Overview
Statistical methods for trial design are indispensable for ensuring the scientific rigor and reliability of clinical studies. Techniques such as hypothesis testing, confidence intervals, and adaptive study designs are integral to this process. This article underscores how these methods, combined with a patient-centric approach, significantly enhance study efficiency and outcomes. This is particularly relevant in light of evolving regulatory frameworks and the integration of real-world data, which present both challenges and opportunities for clinical research.
Introduction
In the realm of clinical research, the application of statistical methods transcends mere technical necessity; it serves as a foundational element that shapes the integrity and outcomes of trials. As the complexity of medical studies escalates, a comprehensive understanding of key statistical concepts—such as hypothesis testing, confidence intervals, and adaptive trial designs—becomes essential for researchers aiming to achieve reliable and meaningful results.
The continuous emergence of innovations in statistical methodologies, coupled with the integration of patient-centric approaches and robust analytical frameworks, is revolutionizing the landscape of clinical trials. This article explores the pivotal role of statistical techniques in clinical trial design, emphasizing recent advancements, challenges, and the critical importance of upholding ethical standards, all while striving to enhance patient outcomes and advance medical knowledge.
Understanding Statistical Methods in Clinical Trial Design
Statistical methods for trial design are pivotal for the effective planning, execution, and analysis of clinical studies. These techniques ensure that experiments maintain scientific rigor and yield reliable outcomes. Central to these methods are key concepts such as hypothesis testing, confidence intervals, and the differentiation between descriptive and inferential statistics.
Hypothesis testing is crucial as it allows researchers to assess whether the observed effects in an experiment are statistically significant, thereby informing decisions about the efficacy of medical interventions. A well-structured hypothesis test can reveal whether a new medical device significantly improves patient outcomes compared to existing treatments. Confidence intervals enhance this by presenting a range of values that probably encompass the true effect size, providing understanding into the accuracy of the estimates obtained from the data.
Recent progress in statistical methods for trial design, especially in 2025, has led to the development of creative strategies for study structure, including adaptive studies that permit adjustments based on preliminary results. Christina Yap, a professor of Clinical Trials Biostatistics with over 20 years of experience, emphasizes the significance of openness and quality in study development, advocating for methods that prioritize patient-reported outcomes and adaptive strategies. This flexibility can enhance the efficiency of medical studies and improve patient outcomes.
Moreover, the increasing trend of multi-regional research underscores the need for robust statistical frameworks that can accommodate diverse populations and regulatory environments. The continuing growth of MRCTs is significant, and a free tutorial is available to provide an overview of these developments and major regulatory guidances expected in 2025. As the environment of medical research changes, understanding statistical methods for trial design becomes more crucial for researchers seeking to create studies that not only add to medical knowledge but also improve patient care.
The integration of hypothesis testing and confidence intervals, as part of the statistical methods for trial design, remains a cornerstone of this endeavor, ensuring that scientific conclusions are drawn from sound statistical foundations. As stated by the American Statistics Association, 'Scientific conclusions and business or policy decisions should not be based on whether the value passes a specified threshold.' This viewpoint emphasizes the significance of stringent statistical methods for trial design in medical studies.
Furthermore, bioaccess®'s 20+ years of experience in the Medtech sector and its customized approach to advancing medical devices sooner highlight the practical implications of these statistical methods in the context of Medtech advancements, particularly in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF) in Latin America. Comprehending the regulatory landscape in Latin America is essential, as it poses unique challenges that necessitate the use of statistical methods for trial design to ensure compliance and successful results.
The Role of Patient-Centric Design in Statistical Methodology
Patient-focused planning in medical studies underscores the critical need to integrate patient perspectives throughout the research process. This approach not only enhances participant engagement but also ensures that the outcomes measured resonate with patients' real-world experiences. In this context, statistical methods for trial design are pivotal, enabling researchers to analyze data in ways that reflect patient experiences and preferences.
For instance, the combination of qualitative data with quantitative measures yields a richer understanding of treatment effects, facilitating a more nuanced interpretation of results.
The recent partnership between bioaccess™ and Caribbean Health Group (CHG), which encompasses eight prominent healthcare institutions in Barranquilla, aims to position Barranquilla as a premier destination for medical studies in Latin America. This partnership, unveiled on March 29, 2019, during a meeting in Miami, FL, exemplifies the commitment to advancing clinical research in the region, ensuring that patient-centric methods remain at the forefront of research frameworks.
