Overview
The future of medtech research in Latin America is marked by significant economic growth, technological advancements, and a supportive regulatory environment, positioning the region as a vital player in global medical technology innovation. This article underscores that countries such as Brazil, Mexico, and Colombia are at the forefront of this development, bolstered by increased healthcare investments, diverse patient populations, and strategic partnerships that enhance clinical research capabilities and streamline regulatory processes. As these nations continue to lead the charge, the implications for clinical research are profound, suggesting a landscape ripe for innovation and collaboration.
Introduction
Latin America is emerging as a vibrant hub for Medtech research, driven by a unique blend of demographic diversity, increasing healthcare investments, and innovative collaborations. The Medtech market, surpassing 44 billion USD in 2023, is poised for significant growth, with countries like Brazil, Mexico, and Argentina leading the charge. This dynamic landscape not only attracts international attention but also transforms clinical research methodologies, particularly through the integration of advanced technologies and strategic partnerships. As the region navigates the complexities of regulatory challenges and capitalizes on its rich patient demographics, the potential for groundbreaking medical innovations is vast. This article delves into the current state of Medtech research in Latin America, exploring key drivers of growth, emerging technologies, and the critical role of clinical trials in shaping the future of healthcare.
Current Landscape of Medtech Research in Latin America
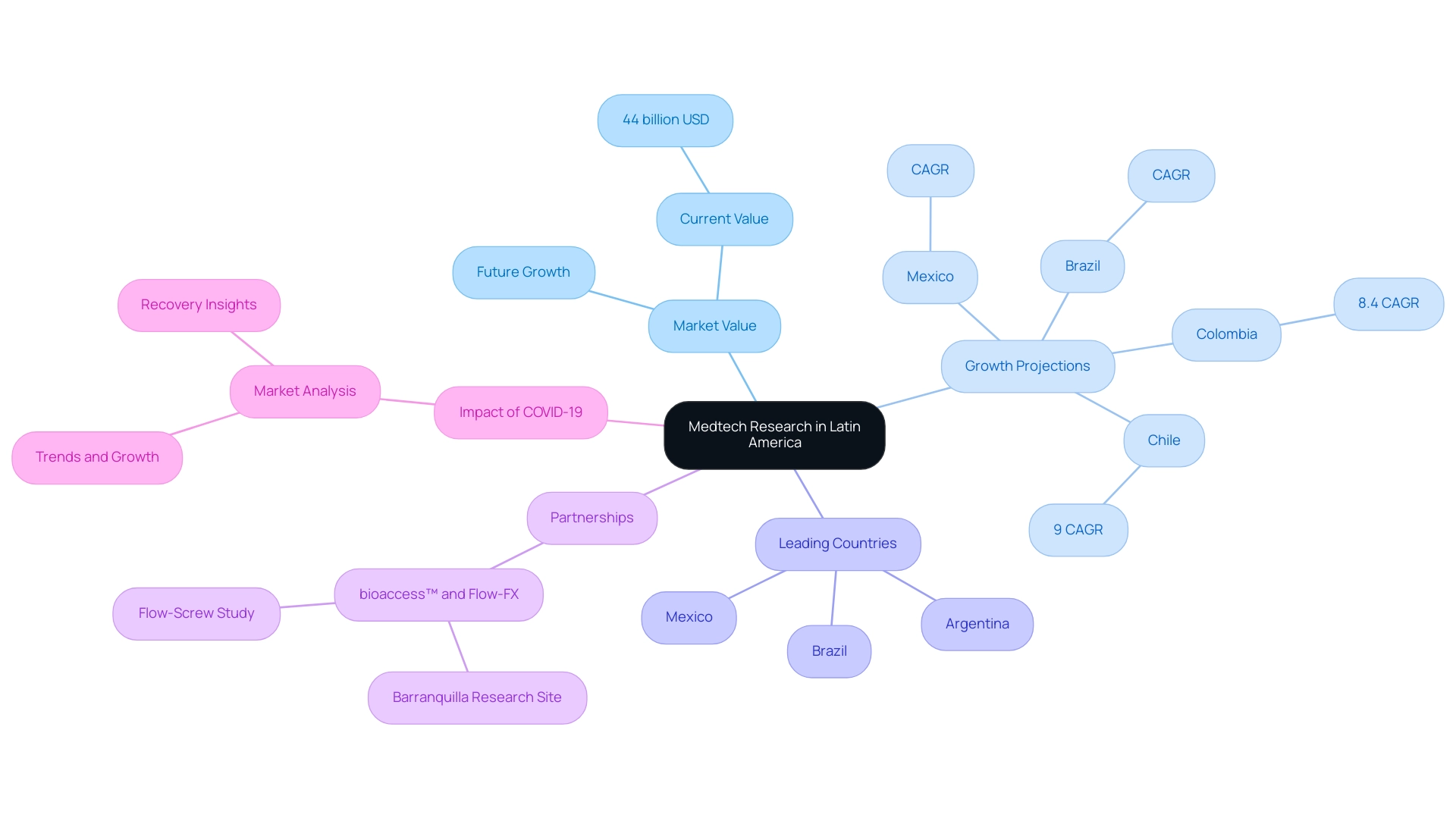
Latin America is swiftly establishing itself as a pivotal player in the global medical technology research landscape, embodying the future of medtech research in the region, driven by a diverse patient base and escalating healthcare investments. As of 2023, the medical technology market in this area was valued at over 44 billion USD, with projections indicating robust growth in the years ahead. Countries such as Brazil, Mexico, and Argentina are poised to lead this expansion, benefiting from favorable regulatory environments and a strong commitment to innovation.
