into the device's real-world application.
Familiarity with various types of clinical experiments, such as randomized controlled trials (RCTs) and observational research, is essential, as each design presents unique advantages and challenges. A well-structured protocol serves as a comprehensive roadmap for understanding how to design medtech clinical trials, outlining the study's design, methodology, and statistical analysis plan. This careful planning is particularly vital in the evolving landscape of 2025, where advancements in artificial intelligence are transforming information creation and enhancing efficiency in experiments.
As Danish Mairaj, principal engineer of medical device design at RESYCA, notes, "This is one of the areas where AI can utilize a lot of information, but gaining insights from this information is only possible if you have certain use cases being implemented in relevant facilities."
Recent statistics reveal that 45% of data from leading companies like Alcon is entered on the same day as the visit date, underscoring the significance of real-time data management in enhancing outcomes. Furthermore, the research landscape increasingly concentrates on fair access, particularly in Latin America, which presents both challenges and opportunities for Medtech firms. Addressing these challenges is crucial for ensuring that innovative medical devices reach underserved populations, thereby maximizing their potential impact.
Successful examples of study objectives and endpoints in Medtech studies illustrate the potential for innovation. For instance, bioaccess® has demonstrated proficiency in overseeing Early-Feasibility Studies, First-In-Human Studies, and Post-Market Clinical Follow-Up Studies, which are essential for navigating the complexities of clinical research in Latin America. Additionally, the case of ReGelTec's Early Feasibility Study on HYDRAFIL™ for addressing chronic low back pain in Colombia exemplifies how effective study design can lead to successful patient outcomes.
Companies are now prioritizing the simplification of site experiences, as evidenced by case studies where addressing site pain points—such as query response times—has resulted in enhanced patient care and more efficient studies. As the Medtech sector continues to progress, learning how to design medtech clinical trials by integrating these best practices and strategies into research design will be crucial for advancing medical devices and ensuring their successful commercialization.