Adaptive study approaches illustrate how statistical methods for trial design can enhance patient-centricity. These designs employ statistical methods that allow modifications based on interim results, enabling researchers to respond dynamically to patient feedback and adjust study parameters accordingly. This flexibility is particularly beneficial in studies centered on patient needs, fostering a more adaptive research environment.
Moreover, GlobalCare Clinical Trials' collaboration with bioaccess™ has led to significant improvements in ambulatory services for studies in Colombia, achieving over a 50% reduction in recruitment duration and 95% retention rates. Such outcomes underscore the effectiveness of integrating patient feedback into the testing process.
Recent research highlights the importance of addressing barriers to patient participation and ensuring diverse representation in medical evaluations. For example, one study conducted four stakeholder meetings within the first 18 months to gather insights on patient engagement strategies, demonstrating a proactive approach to enhancing participation. Furthermore, the examination of patient-reported outcome measures in research studies, as discussed in the PCT Grand Rounds on September 22, 2021, reflects the growing recognition of patient viewpoints in shaping research results.
The impact of patient-focused design on research outcomes is significant. Statistics reveal that studies incorporating patient feedback tend to achieve higher retention rates and more meaningful results. By prioritizing patient engagement, researchers not only enhance the relevance of their findings but also contribute to the overall success of research initiatives.
Additionally, understanding the challenges of implementing patient-focused designs, as emphasized in the case analysis titled "Challenges of EHR Systems in Pragmatic Research," is essential for effective research design.
As Jill Feldman pointed out, addressing potential conflicts of interest is vital in research studies, further highlighting the importance of transparency and integrity in investigations. As the field continues to evolve, the integration of patient perspectives remains a cornerstone of effective research planning and execution.
Key Statistical Techniques for Clinical Trials
In clinical trials, various statistical methods for trial design are employed, each tailored to address specific research needs and enhance the validity of findings. These techniques include:
- Descriptive Statistics: This foundational method summarizes and describes the main features of a dataset, offering insights into the characteristics of the study population. By effectively presenting data, descriptive statistics help researchers understand trends and patterns that may influence study outcomes.
- Inferential Statistics: Techniques such as t-tests, ANOVA, and regression analysis enable researchers to draw conclusions about a larger population based on sample data. These statistical methods for trial design are essential for hypothesis testing and determining the significance of results, thereby allowing for informed decision-making in clinical research. As Meri Beckwith, Co-Founder, notes, "Researchers often conduct power analysis to determine the minimum sample size needed to detect an effect if one exists," highlighting the critical role of sample size determination in study design.
- Survival Analysis: Particularly important for studies involving time-to-event data, survival analysis estimates the duration until an event occurs, such as disease progression or mortality. This technique is vital for understanding treatment effects over time and is increasingly relevant in research focused on chronic conditions.
- Bayesian Methods: These innovative approaches utilize prior knowledge and evidence to update the probability of a hypothesis as new data emerges. Bayesian techniques offer a versatile framework for experimentation, adapting to changing information and improving the reliability of conclusions derived from medical research.
Each statistical technique possesses unique strengths and limitations, making the selection process critical. The choice of approach should correspond with the particular research question, framework, and features of the data. For example, grasping fundamental statistical ideas like population, sample size, and variability is essential for navigating trial statistics.
A well-defined population not only guides research design but also enhances the relevance of findings, ensuring that results are valid and generalizable. Careful selection of sample size is paramount to avoid inconclusive or misleading outcomes, highlighting how statistical methods for trial design are essential for maintaining statistical rigor in clinical research. A case analysis titled 'Basic Concepts in Clinical Trial Statistics' illustrates this point, emphasizing that a careful selection of sample size ensures valid and generalizable results.
Moreover, current industry trends, such as the emphasis on MDR solutions to connect study design, data collection, analysis, and submission, highlight the challenges many companies face in scaling metadata management, often relying on spreadsheets. This context underscores the importance of statistical methods for trial design in navigating these complexities.
At bioaccess®, with over 20 years of experience in Medtech, our expertise and customized approach aim to help advance medical devices sooner for companies in the Medtech industry. We focus on extensive research study management services, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies. Our commitment ensures that research strategies incorporate statistical methods for trial design, making them effective and aligned with the latest developments while addressing the challenges faced in metadata management.