The trajectory of growth is particularly promising, with the Hospitals and Ambulatory Surgical Centers (ASCs) segment expected to experience significant compound annual growth rates (CAGR) throughout the forecast period. In Chile and Colombia, for example, the Medtech market is anticipated to grow at CAGRs of 9% and 8.4%, respectively, propelled by a high incidence of chronic diseases and enhancements in health systems supported by government initiatives.
A notable instance of this expansion is the partnership between bioaccess™ and Flow-FX, which has positioned Barranquilla as a leading site for research studies in Latin America. Their first-in-human research study on the Flow-Screw medical device for intraosseous antibiotic delivery exemplifies the region's capabilities. Conducted at Clinical La Misericordia and led by Dr. Carlos Severini, this study showcases the potential for innovative medical devices to flourish in this environment.
This collaboration has not only advanced clinical research but also demonstrated the effectiveness of bioaccess in managing complex studies. Dushyanth Surakanti, Founder & CEO of Sparta Biomedical, shared his experience with bioaccess during its first human study in Colombia, stating, "The expertise and support provided by bioaccess were instrumental in navigating the complexities of the process, ensuring its success and efficiency."
The impact of the COVID-19 pandemic has also transformed the Medtech landscape. A comprehensive analysis of the South American medical equipment market reveals that the pandemic has significantly influenced market value, trends, and growth. This study offers stakeholders insights to navigate recovery and future developments, underscoring the necessity for adaptability in the sector.
Marlene Greenfield, Vice President, noted, "Statista has been my savior on several occasions. The site is easy to maneuver and the data is in a format that can go right into a report or presentation." This highlights the significance of accessible data in comprehending market dynamics.
With its unique demographic diversity, Latin America provides a rich backdrop for the future of medtech research and various research projects and investigations. Successful trials in Brazil, Mexico, and Argentina underscore the region's potential, illustrating the future of medtech research in Latin America and its capacity to support innovative medical devices and technologies. As the medical technology sector evolves, bioaccess®'s commitment to advancing medical devices sooner through expertise and a tailored approach will be vital in meeting the demands of the global market.
Their extensive experience in managing research studies, including Early-Feasibility, First-In-Human, and Post-Market Follow-Up Studies, positions them as a leader in the field, driving economic growth and healthcare improvement in the region.
Key Drivers Fueling Medtech Growth in Latin America
Several pivotal drivers are propelling the growth of Medtech in Latin America, including:
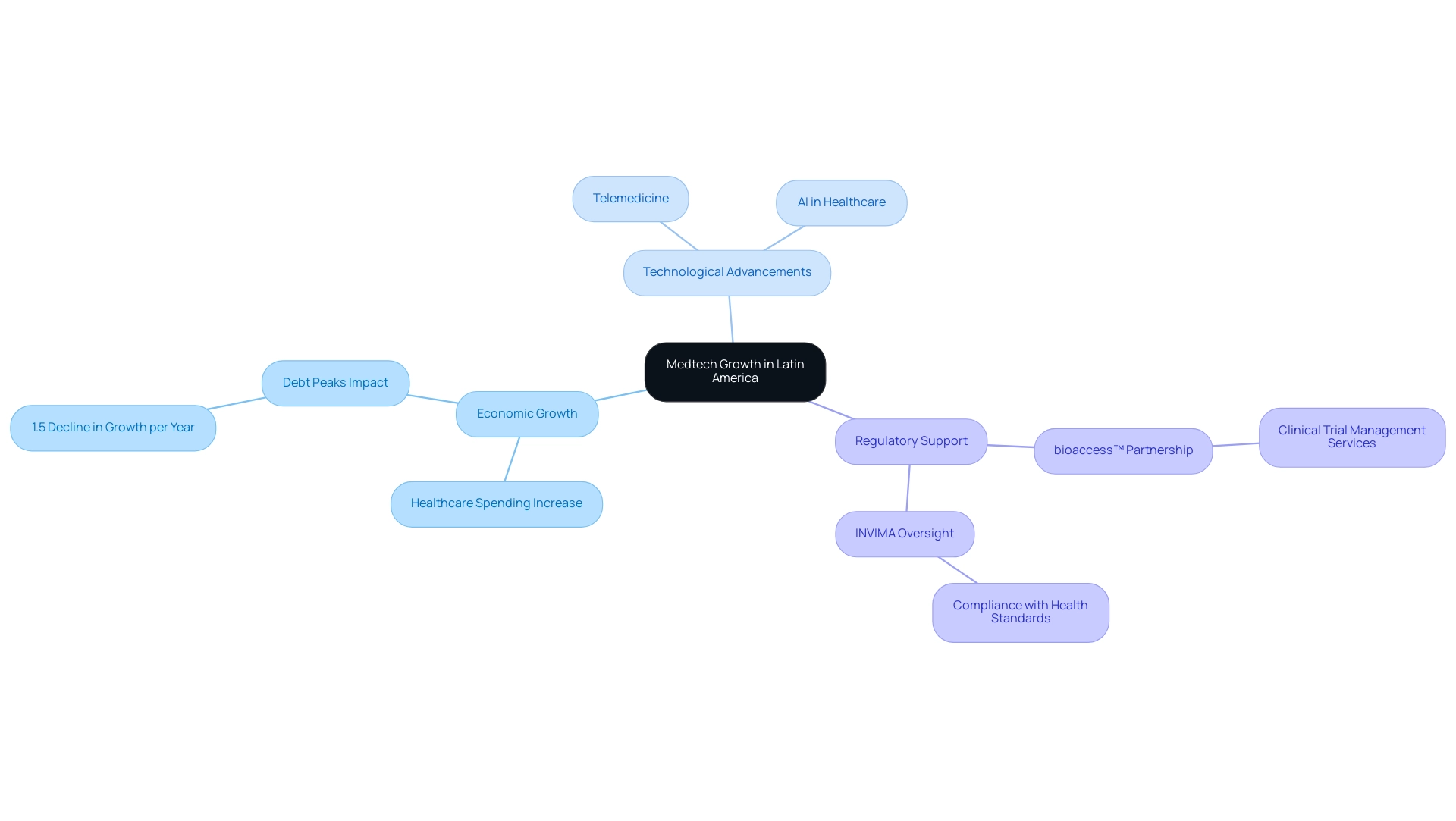
- Economic Growth: The region is witnessing a significant increase in healthcare spending, bolstered by investments in infrastructure that create a favorable environment for Medtech innovations. According to recent analysis, economic growth is projected to enhance health expenditures, particularly as aging populations and non-communicable diseases (NCDs) become more prevalent. Specifically, studies from the Inter-American Development Bank (IDB) indicate that 'debt peaks' can have a considerable negative impact, with each year following a debt peak resulting in an additional decline in economic growth by 1.5 percentage points. This trend underscores the necessity for efficient health resource management to accommodate rising costs.
- Technological Advancements: The emergence of digital health technologies, such as telemedicine and artificial intelligence, is revolutionizing healthcare delivery and research methodologies. These advancements not only improve patient outcomes but also streamline clinical trials, making them more efficient and accessible. The integration of these technologies is expected to drive further innovation within the medical technology sector.