Navigating the Stages of Medtech Clinical Trials: From Pilot to Post-Market
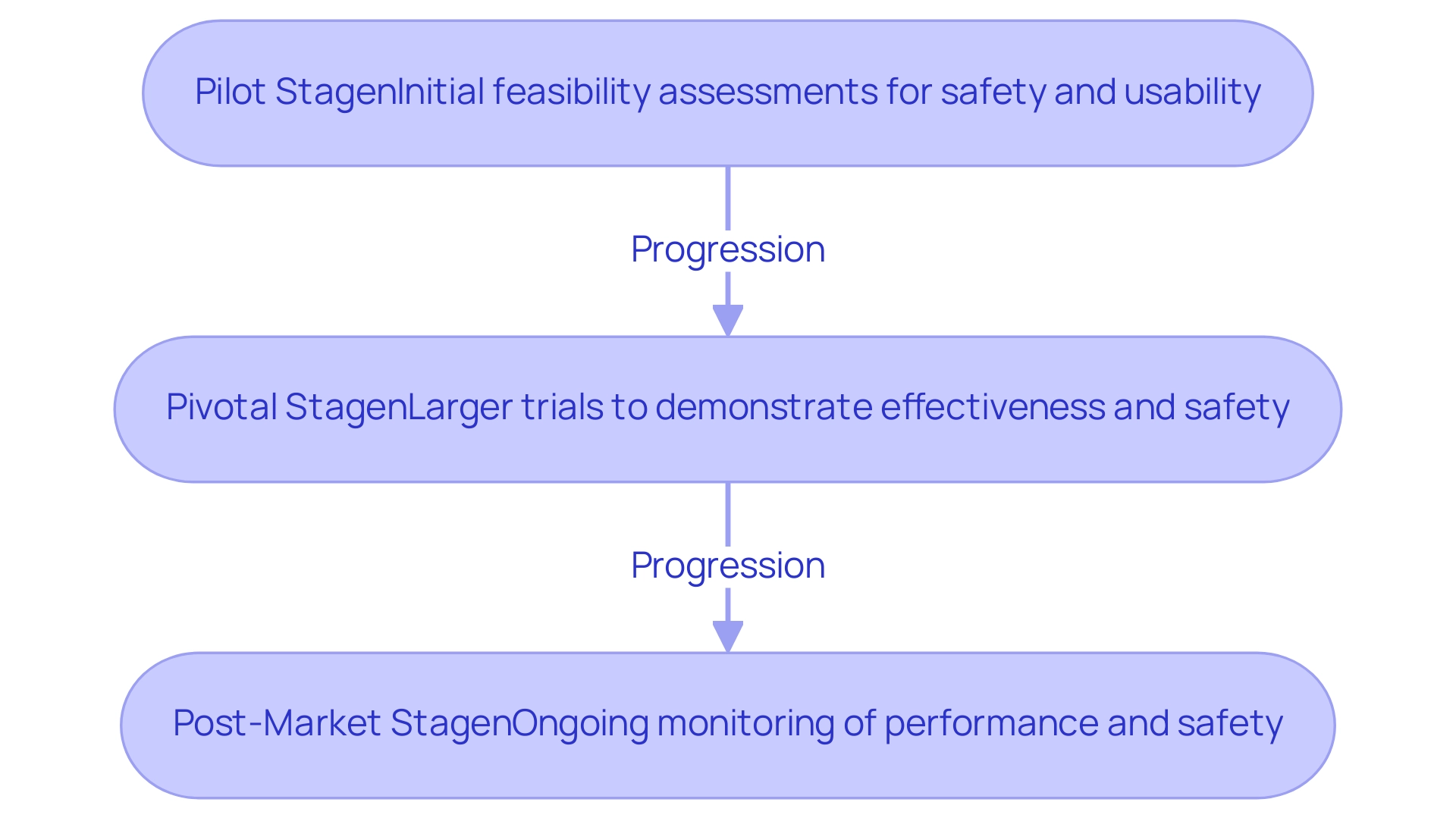
Medtech clinical studies are organized into three crucial stages: Pilot, Pivotal, and Post-Market, each fulfilling a unique role in the development and assessment of medical devices.
The Pilot stage, frequently known as initial feasibility assessments, is essential for evaluating the safety and usability of a device within a restricted participant group. A notable example is ReGelTec's Early Feasibility Study on HYDRAFIL™, which successfully treated eleven patients with chronic low back pain in Colombia. This initial phase enables researchers to identify potential issues and refine the device before progressing to larger studies.
Success rates for pilot projects in Medtech have shown promising trends, with many reports indicating favorable outcomes that pave the way for subsequent phases. bioaccess® specializes in managing Early-Feasibility Studies, ensuring that assessments are conducted efficiently and effectively.
Following the Pilot phase, the Pivotal phase includes larger, more rigorous trials aimed at demonstrating the device's effectiveness and safety across a broader population. This stage is essential for obtaining regulatory approval, as it provides the necessary evidence to support claims of the device's benefits. Interventional trials are anticipated to dominate the clinical trial market, fueled by the rising demand for enhanced diagnostic tests and vaccines, emphasizing the significance of strong Pivotal research in the current landscape. bioaccess® provides extensive assistance in Pivotal trials, increasing the chances of favorable results.
The final stage, Post-Market, involves ongoing monitoring of the device's performance and safety after it has received regulatory approval. This phase is vital for collecting long-term data, ensuring compliance with regulatory standards, and addressing any emerging safety concerns. As the Medtech sector develops, the importance of Post-Market evaluations keeps increasing, especially considering recent obstacles in participant recruitment and retention rates, which have led to a move towards more engaging and personalized methods.
A case study on challenges in medical research recruitment illustrates the significant issues faced by the biopharma industry, emphasizing the need for innovative recruitment strategies to improve delivery times and retention rates. bioaccess® has shown success in attaining over 50% decrease in recruitment duration and 95% retention rates in their studies.