Sample Size Determination and Randomization Strategies
Sample size determination stands as a cornerstone of study design, employing statistical methods that significantly influence a research project’s ability to identify meaningful differences between treatment groups. Researchers must weigh several critical factors, including expected effect size, data variability, and the desired level of statistical power, when utilizing statistical methods for trial design to calculate the appropriate sample size. For instance, in a recent pain study involving Drug A, the clinically important difference was established at 0.5, a metric that bioaccess® incorporates into its clinical research services, thereby ensuring that studies are designed to yield robust outcomes.
A variety of formulas and software tools, incorporating statistical methods for trial design, are readily available to assist researchers in this vital process, facilitating informed decision-making. Equally crucial are randomization strategies, which play an essential role in minimizing bias during participant assignment to treatment groups. Methods such as:
- simple randomization
- stratified randomization
- block randomization
can significantly enhance the integrity of the study. For example, stratified randomization guarantees that specific subgroups are adequately represented, thereby improving the generalizability of the results.
As Tushar Vijay Sakpal, Principal Statistician at PharmaNet Clinical Services, aptly notes, "sample size is an important factor in approval/rejection of CT results irrespective of how clinically effective/ineffective the test drug may be." By meticulously planning both sample size and randomization methods, researchers can leverage statistical methods for trial design to bolster the credibility and reliability of their findings, ultimately advancing the development of medical devices and enhancing patient outcomes. This strategic approach is particularly pertinent in the context of Latin America, where bioaccess® draws upon its extensive experience in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF) to effectively support Medtech startups in navigating these complexities, as demonstrated in the case study titled "bioaccess®: Advancing Medtech in Latin America.
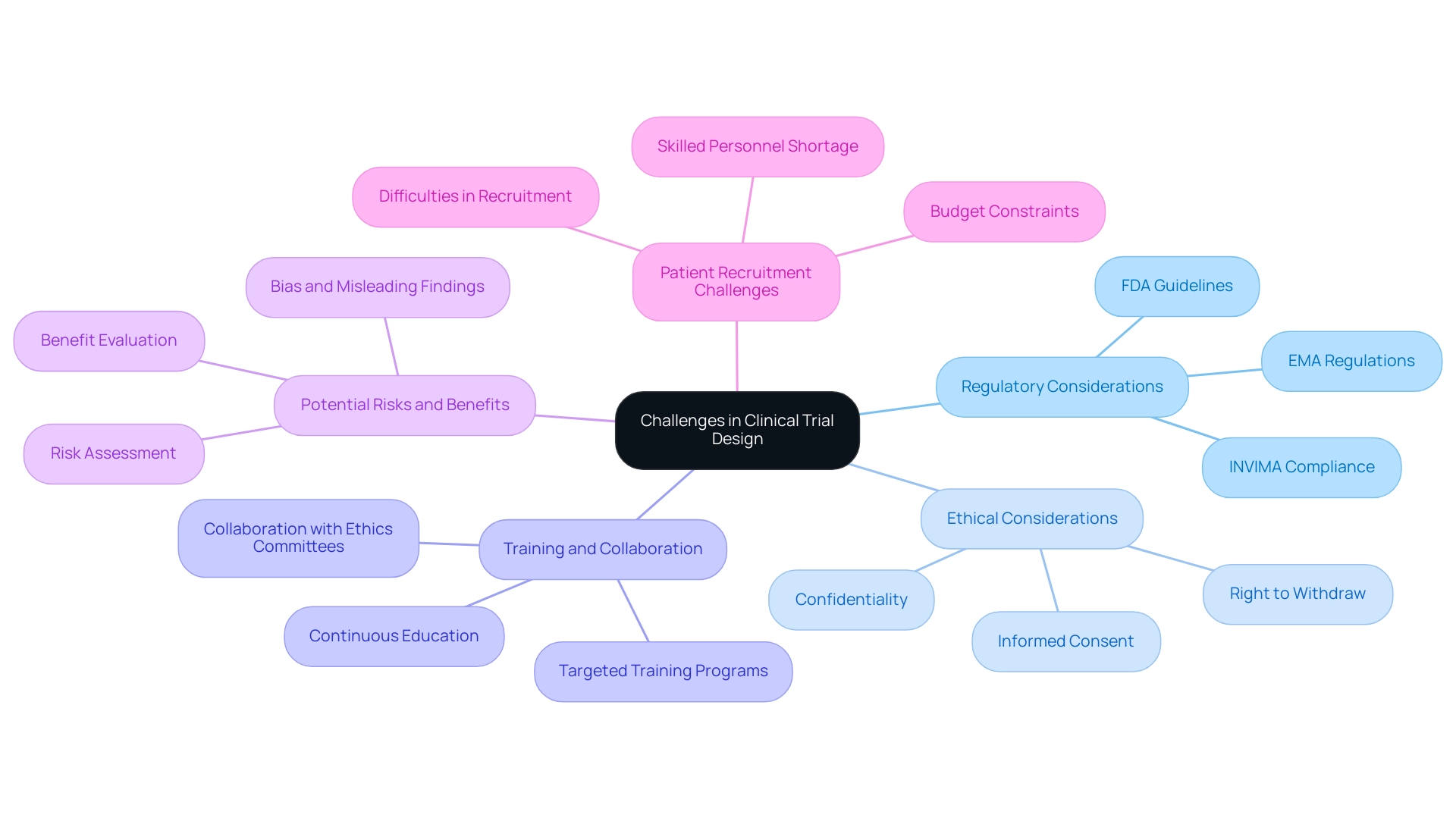
