Overview
Latin America is key for medtech research due to its diverse patient populations, increasing healthcare investments, and a strong focus on innovation, which create an ideal environment for clinical studies and technological advancements. The article highlights various successful case studies, regulatory frameworks, and local collaborations that demonstrate the region's capacity for impactful research, making it an attractive destination for medical technology firms seeking to optimize their studies while addressing unique healthcare needs.
Introduction
Latin America is rapidly emerging as a pivotal player in the Medtech research landscape, driven by a unique combination of diverse demographics, increasing healthcare investments, and a strong emphasis on innovation. As illustrated by groundbreaking initiatives like ReGelTec's Early Feasibility Study for the HYDRAFIL™ treatment in Colombia, the region is not only advancing medical technology but also establishing itself as a prime destination for clinical trials.
The rich tapestry of patient populations across Latin America offers invaluable insights that can shape tailored healthcare solutions, while local collaborations and robust regulatory frameworks further enhance the region's appeal to Medtech companies.
This article delves into the strategic importance of Latin America in Medtech research, exploring its advantages, regulatory challenges, and the cultural dynamics that influence successful collaborations, ultimately spotlighting the vast opportunities for innovation and investment in this burgeoning market.
The Strategic Importance of Latin America in Medtech Research
The region of Central and South America has solidified its status as a critical area for medical technology research, which illustrates why Latin America is key for medtech research, fueled by a confluence of diverse demographics, escalating healthcare investments, and a heightened focus on innovation. A recent case study on ReGelTec's Early Feasibility Study involving the innovative HYDRAFIL™ treatment for chronic low back pain in Colombia exemplifies the region's commitment to advancing medical technology. This study successfully treated eleven patients with degenerative disc disease, utilizing a patented hydrogel that is melted before injection into the nucleus of a degenerated disc via a 17-gauge needle.
The procedures were supervised remotely through Zoom, highlighting how local collaborations, such as those between bioaccess™ and the Caribbean Health Group, establish Barranquilla as a prominent destination for medical trials in South America, with the backing of Colombia's Minister of Health. The varied patient populations across the region provide an invaluable source of data, enabling the development of technologies tailored to distinct healthcare needs. Furthermore, the impact of medical technology clinical studies extends to local economies, creating jobs, promoting economic growth, and improving healthcare services.
This is further supported by collaboration efforts, such as those by IDx Technologies with bioaccess™ to identify American ophthalmology centers for AI-based disease detection. As healthcare systems progress, the demand for advanced medical technologies rises, creating an appealing environment for medical technology firms. Enhanced regulation and supervision through consolidated sustainable finance frameworks will be essential to ensure the effectiveness of these initiatives and mitigate risks.
By capitalizing on these dynamics, South America illustrates why Latin America is key for medtech research initiatives, fostering an environment ripe for innovation and investment.
Leveraging Latin America's Unique Advantages for Medtech Innovation
The distinct benefits that Latin America offers for medical technology innovation highlight why Latin America is key for medtech research, primarily due to its diverse patient population. This demographic variability is crucial for research that seeks to assess the effectiveness of medical devices among various groups, highlighting why Latin America is key for medtech research. Furthermore, the cost-efficiency of performing medical studies in this region illustrates why Latin America is key for medtech research, as it allows Medtech firms to optimize their research funds while upholding high quality standards.
Significantly, extensive research study management services, including:
- feasibility assessments
- site selection
- compliance evaluations
- study setup
- import permits
- project oversight
- reporting
are expertly managed by organizations like bioaccess®. This expertise is exemplified by Flow-FX’s recent first-in-human medical study of the Flow-Screw device for intraosseous antibiotic delivery, conducted in Barranquilla, Colombia, under the guidance of Dr. Carlos Severini, demonstrating the region's capability for advanced medical research. Additionally, bioaccess® specializes in various types of studies, including:
- Early-Feasibility Studies
- First-In-Human Studies
- Pilot Studies
- Pivotal Studies
- Post-Market Clinical Follow-Up Studies
Colombia's National Food and Drug Surveillance Institute (INVIMA) plays a crucial role as a Level 4 health authority, ensuring robust oversight and regulation of medical devices. The influence of medical technology studies goes beyond separate experiments, aiding local economies through:
- job creation
- economic advancement
- healthcare enhancement
- promoting international cooperation
As emphasized by Julio G. Martinez-Clark, CEO of bioaccess, Colombia's ambitious science, technology, and innovation plan for 2022–2031 reflects a broader regional commitment to becoming a knowledge economy.