In 2025, the terrain of Medtech research is defined by an emphasis on enhancing development pathways to not only reach approval-enabling endpoints but also to guarantee commercial success. As Max Baumann from Treehill Partners notes, "We expect continued focus on optimizing the development journeys of assets to achieve not only an approval-enabling endpoint but to qualify for commercial success." The sector encounters essential business model challenges as end-markets become progressively crowded.
Therefore, understanding the intricacies of each phase is paramount for Medtech companies looking to grasp how to design medtech clinical trials successfully. Additionally, the last major transformation in Japan's regulatory environment in 2017 serves as a relevant backdrop to the current changes being implemented, underscoring the dynamic nature of the Medtech landscape.
Ensuring Regulatory Compliance in Medtech Clinical Trials
Understanding how to design medtech clinical trials is essential, as regulatory compliance acts as a fundamental pillar in the design and execution of Medtech research studies. Following the guidelines set by regulatory bodies like the FDA and EMA is crucial for maintaining the integrity and safety of research. Familiarity with pertinent regulations, particularly the Good Clinical Practice (GCP) guidelines, is vital.
These guidelines provide insight into how to design medtech clinical trials while ensuring ethical and scientific quality standards are met for their conduct and reporting.
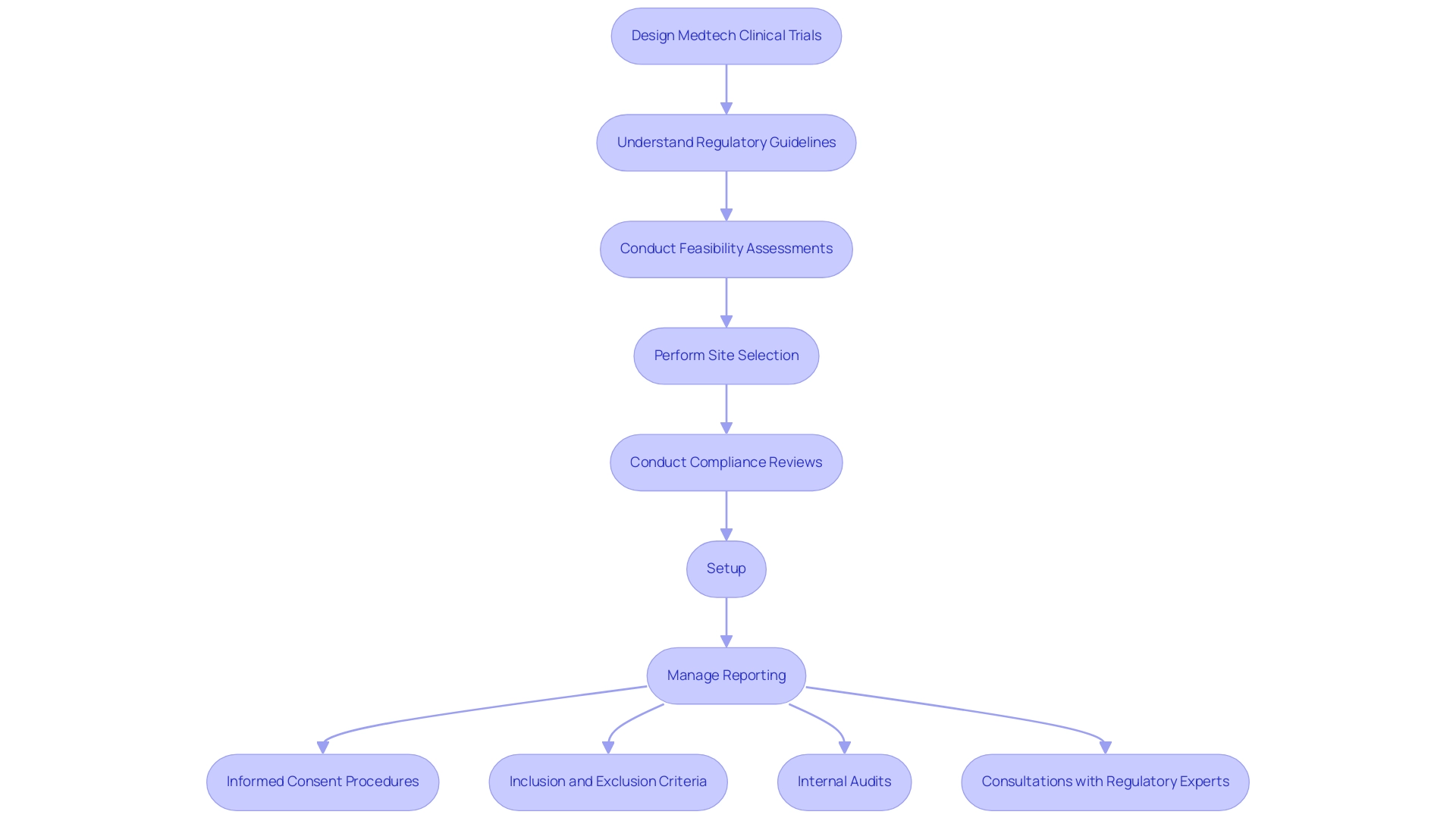
At bioaccess, our comprehensive clinical research management services encompass:
- Feasibility assessments
- Site selection
- Compliance reviews
- Setup
- Import permits
- Nationalization of investigational devices
- Project management
- Reporting (research status, inventory, serious and non-serious adverse events)
A strong research protocol must encompass all critical components, including informed consent procedures, clear inclusion and exclusion criteria, and thorough management plans. As the landscape of regulatory compliance evolves, particularly with the anticipated emphasis on sustainability in 2025—where 77% of US respondents expect increased regulatory scrutiny—staying informed is paramount.