Challenges in Clinical Trial Design: Regulatory and Ethical Considerations
Creating clinical studies necessitates a thorough understanding of the intricate regulatory and ethical landscape that governs research practices. Regulatory authorities, including the FDA, EMA, and INVIMA—the Colombian National Food and Drug Surveillance Institute—enforce stringent guidelines to safeguard the safety and efficacy of new treatments. Familiarity with these regulations is essential for researchers to avoid compliance issues that could jeopardize the integrity of their studies.
Equally important are the ethical considerations that safeguard the rights and welfare of participants. Key components such as informed consent, confidentiality, and the right to withdraw from a research project are fundamental to ethical conduct. Researchers must carefully evaluate the potential risks and benefits associated with participation, ensuring that study designs are structured to minimize harm while maximizing potential advantages.
Recent statistics indicate that non-compliance with best practices can lead to biased and misleading findings, ultimately undermining research integrity. The CONSORT Statement, which includes a 22-item checklist and a flow diagram for reporting randomized experiments, serves as an essential tool for emphasizing the need for transparency and rigor in research design.
A systematic review analyzing challenges in medical experiments from 1991 to 2017 highlights significant barriers, including ethical and regulatory complexities, difficulties in patient recruitment, and constraints related to budgets and skilled personnel. This review underscores the necessity for targeted training to improve the quality of research studies, particularly in navigating the evolving regulatory challenges of 2025.
As Katherine Ruiz, an expert in regulatory affairs for medical devices and in vitro diagnostics in Colombia, emphasizes, addressing these multifaceted challenges requires meticulous planning, continuous education, and collaboration with regulatory and ethics committees. By prioritizing thorough management services for research—such as feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting—researchers can design experiments that not only adhere to regulatory standards but also maintain the highest ethical principles, ultimately contributing to the progress of medical technology.