- Regulatory Support: A supportive regulatory landscape is crucial for fostering innovation in Medtech. The partnership between bioaccess™ and Caribbean Health Group (CHG) seeks to establish Barranquilla as a premier location for medical studies in Latin America, supported by Colombia's Minister of Health. This initiative is anticipated to improve outpatient services in Colombia, achieving over 50% reduction in recruitment time and 95% retention rates. bioaccess™ offers comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting. Additionally, INVIMA, the Colombia National Food and Drug Surveillance Institute, plays a vital role in overseeing medical devices and ensuring compliance with health standards, classified as a Level 4 health authority by PAHO/WHO. Streamlined processes and government initiatives are increasingly attracting both local and international companies to the region. Workshops like the 'Latin America is Open for Business' at the Florida International Medical Expo 2023 exemplify this trend, offering opportunities for knowledge sharing and networking that can lead to substantial income increases for participating firms. In fact, the workshop guarantees an income increase of at least 50 percent for the first year for clients using the Buyer Representative service.
These factors collectively emphasize the dynamic environment of medical technology in South America, highlighting that the future of medtech research in Latin America will be shaped by economic growth and technological advancement. Furthermore, the case study titled "Impact of Demographic Changes on Health Expenditures" illustrates how demographic transitions, particularly population aging and the rise of NCDs, affect health expenditures in LAC countries, emphasizing the need for efficient health resource management.
Navigating the Regulatory Landscape for Medtech in Latin America
The regulatory landscape presents a multifaceted challenge for the future of medtech research in Latin America, primarily due to the lack of harmonization among countries. Each nation operates under its own regulatory authority, leading to a diverse array of requirements that companies must navigate. This complexity necessitates thorough research and often the engagement of local experts to ensure compliance and successful execution of studies.
In 2025, significant strides are being made towards streamlining regulatory processes, particularly in Brazil and Chile. These nations are actively improving their regulatory structures to attract more research studies, reflecting a growing acknowledgment of the significance of efficient regulatory pathways for the future of medtech research in Latin America. For instance, Brazil has introduced measures intended to shorten approval times for research studies, while Chile is concentrating on harmonizing its regulations with global standards.
Grasping these regulatory subtleties is crucial for medical technology firms aiming to contribute to the future of medtech research in Latin America. The capacity to navigate these challenges effectively can yield successful outcomes, as demonstrated by bioaccess®'s experience in connecting innovative medical technology companies with the future of medtech research in Latin America. With over 15 years of expertise, bioaccess® has successfully supported numerous startups in overcoming regulatory hurdles, thereby accelerating the advancement of medical devices from initial studies to commercialization.
As the regulatory environment continues to evolve, the future of medtech research in Latin America necessitates that medical technology companies prioritize compliance to avoid severe penalties from increasingly coordinated regulatory bodies. Gartner forecasts that by 2025, a third of all nations will enact laws specifically concerning ransomware payments, emphasizing the critical need for regulatory adherence and security in medical technology. Furthermore, starting in 2025, connected medical devices must be secure against cyber threats, with technology firms accountable for protecting patient data.