Regular consultations with regulatory experts, such as Katherine Ruiz, who specializes in Regulatory Affairs for medical devices and in vitro diagnostics in Colombia, can provide insights into any changes that may affect your trial.
Moreover, internal audits serve as a proactive measure to evaluate compliance and pinpoint areas for enhancement. The recent resurgence of Medical Device Regulation (MDR) solutions emphasizes the need for enhanced research design, including how to design medtech clinical trials and improve information management practices. Effective metadata management and the integration of electronic data capture (EDC) systems are becoming increasingly essential for automating study builds and improving data management efficiency.
Adhering to FDA and EMA guidelines not only enhances the credibility of your study but also protects the rights and safety of participants. In 2025, as the industry adapts to new regulatory frameworks, the importance of Good Clinical Practice guidelines cannot be overstated. As noted by a Senior Vice President of a small German pharma company, "We are full of expectations for personalized medicine. With advances in genomics and biomarkers, we can provide patients with more precise treatment options. This not only improves efficacy but also significantly reduces side effects, allowing patients to have a better treatment experience." By prioritizing regulatory compliance, Medtech firms can manage the intricacies of studies more effectively, ultimately resulting in improved patient outcomes and expedited routes to market.
Additionally, the NHS Health Research Authority’s framework offers a structured method for integrating patient input while upholding scientific standards, which is crucial for maintaining regulatory compliance. By incorporating these elements, Medtech companies can enhance their study designs and learn how to design medtech clinical trials that meet the evolving demands of regulatory authorities.