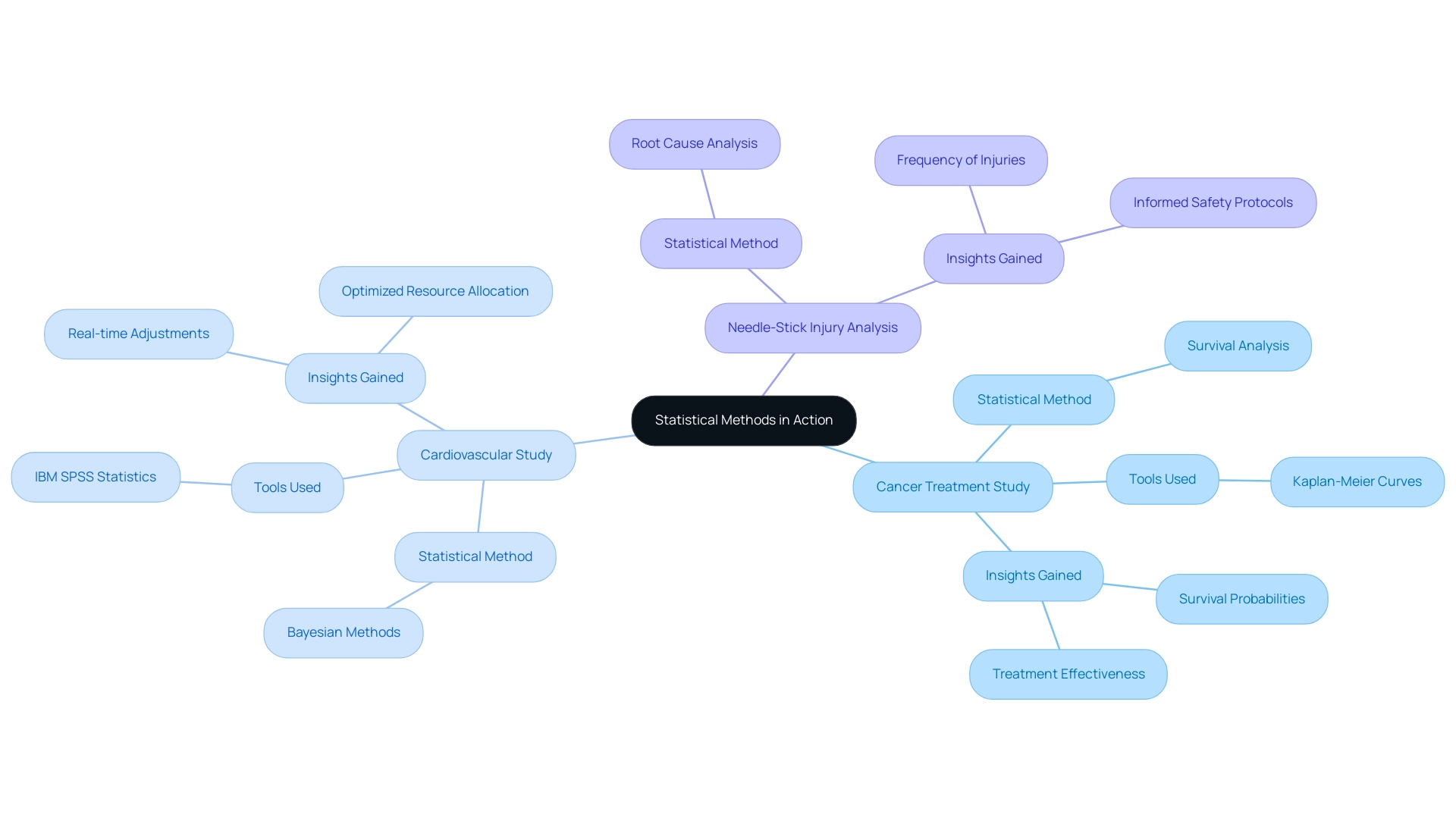
Case Studies: Statistical Methods in Action
Case analyses serve as essential examples of the effective utilization of statistical techniques in research, showcasing their significant influence on results. A notable instance is a recent study assessing a novel cancer treatment, which employed survival analysis to evaluate the time to progression among patients. By utilizing Kaplan-Meier curves, researchers adeptly illustrated survival probabilities over time, providing critical insights into the treatment's effectiveness and informing future medical decisions.
In another compelling case, a cardiovascular study employed Bayesian methods, allowing for adaptive alterations to the study framework based on interim results. This innovative approach enabled researchers to make real-time adjustments, optimizing resource allocation and enhancing the overall efficiency of the experiment. The use of IBM SPSS Statistics for Windows, Version 26.0, facilitated multivariate analysis, underscoring the importance of robust statistical tools in clinical research. Furthermore, sequential methods have demonstrated the ability to save time, sample size, and costs compared to standard fixed-sample procedures, further emphasizing the efficiency of statistical methods for trial design.
A pertinent case examination is the Needle-Stick Injury Incidence Analysis, which investigates the occurrence of needle-stick injuries among healthcare workers. This research highlights the application of statistical methods in identifying root causes and informing safety protocols.
As noted by the American Statistics Association, 'Scientific conclusions and business or policy decisions should not be based on whether the value passes a specified threshold.' This statement underscores the importance of selecting appropriate statistical techniques that align with the specific goals and challenges present in each research study. Ultimately, these considerations contribute to more robust and reliable outcomes, aligning with bioaccess®'s mission to bridge the gap between innovative Medtech companies and the potential for conducting research studies in Latin America.