Moreover, the partnership between bioaccess™ and Caribbean Health Group establishes Barranquilla as a prominent location for medical studies in South America, backed by Colombia's Minister of Health. The expanding infrastructure and rising investments in healthcare technology reinforce the region's status as a prime location for medical technology research, illustrating why Latin America is key for medtech research through its attractive combination of diversity, affordability, and expertise that positions the area advantageously for impactful and broadly applicable research outcomes.
Navigating the Regulatory Landscape for Medtech in Latin America
The regulatory environment in South America presents both challenges and opportunities for medical technology firms, highlighting why Latin America is key for medtech research. With bioaccess® as your reliable CRO, we assist in speeding up medical device research studies amidst a population of around 7 million in Paraguay and an urbanization rate of 61.6%, emphasizing the considerable market opportunity for healthcare innovations in urban areas. Each South American nation has a diverse set of regulations governing clinical studies, leading to unique approval timelines and requirements that can greatly influence research endeavors.
Notably, with around 30% of the population in Latin America under the age of 14, this demographic shift illustrates why Latin America is key for medtech research, as there is a growing elderly patient demographic and an expanding drug market potential that medical technology firms can tap into. While some nations may provide expedited approval processes for innovative medical devices, others impose stringent regulations that can prolong testing timelines. Engaging with local authorities and remaining informed about the regulatory environment is crucial for medical technology companies.
In Chile, for example, compliance with international standards has led to 85% of international sponsors evaluating the data quality from Chilean studies as 'excellent' or 'very good,' based on recent survey findings. Moreover, the integration of AI and automation in regulatory affairs is revolutionizing the way companies navigate these challenges, enhancing efficiency and compliance. By adopting a proactive approach to regulatory compliance and fostering relationships with local regulatory bodies, Medtech companies can better understand why Latin America is key for medtech research, enhancing their opportunities for successful evaluations and building essential trust for long-term partnerships in the region, a strategy that bioaccess® excels in.
Additionally, it is important to address any queries or concerns regarding the processing of your information. You can reach out to our Grievance Officer at IMH ASSETS CORP (doing business as 'bioaccess®'), ensuring that your data protection inquiries are handled with care. As Dr. Adrian Ebner highlighted, effectively navigating these regulatory challenges can lead to significant advancements in clinical research outcomes.
Furthermore, understanding best practices for regulatory compliance can enhance the effectiveness of your trials, ensuring that you are well-prepared to meet the diverse requirements across countries in South America.
Overcoming Cultural and Operational Challenges in Medtech Collaborations
Cultural and operational challenges pose significant barriers to medical technology collaborations, highlighting why Latin America is key for medtech research. Variations in communication styles, work ethics, and expectations can lead to misunderstandings that may delay project timelines. For instance, the preference for less invasive diagnostic methods is indicative of broader cultural inclinations; as noted,
‘For example, with the rapid test … they were very happy with that.
But I mean, the people likes that, you know, it’s because, less invasive.’
This insight highlights the necessity for Medtech companies to invest in cultural competency training for their teams, equipping them to navigate these differences effectively. Furthermore, the experiences of industry leaders, such as Dushyanth Surakanti of Sparta Biomedical during the bioaccess® research in Colombia, serve as a testament to the region's potential. The competitive benefits of Colombia for first-in-human clinical studies, including:
- Over 30% cost savings compared to North America
- A swift IRB/EC and MoH review process of just 90-120 days
- A high-quality healthcare infrastructure ranked among the best in South America
illustrate why Latin America is key for medtech research.
Furthermore, Colombia provides attractive R&D tax incentives, such as:
- A 100% tax deduction for investments in science, technology, and innovation projects
- A 25% tax discount
- A 50% future tax credit
- Approximately $10 million in government grants
which illustrates why Latin America is key for medtech research. The orthopedic devices market in Latin America, valued at 3.6 billion USD, illustrates why Latin America is key for medtech research and underscores the potential for growth. Establishing clear communication channels is vital in fostering an inclusive environment that respects local customs and practices, thereby enhancing trust and collaboration among stakeholders.
Effective communication strategies, such as regular updates and culturally tailored messaging, can help medical technology companies address misunderstandings and build stronger partnerships. The evolving landscape for US-Latin American MedTech collaboration illustrates why Latin America is key for medtech research, revealing innovative solutions and partnerships emerging in telemedicine and personalized medicine, as detailed in the case study 'Future Trends and Opportunities in US-Latin American MedTech Collaboration.'. Furthermore, effective communication of the benefits of technology to the community is crucial for its adoption and sustainability, as differing perspectives on benefits can lead to rejection.