This proactive approach will not only facilitate smoother processes but also enhance the overall integrity and safety of clinical research in the region, which is crucial for the future of medtech research in Latin America.
bioaccess® offers comprehensive services, including feasibility studies, site selection, compliance reviews, setup, project management, and reporting. The company specializes in various types of studies such as Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF). This extensive expertise positions bioaccess® as a leader in navigating the complexities of the future of medtech research in Latin America.
The Role of Clinical Trials in Advancing Medtech Innovations
Clinical trials are the cornerstone of Medtech innovation, providing essential insights into the safety and efficacy of emerging devices. In 2025, America emerges as a prime location for Medtech research in Latin America, presenting numerous compelling advantages. Notably, the region exhibits significantly faster patient recruitment rates, with dropout rates one-third of those seen in the U.S. and EU, thereby enhancing the credibility of research outcomes.
This efficiency is further enhanced by a diverse patient demographic, which enriches the data collected and improves the generalizability of study results.
A prime illustration of this trend is Flow-FX's selection of Colombia and bioaccess™ as its contract research organization (CRO) for a first-in-human study on its innovative Flow-Screw medical device for the delivery of intraosseous (IO) antibiotics. Conducted at Clinica La Misericordia in Barranquilla, Colombia, under the guidance of Dr. Carlos Severini, this study exemplifies collaborative efforts to position Barranquilla as a leading destination for medical studies in South America, with support from Colombia's Minister of Health.
Moreover, operational expenses in South America are significantly lower than in other regions, enabling medical technology firms to conduct high-quality research without the financial burdens typically associated with medical studies. As pharmaceutical companies increasingly engage in multicentric studies, the future of Medtech research in Latin America becomes increasingly apparent, establishing the region as a key player in the global research landscape.
The importance of medical research studies in propelling Medtech innovation cannot be overstated. They not only facilitate the development of new technologies but also ensure that these innovations comply with regulatory standards and address real-world patient needs. As the industry evolves, the focus on risk-based quality management and data transparency is transforming trial conduct, thereby enhancing their value.
For example, a Senior Director within a global biopharma’s research sciences and study management group remarked that "eliminating one 20-minute task per visit across 130,000 visits avoids 43,000 hours of work, allowing CRAs to focus on what matters."
Additionally, case studies underscore the shift towards increased data ownership and transparency, with sponsors adopting insourced models for data management, granting them direct access to live data and improving study quality. The ICH guideline E8(R1) emphasizes the necessity for sponsors to focus on critical-to-quality factors and manage risks appropriately, a principle that is becoming increasingly vital in the research environment.