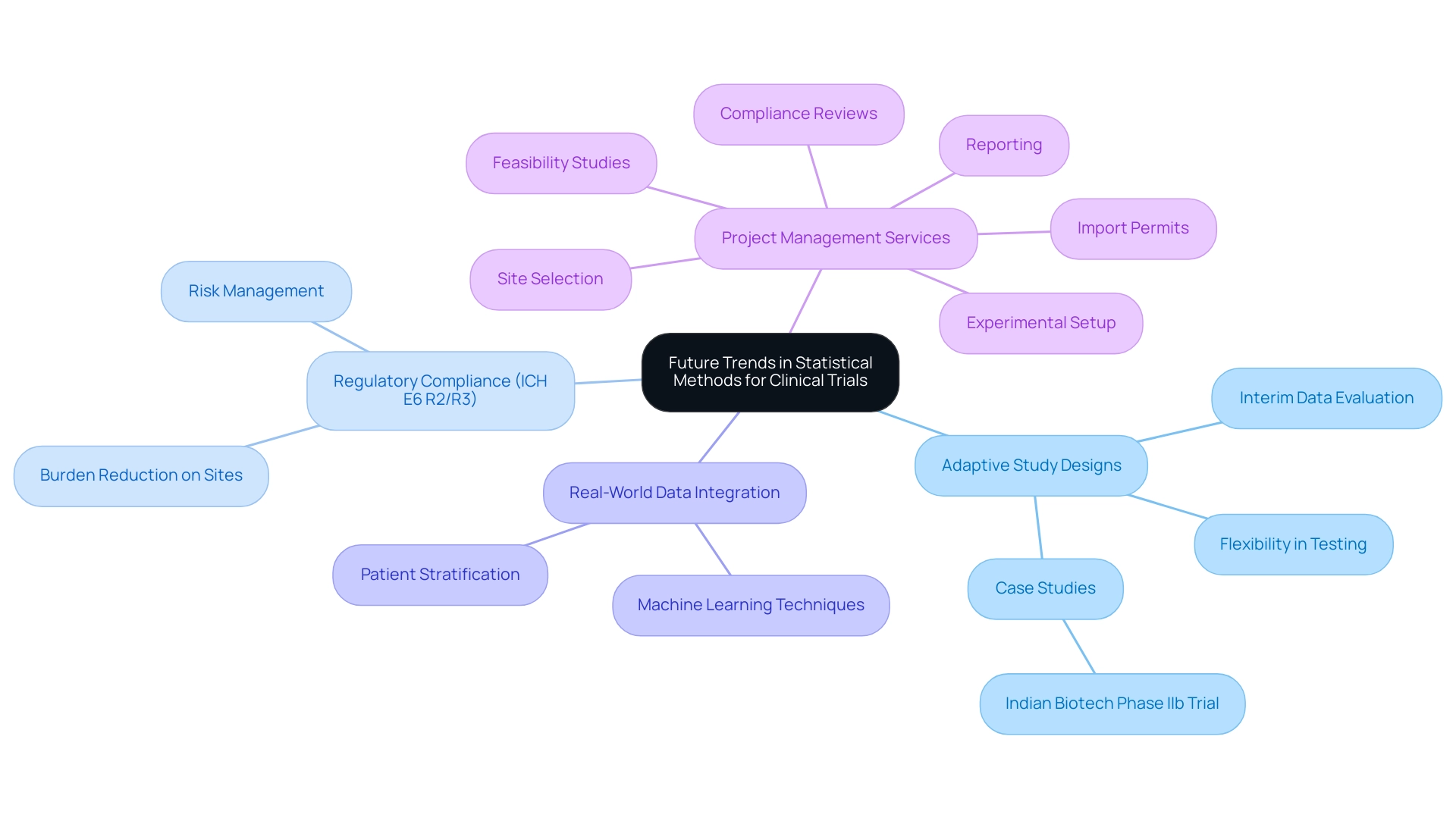
Future Trends in Statistical Methods for Clinical Trials
The framework of clinical experimentation is undergoing significant transformation, driven by advancements in statistical techniques and comprehensive clinical research management services. A noteworthy trend is the adoption of adaptive study formats, which facilitate adjustments based on interim data evaluation. This flexibility not only enhances testing efficiency but also expedites decision-making processes, leading to more timely and relevant outcomes.
In this context, the ICH E6 R2 (and R3 revisions) requires sponsors to manage risk in a manner that alleviates the burden on sites, thereby further bolstering the efficiency of adaptive designs.