By proactively addressing these cultural dynamics and communication challenges, medical technology companies can create a more cohesive working environment, which illustrates why Latin America is key for medtech research, paving the way for successful research outcomes and capitalizing on innovative opportunities in the region.
Future Trends and Opportunities in Latin America's Medtech Sector
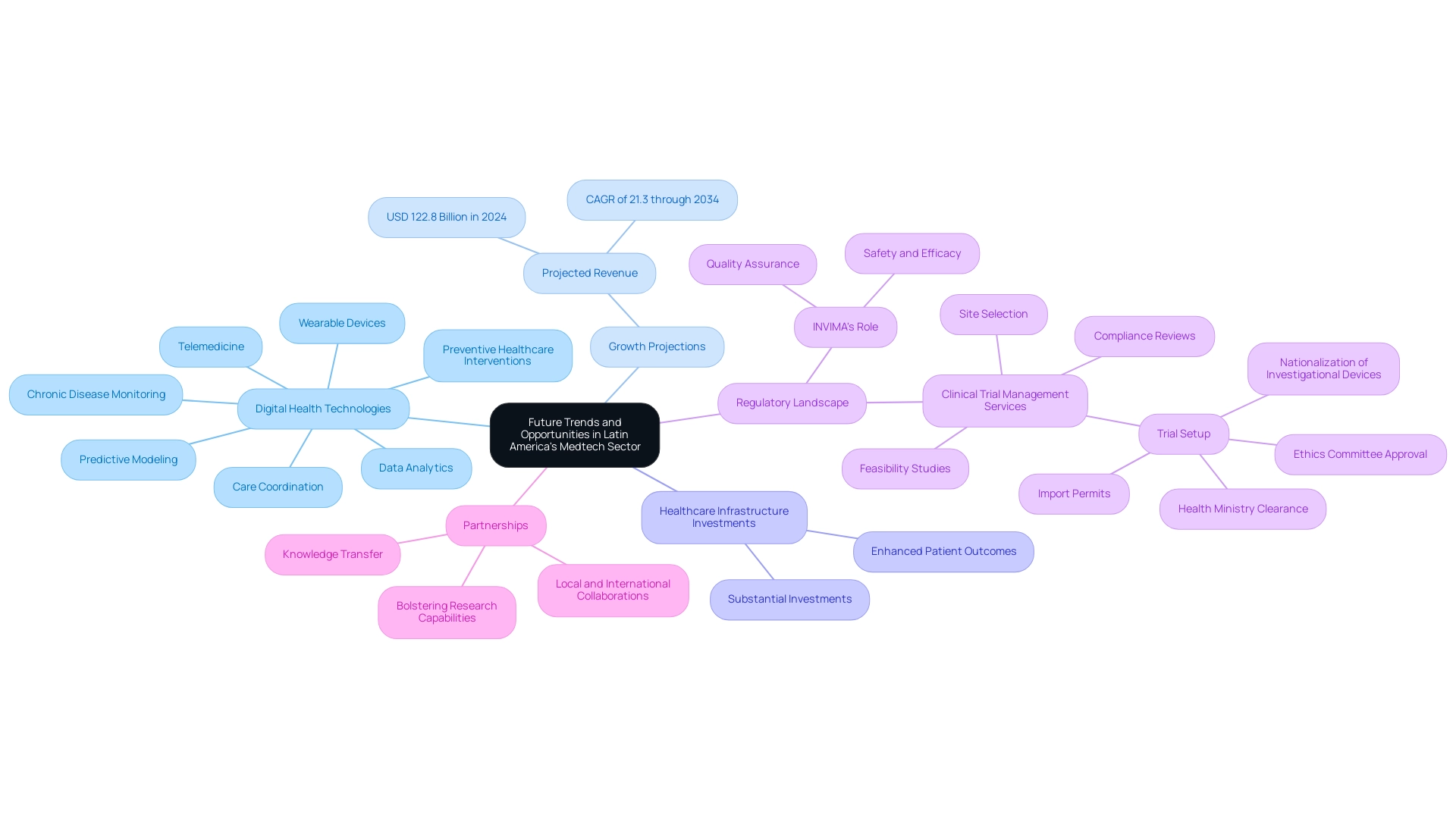
The significant transformation occurring in the medical technology sector in Latin America illustrates why Latin America is key for medtech research, driven by a variety of promising trends. The continuous integration of digital health technologies, especially in fields like telemedicine and wearable devices, has been hastened by the COVID-19 pandemic, transforming the healthcare landscape and creating opportunities for novel medical technology solutions. As highlighted by Mariam Faizullabhoy,
The services segment is expected to generate USD 122.8 billion in 2024 and maintain its dominance with a CAGR of 21.3% through 2034.