Strategies for Successful Recruitment in Clinical Trials
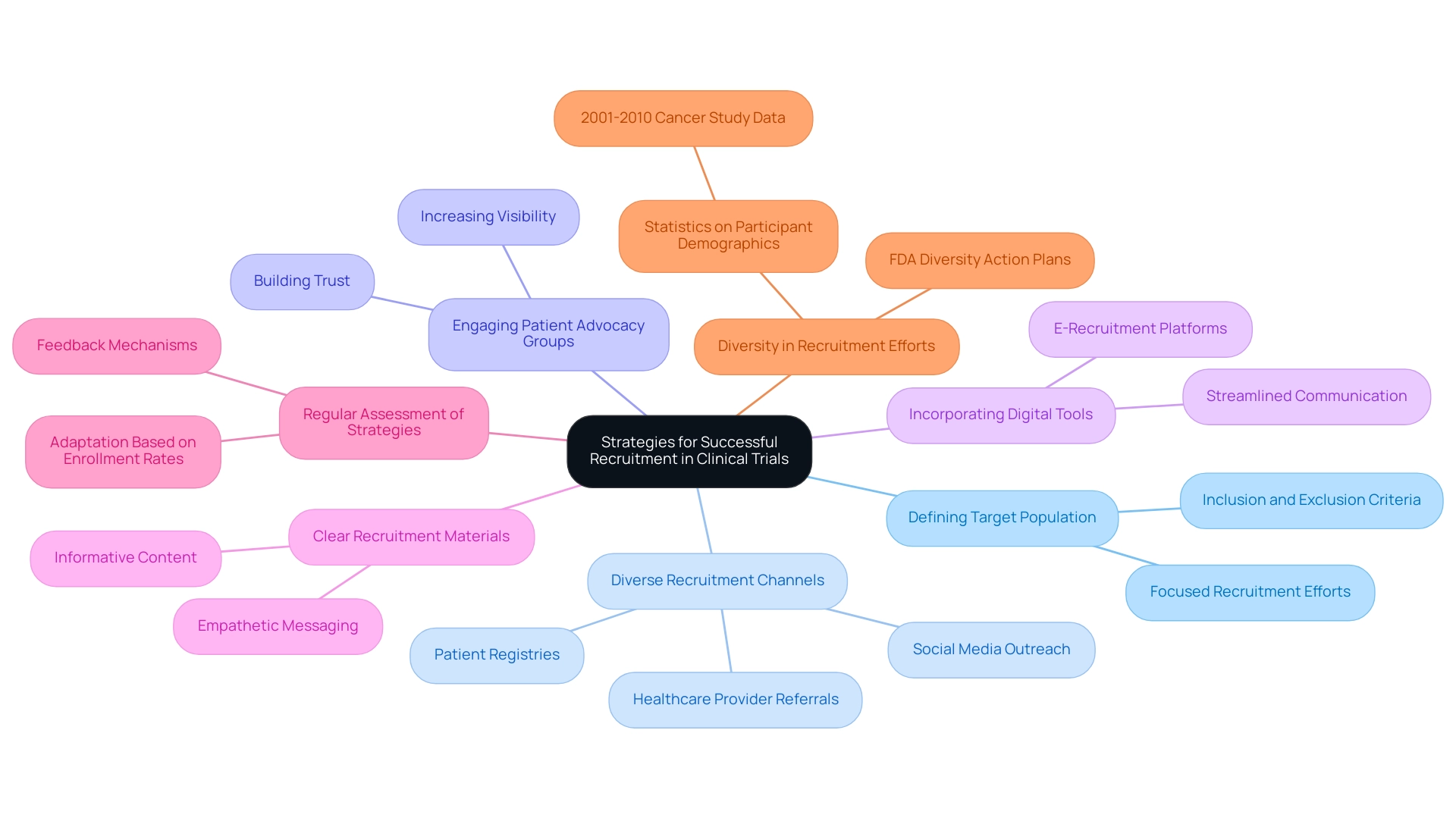
Recruiting participants for clinical studies presents significant challenges. However, employing effective strategies can streamline this process considerably. Begin by precisely defining your target population in accordance with the study's inclusion and exclusion criteria. This foundational step ensures that recruitment efforts are focused and relevant.
Utilizing a diverse array of recruitment channels is crucial. Social media platforms have emerged as powerful tools for reaching potential participants, allowing for targeted outreach and engagement. Additionally, leveraging healthcare provider referrals and patient registries can broaden your reach and enhance recruitment efforts.