In summary, the advantages of conducting research studies in South America in 2025 are clear and illustrate the future of Medtech research in Latin America: quicker recruitment, diverse populations, and reduced expenses all contribute to a more efficient research process. As the region continues to solidify its position as a hub for medical research, the significance of these studies in fostering healthcare technology innovations will only grow, with bioaccess® playing a crucial role in supporting these initiatives alongside collaborators like GlobalCare Clinical Studies, which has achieved over a 50% reduction in recruitment time and 95% retention rates.
Emerging Technologies Transforming Medtech Research
Emerging technologies are poised to revolutionize Medtech research in Latin America, with several key innovations leading the charge:
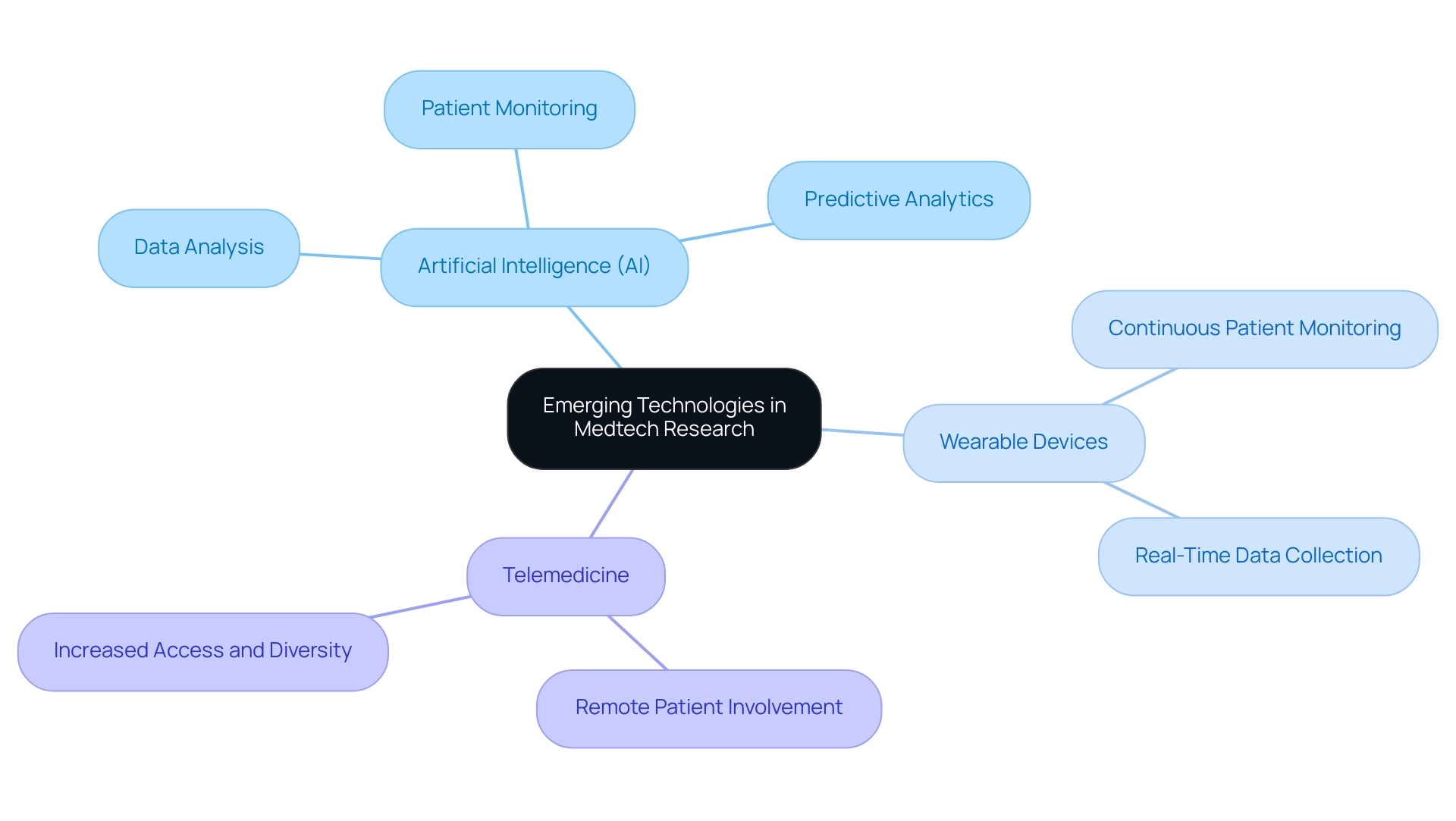
- Artificial Intelligence (AI): The integration of AI in clinical trials is transforming data analysis, patient monitoring, and predictive analytics. This technology not only enhances the efficiency of tests but also facilitates more precise patient selection and outcome forecasts, ultimately accelerating the path to market for new medical devices.
- Wearable Devices: The proliferation of wearable health technology enables continuous patient monitoring and real-time data collection. This capability is invaluable for medical research, as it allows researchers to gather extensive data on patient health metrics outside of traditional medical settings, leading to more robust and comprehensive study results.
- Telemedicine: The growth of telemedicine facilitates remote patient involvement in research studies, thereby increasing access and improving diversity within study groups. This trend is particularly significant in Latin America, where geographical barriers can limit patient recruitment. By utilizing telemedicine, researchers can engage a broader demographic, ensuring that studies are more representative of the general population.
As we look towards 2025, nearly 90% of health system executives anticipate that the future of medtech research in Latin America, along with the expanding adoption of digital tools and virtual health, will significantly influence their strategies. Furthermore, 72% of these executives recognize the importance of investing in platforms that support the use of digital tools, underscoring the critical role of technology in shaping the future of healthcare.
In this evolving landscape, bioaccess® distinguishes itself with its extensive research management services, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF). These services are essential for ensuring successful outcomes in clinical trials, especially as collaborations like that of bioaccess™ and Caribbean Health Group aim to position Barranquilla as a leading destination for clinical trials in South America, supported by Colombia's Minister of Health.
Case studies highlight the future of medtech research in Latin America, showcasing the evolving landscape of US-Latin American MedTech collaboration, driven by technological advancements and changing healthcare needs. These collaborations are paving the way for innovations in telemedicine and personalized medicine, illustrating the region's potential for growth. As Monica Mora, Chief Operating Officer focusing on operations, logistics, and compliance strategies for medical device firms in South America, states, "Navigating these complexities is vital to ensure that partnerships are both effective and in accordance with local regulations."
Stakeholders must remain proactive in addressing these emerging trends to fully capitalize on the opportunities presented by advancements in medical technology research.