This growth is further fueled by substantial investments in healthcare infrastructure, which will inevitably heighten the demand for advanced medical technologies aimed at enhancing patient outcomes. Digital technology supports population health management by enabling:
- Data analytics
- Predictive modeling
- Care coordination
- Chronic disease monitoring
- Preventive healthcare interventions
Moreover, comprehensive clinical trial management services, including:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup (which involves ethics committee approval and health ministry clearance)
- Import permits
- Nationalization of investigational devices
are crucial for navigating the regulatory landscape established by INVIMA, Colombia's National Food and Drug Surveillance Institute.
This regulatory authority plays a pivotal role in ensuring the safety, efficacy, and quality of medical devices, further promoting local economic growth through job creation and improved healthcare systems. Additionally, we expect a surge in partnerships between local firms and international medical technology companies, which will facilitate essential knowledge transfer and bolster research capabilities. By remaining attuned to these evolving trends, stakeholders can strategically position themselves to understand why Latin America is key for medtech research and seize the burgeoning opportunities within the sector.
Conclusion
Latin America is increasingly recognized as a vital hub for Medtech research, characterized by its diverse demographics, innovative spirit, and growing investment in healthcare. The region's unique advantages, including a wide-ranging patient population and cost-effective clinical trial management, enable Medtech companies to conduct meaningful research while maximizing their resources. Initiatives such as ReGelTec's Early Feasibility Study in Colombia serve as prime examples of how local collaborations and regulatory support can enhance the development of novel medical technologies.
Navigating the regulatory landscape remains a key challenge for Medtech firms; however, proactive engagement with local authorities can facilitate smoother approval processes and foster long-term partnerships. Understanding cultural dynamics and communication styles is equally crucial, as it helps bridge gaps that could impede collaboration. By investing in cultural competency and establishing clear communication channels, Medtech companies can enhance trust and cooperation, ultimately leading to successful research outcomes.
Looking ahead, the Medtech sector in Latin America is poised for transformation, driven by trends such as the rise of digital health technologies and increased healthcare infrastructure investment. As stakeholders align with these developments, there is a significant opportunity to leverage the region's strengths for innovation and growth. Embracing the potential of Latin America not only paves the way for advancements in medical technology but also contributes to improved healthcare outcomes and economic prosperity in the region.
Frequently Asked Questions
Why is Latin America considered a key region for medical technology research?
Latin America is crucial for medtech research due to its diverse demographics, increasing healthcare investments, and a strong focus on innovation, making it an appealing environment for medical technology firms.
What recent case study highlights the region's commitment to advancing medical technology?
A recent case study on ReGelTec's Early Feasibility Study of the HYDRAFIL™ treatment for chronic low back pain in Colombia demonstrated the region's commitment, successfully treating eleven patients with degenerative disc disease.
How does the patient diversity in Latin America benefit medical technology research?
The varied patient populations provide invaluable data for assessing the effectiveness of medical devices across different demographic groups, which is essential for developing technologies tailored to specific healthcare needs.
What are some of the economic impacts of medical technology clinical studies in the region?
Medical technology studies contribute to local economies by creating jobs, promoting economic growth, and enhancing healthcare services.
What role does bioaccess™ play in medical technology research in Latin America?
Bioaccess™ manages extensive research study services, including feasibility assessments, site selection, compliance evaluations, study setup, and project oversight, facilitating various types of medical studies in the region.
What regulatory body oversees medical devices in Colombia?
Colombia's National Food and Drug Surveillance Institute (INVIMA) serves as a Level 4 health authority, ensuring robust oversight and regulation of medical devices.
How do collaborations enhance medical research in Latin America?
Collaborations, such as those between bioaccess™ and Caribbean Health Group, help establish prominent locations for medical studies, supported by local health authorities, enhancing the region's research capabilities.
What future plans does Colombia have for science and technology?
Colombia's science, technology, and innovation plan for 2022–2031 reflects a commitment to becoming a knowledge economy, emphasizing the region's dedication to advancing medical technology research.
What are the types of studies bioaccess® specializes in?
Bioaccess® specializes in various studies, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies.
What factors contribute to Latin America's attractiveness for medtech research?
The region's attractiveness stems from its demographic variability, cost-efficiency in conducting studies, expertise in research management, and the supportive role of health authorities, all fostering impactful research outcomes.