Engaging with patient advocacy groups not only increases visibility but also fosters trust within the community, which is essential for participant buy-in.
Incorporating digital tools for e-recruitment can further simplify the process and improve participant engagement. These tools facilitate communication and streamline the onboarding process, making it easier for potential participants to understand the study's purpose and their role within it.
Your recruitment materials should be clear, informative, and empathetic, addressing common concerns and questions that potential participants may have. Regularly assessing your recruitment strategies is vital; be prepared to adapt based on feedback and enrollment rates. For instance, recent findings suggest that 73% of patients favor discovering research opportunities through their healthcare providers, highlighting the significance of efficient communication channels.
Moreover, the FDA's recent draft guidance on Diversity Action Plans emphasizes the need for increased enrollment of underrepresented populations in clinical studies. Statistics from cancer treatment studies from 2001 to 2010 reveal that 82.9% of participants were white, underscoring the critical need for diversity in recruitment efforts. By offering more options for patient onboarding and visit experiences, sponsors can enhance participant satisfaction and broaden the diversity of study populations, ultimately leading to more robust and generalizable results.
A real-world example of effective recruitment strategies can be seen in MAC Clinical Research, which successfully addressed recruitment challenges in a recent study by tripling the recruitment rate after being initiated as a rescue site. This demonstrates the potential for innovative approaches to significantly improve participant recruitment.
Furthermore, the partnership between bioaccess™ and Caribbean Health Group (CHG) seeks to establish Barranquilla as a prominent location for medical studies in Latin America, backed by Colombia's Minister of Health. This initiative enhances the infrastructure for clinical research and contributes to local economic growth through job creation and improved healthcare services. The partnership has led to a significant reduction in recruitment time and high retention rates, showcasing the effectiveness of their combined efforts.
As Bree Burks, Vice President of Site Strategy at Veeva, observes, "As sponsors reconsider their site engagement approaches in 2025, they will emphasize consistent site technology and standardization across sponsors for all studies." By prioritizing participant recruitment and employing these strategies, you can significantly enhance the likelihood of success in designing medtech clinical trials and ensure timely completion, thereby paving the way for advancements in medical technology.
Leveraging Technology and Innovation in Clinical Trial Design
Integrating technology is essential for designing medtech clinical trials effectively, enhancing efficiency and ensuring high-quality information. Begin by leveraging electronic information capture (EDC) systems, which facilitate streamlined collection and management, significantly reducing potential errors and enhancing overall integrity. The adoption of remote monitoring technologies, such as wearable devices, allows for the gathering of real-time information from participants, fostering greater engagement and adherence throughout the study process.
As we progress through 2025, the impact of EDC systems is becoming increasingly evident, with research indicating that their use can lead to quicker data accessibility and improved patient outcomes. Current trends show a growing focus on decentralized research models, which are crucial for understanding how to design medtech clinical trials that provide participants with enhanced flexibility and accessibility, ultimately broadening the demographic reach of medical investigations. To further enhance this reach, it is vital to implement more options for patient onboarding and study visits, significantly improving the enrollment of diverse populations in research studies.
bioaccess offers extensive study management services that encompass feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting on study status, inventory, and adverse events. These services are essential for ensuring that research studies are conducted effectively and in accordance with local regulations, positively influencing local economies through job creation and healthcare enhancement. Additionally, artificial intelligence (AI) plays a pivotal role in optimizing study design, which is critical for understanding how to design medtech clinical trials, enhancing patient recruitment strategies, and streamlining data analysis.
By utilizing these advanced technologies, researchers can learn how to design medtech clinical trials that not only improve the quality of their studies but also reduce expenses and accelerate the timeline from study initiation to market entry. This strategic approach aligns with the industry's shift towards achieving both approval-enabling endpoints and commercial viability. As Max Baumann, Head of Execution, states, "We expect continued focus on optimizing the development journeys of assets to achieve not only an approval-enabling endpoint but to qualify for commercial success."