Building Strategic Partnerships for Medtech Success
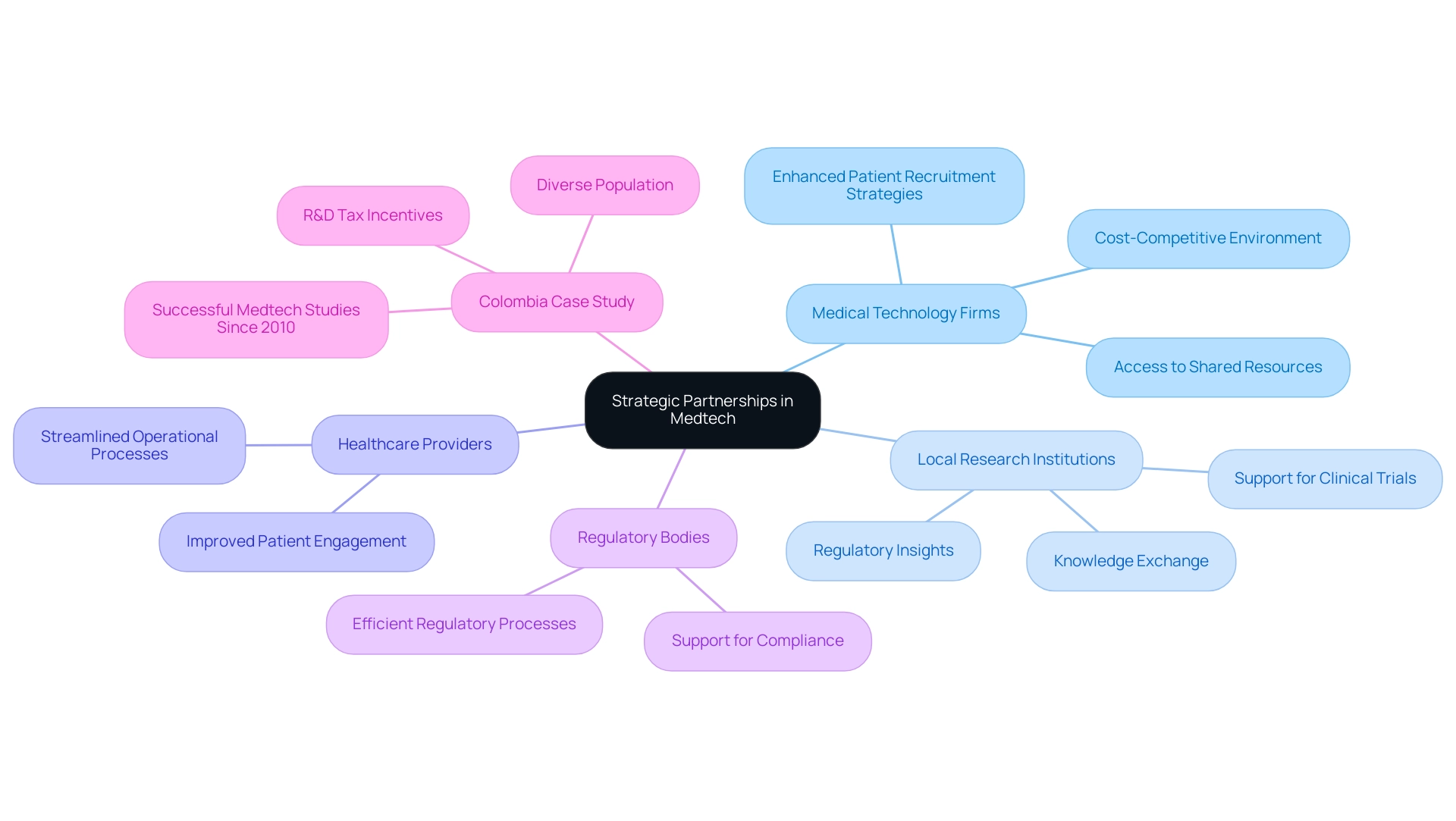
Strategic alliances are pivotal for the future of medtech research in Latin America, empowering medical technology firms to thrive. Collaborations with local research institutions, healthcare providers, and regulatory bodies not only enhance research capabilities but also streamline operational processes. By leveraging the strengths of these partnerships, companies gain access to shared resources, facilitate knowledge exchange, and implement more effective patient recruitment strategies.
In 2025, the landscape of medtech research in Latin America is increasingly shaped by proactive partnerships. For instance, Colombia, with its diverse population of nearly 52 million—80% of whom reside in urban areas—offers a fertile ground for Medtech studies. The country’s efficient regulatory processes and a history of successful studies dating back to 2010 make it an appealing destination for U.S. medical device companies.
As Julio G. Martinez-Clark, CEO of bioaccess™, notes, "Colombia’s combination of a large and diverse population, experienced research sites, efficient regulatory processes, a cost-competitive environment, and a track record of successful medtech studies since 2010 exemplifies why U.S. medical device firms should invest in South American studies."
The significance of strategic partnerships cannot be overstated. They not only bolster individual organizations but also fortify the overall healthcare ecosystem. Experts emphasize that fostering collaborations with local institutions is crucial for the future of medtech research in Latin America, as it can substantially mitigate challenges related to logistics and regulatory compliance.
Dushyanth Surakanti, Founder & CEO of Sparta Biomedical, shared insights from bioaccess® during its first human trial in Colombia, illustrating how such partnerships can enhance trial efficiency and patient engagement. George Frazer Moreira, Founder of FRAGIA CORP., reinforces this point by stating, "With challenges ranging from logistics to import regulations, creating an efficient supply chain in Latin America can seem daunting. But with the right knowledge and strategies, these challenges can be overcome."
This underscores how strategic partnerships can pave the way for more efficient pathways to market for innovative medical devices, significantly contributing to the future of medtech research in Latin America.
Moreover, the case study of Welwaze Medical Inc.'s collaboration with bioaccess™ for the launch of the Celbrea® medical device in Colombia highlights the necessity of collaboration in addressing regulatory challenges. By partnering with local research organizations, healthcare technology firms can tap into valuable insights and expertise that enhance their clinical research efforts. bioaccess™ provides essential regulatory and market access consulting services that facilitate these collaborations, ensuring that companies navigate the complexities of the Colombian market effectively.
Additionally, Colombia offers substantial R&D tax and financial incentives, including a 100% tax deduction for investments in science, technology, and innovation projects, further enhancing the appeal of conducting trials in the region. This collaborative approach is vital for navigating the complexities of the medical technology landscape and ensuring that life-changing therapies reach patients more effectively.