Moreover, bioaccess offers a wide array of services including:
- Feasibility studies
- Site selection
- Compliance reviews
- Experimental setup
- Import permits
- Project management
- Reporting
This holistic approach ensures that research studies not only comply with national requirements but are also optimized for success. Bioaccess's compliance evaluations are designed to guarantee adherence to regulatory standards, while its project management processes facilitate the seamless execution of studies.
The integration of real-world data with advanced machine learning techniques is gaining prominence, enabling researchers to refine their analyses and enhance patient stratification, which ultimately fosters more personalized medicine strategies. Such innovations strengthen test outcomes and align with the increasing emphasis on optimizing development pathways to achieve commercial success in medical studies.
As Max Baumann, Head of Execution at Treehill Partners, noted, "Entering 2025, we still witness biotech encountering essential business model obstacles as medical end-markets become increasingly saturated," highlighting the necessity for flexible study frameworks to navigate these complexities. As we progress into 2025, it is imperative for medical researchers to stay abreast of these evolving methodologies. The shift towards flexible study frameworks, underpinned by statistical methods for trial design and extensive management services from bioaccess, is reshaping the future of clinical research, necessitating that professionals in the field adapt their strategies accordingly.
Insights from recent case studies, such as the Phase IIb trial conducted by an Indian biotech firm, underscore the potential of adaptive designs to generate valuable data that informs treatment development and commercialization pathways.
Conclusion
The integration of statistical methods into clinical trial design stands as a fundamental pillar, essential for producing reliable and meaningful results that advance medical knowledge and enhance patient care. Key statistical techniques such as hypothesis testing, confidence intervals, and adaptive trial designs are crucial in ensuring the scientific rigor of studies. As emphasized throughout the article, these methodologies empower researchers to discern significant treatment effects, accommodate patient perspectives, and respond dynamically to interim results, thereby enhancing trial efficiency and relevance.
Furthermore, the significance of patient-centric designs cannot be overstated. By incorporating patient feedback and focusing on outcomes that matter to individuals, researchers can markedly improve participant engagement and retention, leading to more robust findings. The highlighted case studies illustrate the practical application of statistical methods in real-world settings, reinforcing the necessity of selecting appropriate techniques tailored to specific research needs.
As the clinical research landscape continues to evolve, it is imperative for researchers to stay abreast of emerging trends and innovations in statistical methodologies. The adoption of adaptive trial designs, the integration of real-world data, and a commitment to ethical standards will shape the future of clinical trials. By prioritizing these approaches, researchers can enhance the quality and integrity of their studies and contribute to the broader goal of improving patient outcomes and advancing the field of medicine. The journey towards more effective clinical trials is ongoing, and the commitment to rigorous statistical practices remains a vital element of success.
Frequently Asked Questions
Why are statistical methods important for trial design in clinical studies?
Statistical methods are crucial for the effective planning, execution, and analysis of clinical studies, ensuring scientific rigor and reliable outcomes.
What are some key concepts in statistical methods for trial design?
Key concepts include hypothesis testing, confidence intervals, and the differentiation between descriptive and inferential statistics.
How does hypothesis testing contribute to clinical research?
Hypothesis testing allows researchers to determine if observed effects in an experiment are statistically significant, which informs decisions about the efficacy of medical interventions.
What role do confidence intervals play in trial design?
Confidence intervals provide a range of values that likely encompass the true effect size, enhancing the understanding of the accuracy of estimates obtained from the data.
What advancements in statistical methods were noted in 2025?
Advancements include the development of adaptive study designs that allow modifications based on preliminary results, enhancing the efficiency of medical studies.
How does multi-regional research impact statistical methods for trial design?
Multi-regional research necessitates robust statistical frameworks to accommodate diverse populations and regulatory environments, highlighting the importance of these methods in clinical studies.
What is the significance of patient-focused planning in medical studies?
Patient-focused planning integrates patient perspectives throughout the research process, enhancing engagement and ensuring that outcomes resonate with real-world experiences.
How do adaptive study approaches enhance patient-centricity?
Adaptive study designs allow modifications based on interim results and patient feedback, fostering a more responsive research environment.
What are the benefits of integrating patient feedback into clinical studies?
Integrating patient feedback can lead to higher retention rates and more meaningful results, ultimately enhancing the relevance and success of research initiatives.
Why is transparency and integrity important in research studies?
Addressing potential conflicts of interest is vital to maintain trust and credibility in research, ensuring that findings are valid and reliable.