Furthermore, the FDA's draft guidance on Diversity Action Plans urges sponsors to enroll more patients from underrepresented groups, underscoring the importance of diversity in clinical trials. Additionally, the evolving landscape of electronic patient-reported outcomes (ePRO) in 2025 highlights the necessity for applications that support patient engagement and retention beyond mere data capture.

Conclusion
The design of Medtech clinical trials is a multifaceted endeavor that necessitates a comprehensive understanding of foundational concepts, including study objectives, endpoints, and regulatory compliance. By aligning trial objectives with the intended use of medical devices, researchers can ensure that their investigations yield relevant and impactful results. The integration of real-time data management and innovative recruitment strategies is crucial for enhancing trial efficiency and reaching diverse patient populations, ultimately improving the quality of outcomes.
Navigating the stages of clinical trials—from Pilot to Pivotal and Post-Market—underscores the significance of each phase in evaluating the safety and efficacy of medical devices. Successful trials not only adhere to regulatory guidelines but also incorporate effective recruitment strategies that engage participants and ensure diverse representation. The role of technology, particularly electronic data capture and remote monitoring, emphasizes the necessity of leveraging advancements to streamline processes and enhance data quality.
As the Medtech landscape continues to evolve, prioritizing regulatory compliance remains essential for safeguarding participant rights and ensuring the integrity of clinical research. By focusing on these critical elements, Medtech companies can navigate the complexities of clinical trials more effectively, leading to improved patient outcomes and accelerated pathways to market. Embracing these best practices will not only facilitate the successful commercialization of innovative medical devices but also contribute to the advancement of healthcare as a whole. The future of Medtech is promising, driven by a commitment to innovation, inclusivity, and regulatory adherence.
Frequently Asked Questions
What are the key components to consider when designing medtech clinical trials?
The key components include understanding study goals and endpoints, which should align with the intended use of the medical device. Specific measurable outcomes must be identified to assess the device's safety and efficacy.
What types of endpoints are commonly used in medtech clinical trials?
Commonly utilized endpoints include health outcomes, quality of life assessments, and device performance metrics, all of which provide insights into the device's real-world application.
Why is familiarity with different types of clinical experiments important in medtech?
Familiarity with various clinical experiment types, such as randomized controlled trials (RCTs) and observational research, is essential as each design has unique advantages and challenges that can impact the study's effectiveness.
What role does a well-structured protocol play in medtech clinical trials?
A well-structured protocol serves as a comprehensive roadmap, outlining the study's design, methodology, and statistical analysis plan, which is crucial for successful trial execution.
How is artificial intelligence influencing medtech clinical trials?
Advancements in artificial intelligence are transforming information creation and enhancing efficiency in experiments, allowing for better insights and management of data in clinical trials.
What are the three crucial stages of medtech clinical studies?
The three stages are Pilot, Pivotal, and Post-Market. Each stage has a unique role in the development and assessment of medical devices.
What is the purpose of the Pilot stage in clinical trials?
The Pilot stage, or initial feasibility assessments, is essential for evaluating the safety and usability of a device within a restricted participant group and allows researchers to identify potential issues before larger studies.
What is the significance of the Pivotal phase in clinical trials?
The Pivotal phase involves larger, more rigorous trials aimed at demonstrating the device's effectiveness and safety across a broader population, which is critical for obtaining regulatory approval.
What does the Post-Market stage entail in medtech studies?
The Post-Market stage involves ongoing monitoring of the device's performance and safety after regulatory approval, collecting long-term data, and ensuring compliance with regulatory standards.
How are recruitment and retention challenges being addressed in medtech research?
Innovative recruitment strategies are being employed to improve delivery times and retention rates, with some companies reporting significant decreases in recruitment duration and high retention rates in their studies.
What trends are shaping the future of medtech clinical trials in 2025?
The future of medtech research is focused on enhancing development pathways to achieve approval-enabling endpoints and ensure commercial success, amidst a crowded market and evolving regulatory landscape.