As the medical technology sector continues to evolve, emphasizing strategic collaborations will be essential for fostering innovation and achieving sustainable growth, which is crucial for the future of medtech research in Latin America.
Future Trends and Predictions for Medtech Research in Latin America
Looking ahead, several pivotal trends are poised to redefine Medtech research in Latin America:
-
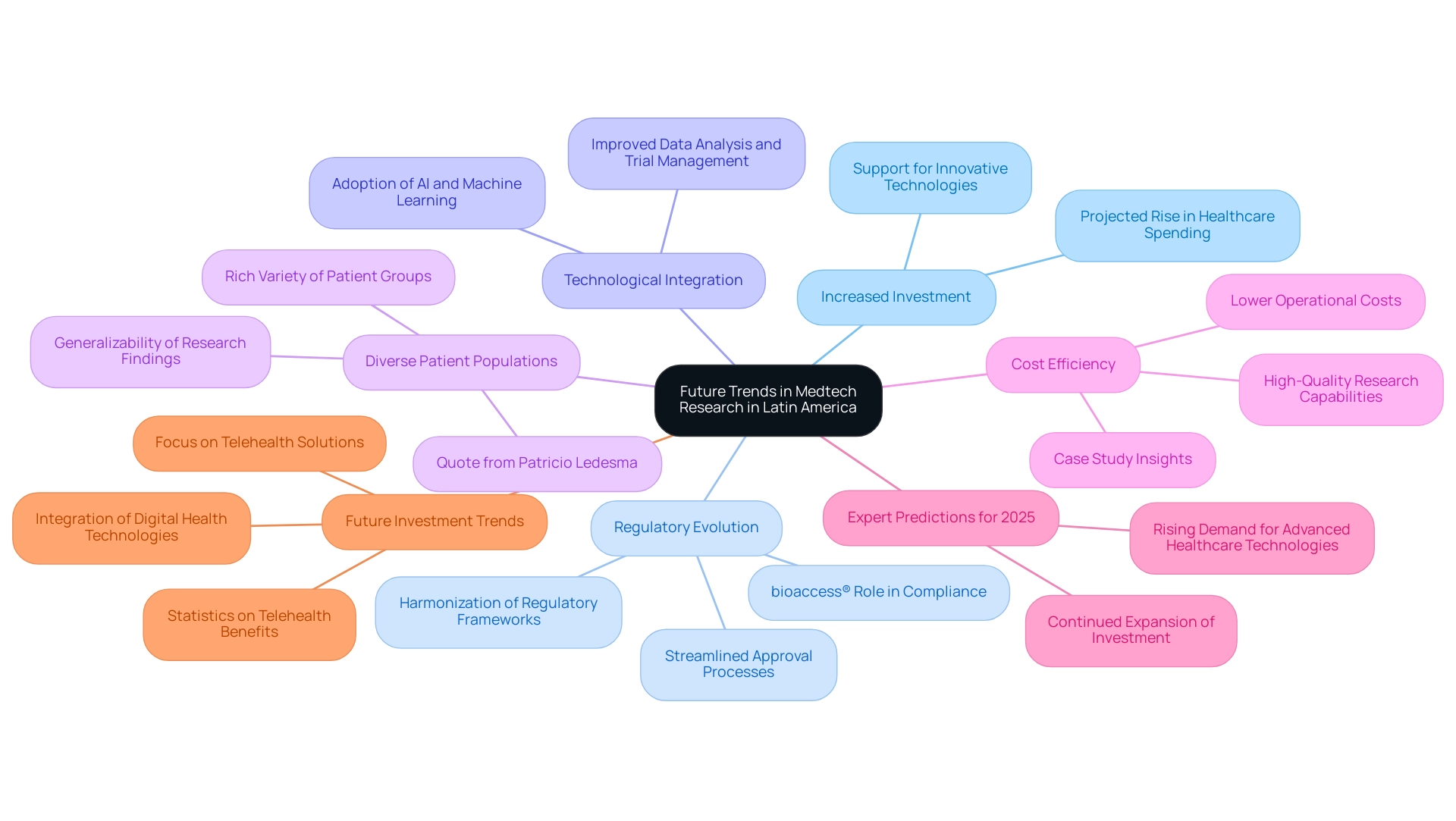
Increased Investment: With healthcare spending projected to rise significantly, the Medtech sector is expected to attract substantial investments. This influx of capital will not only support the development of innovative medical technologies but also enhance the overall infrastructure necessary for clinical research.
-
Regulatory Evolution: The ongoing harmonization of regulatory frameworks across Latin American countries is set to streamline the approval processes for new medical devices. This evolution will facilitate quicker market entry, allowing innovative solutions to reach patients faster and more efficiently. Organizations like bioaccess®, with over 20 years of experience in Medtech, are at the forefront of navigating these regulatory landscapes, ensuring compliance and successful market access for their clients.
-
Technological Integration: The adoption of advanced technologies, including artificial intelligence and machine learning, is anticipated to revolutionize medical studies. These technologies will improve data analysis, patient recruitment, and trial management, ultimately driving innovation and enhancing the quality of research outcomes.
-
Diverse Patient Populations: The distinctive demographic environment of this region provides a rich variety of patient groups, which is essential for research studies. As Patricio Ledesma, Head of Clinical Operations at Sofpromed CRO, observes, "The region of South America presents unique opportunities for biotech companies, especially concerning patient diversity and cost efficiency, which are vital for successful research studies." This diversity not only enhances the generalizability of research findings but also provides valuable insights into the efficacy of medical devices across different demographics.
-
Cost Efficiency: Conducting clinical trials in South America presents a cost-effective alternative compared to traditional markets. The combination of lower operational costs and high-quality research capabilities positions the region as an appealing destination for medical technology firms aiming to optimize their research budgets. The case study titled "Challenges and Opportunities in the Global Medical Devices Market" highlights how emerging markets such as South America present significant growth opportunities, fostering innovation and increasing access to medical technologies.
-
Expert Predictions for 2025: Industry specialists forecast that investment in medical technology within South America will keep expanding, propelled by the area's potential for innovation and the rising demand for advanced healthcare technologies. As emphasized by industry leaders, the unique opportunities offered by South America make it a focal point for biotech firms aiming to expand their reach.
-
Future Investment Trends: As we approach 2025, the medical technology landscape in South America is anticipated to evolve significantly, with increased focus on telehealth solutions and digital health technologies. Notably, 50% of insurers in Europe are already incorporating telehealth benefits, indicating a shift towards more integrated healthcare solutions that could influence investment strategies in the region.
In summary, the future of medtech research in Latin America is bright, characterized by increased investment, regulatory advancements, and the integration of cutting-edge technologies, all of which will contribute to a more robust and innovative healthcare ecosystem. Furthermore, bioaccess® plays a crucial role in bridging the gap between innovative Medtech companies and the potential for conducting clinical research studies in Latin America, specializing in Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), as evidenced by successful collaborations such as the launch of the Celbrea® medical device in Colombia.
Conclusion
Latin America stands at the forefront of Medtech innovation, propelled by a unique combination of economic growth, technological advancements, and a diverse patient demographic. With the region's Medtech market valued at over 44 billion USD in 2023, robust expansion is anticipated, particularly in Brazil, Mexico, and Argentina. Strategic partnerships and collaborations are vital, enhancing research capabilities and streamlining clinical trials, ultimately fostering a more effective healthcare ecosystem.
The regulatory landscape, while complex, is evolving towards harmonization, enabling quicker market access for innovative medical devices. Companies like bioaccess® are leading the charge in navigating these challenges, ensuring compliance and supporting the growth of Medtech initiatives across the region. Emerging technologies, including AI and telemedicine, are transforming clinical research methodologies, enhancing patient engagement, and improving data quality.
Looking forward, the future of Medtech research in Latin America appears promising. Increased investments, regulatory evolution, and the potential for cost-effective trials position the region as a key player in global healthcare innovation. By leveraging its strengths and addressing existing challenges, Latin America is poised to become a vital hub for Medtech research, paving the way for groundbreaking advancements that will benefit patients both locally and globally.
Frequently Asked Questions
What is the current state of the medical technology market in Latin America?
As of 2023, the medical technology market in Latin America was valued at over 44 billion USD, with projections indicating robust growth in the coming years.
Which countries are leading the expansion of the medical technology market in Latin America?
Brazil, Mexico, and Argentina are poised to lead the expansion, benefiting from favorable regulatory environments and a strong commitment to innovation.
What segments of the medical technology market are expected to grow significantly?
The Hospitals and Ambulatory Surgical Centers (ASCs) segment is expected to experience significant compound annual growth rates (CAGR) throughout the forecast period.
What are the projected growth rates for the Medtech market in Chile and Colombia?
The Medtech market in Chile is anticipated to grow at a CAGR of 9%, while Colombia is expected to grow at 8.4%, driven by a high incidence of chronic diseases and improvements in health systems supported by government initiatives.
Can you provide an example of a successful research initiative in Latin America?
A notable example is the partnership between bioaccess™ and Flow-FX, which has established Barranquilla as a leading site for research studies. Their first-in-human study on the Flow-Screw medical device for intraosseous antibiotic delivery highlights the region's capabilities.
How has the COVID-19 pandemic impacted the Medtech landscape in Latin America?
The COVID-19 pandemic has significantly influenced market value, trends, and growth in the Medtech sector, necessitating adaptability among stakeholders to navigate recovery and future developments.
What are the key drivers of growth for Medtech in Latin America?
Key drivers include economic growth leading to increased healthcare spending, technological advancements such as telemedicine and AI, and supportive regulatory environments that foster innovation.
How does regulatory support contribute to Medtech innovation in the region?
A supportive regulatory landscape, exemplified by initiatives such as the partnership between bioaccess™ and Caribbean Health Group, helps streamline clinical trial processes and attract both local and international companies to the region.
What role does demographic diversity play in the future of medtech research in Latin America?
The unique demographic diversity in Latin America provides a rich backdrop for medtech research, supporting various innovative medical devices and technologies, particularly in countries like Brazil, Mexico, and Argentina.
What is bioaccess®'s role in advancing medical technology research?
bioaccess® is committed to advancing medical devices through expertise and a tailored approach, managing various research studies and driving economic growth and healthcare improvement in the region.

