Overview
Entering the Latin America medtech industry necessitates a comprehensive understanding of the region's unique regulatory landscape, market dynamics, and the significance of local partnerships. Successfully navigating these factors is critical. Companies that leverage clinical trial opportunities and engage in sustainable practices are poised for growth in this rapidly expanding market, projected to reach USD 30 billion by 2025.
Introduction
The Latin American Medtech sector is on the brink of a remarkable transformation, poised to reach an estimated USD 30 billion by 2025. This growth is not merely a statistic; it reflects a confluence of rising healthcare expenditures, an aging population, and the aftermath of the COVID-19 pandemic, which has exposed significant healthcare challenges while also unveiling new opportunities.
As major players like Medtronic and Abbott Laboratories lead the charge, countries such as Brazil and Mexico emerge as key markets ripe for investment.
However, navigating this dynamic landscape requires a deep understanding of local regulations, strategic partnerships, and innovative approaches to market access. From clinical trials to the integration of digital health technologies, the landscape is evolving rapidly, offering Medtech companies both challenges and unprecedented opportunities for growth and success.
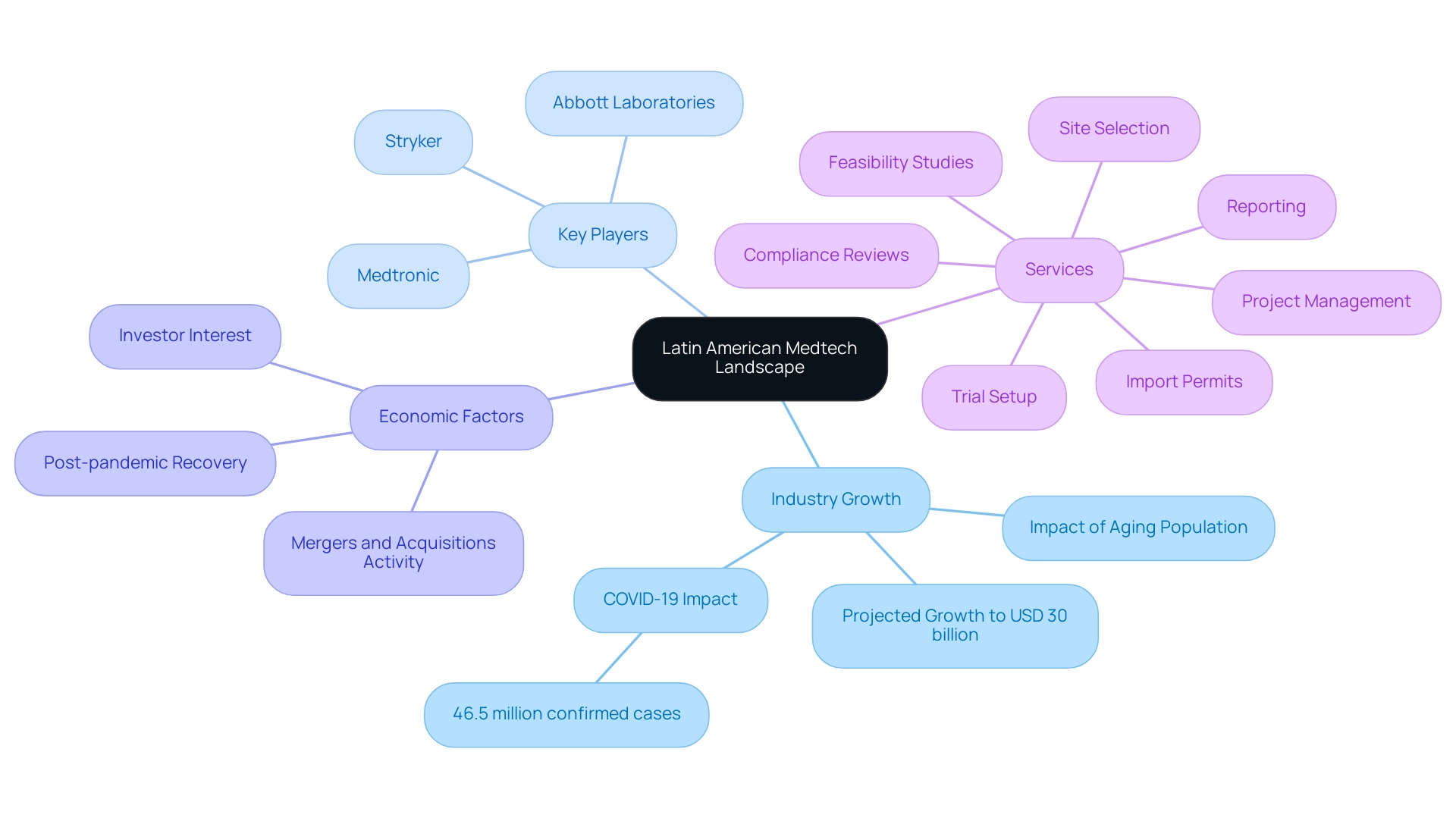
Overview of the Latin American Medtech Landscape
The Latin America medtech industry is experiencing significant growth, with projections indicating it will reach approximately USD 30 billion by 2025. This expansion is driven by rising medical expenditures, anticipated to increase due to an aging population and the prevalence of chronic diseases. Notably, the region has reported around 46.5 million confirmed COVID-19 cases, underscoring both the medical challenges and opportunities that may influence the medical technology sector.
Key players in the burgeoning Latin America medtech industry include:
- Medtronic
- Stryker
- Abbott Laboratories
Brazil and Mexico are at the forefront of this progress. As the region's economy recovers post-pandemic, there has been a surge in investor interest in medical services, fostering a favorable environment for the Latin America medtech sector. The revitalized economic landscape, combined with low competition among investors, presents substantial opportunities for growth.
A case study titled 'Latin America is Open for Business' exemplifies this trend, highlighting the region's resilience and growth, particularly within the healthcare sector. Furthermore, the Latin America medtech industry accounted for 5.4% of the global medical device contract manufacturing market in 2024, emphasizing its increasing significance in the global healthcare technology landscape.
Companies such as Welwaze Medical Inc. are seizing these opportunities by partnering with bioaccess™ for the launch of innovative medical devices like Celbrea® in Colombia. Bioaccess™ stands as a leading contract research organization (CRO) in Latin America, offering comprehensive clinical trial management services that encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
This expertise is vital for Medtech startups in the Latin America medtech industry as they navigate the intricate regulatory environment and local medical systems.
Moreover, bioaccess™'s services contribute to local economies by generating employment and fostering economic development through enhanced medical solutions. However, enterprises looking to enter this sector must develop a thorough understanding of regional medical systems. The rise of innovative technologies is transforming the industry, rendering it a dynamic space ripe for investment and development.
As Anil Kumar P., Research Manager - Healthcare, asserts, 'Whether working with small start-ups or large enterprises, I consistently deliver high-quality research that supports informed decision-making and drives impactful results in the healthcare industry.' Successful companies in the Latin America medtech industry are capitalizing on these trends to propel growth and strengthen their presence, strategically positioning themselves for future success.
Navigating Regulatory Challenges in Latin America
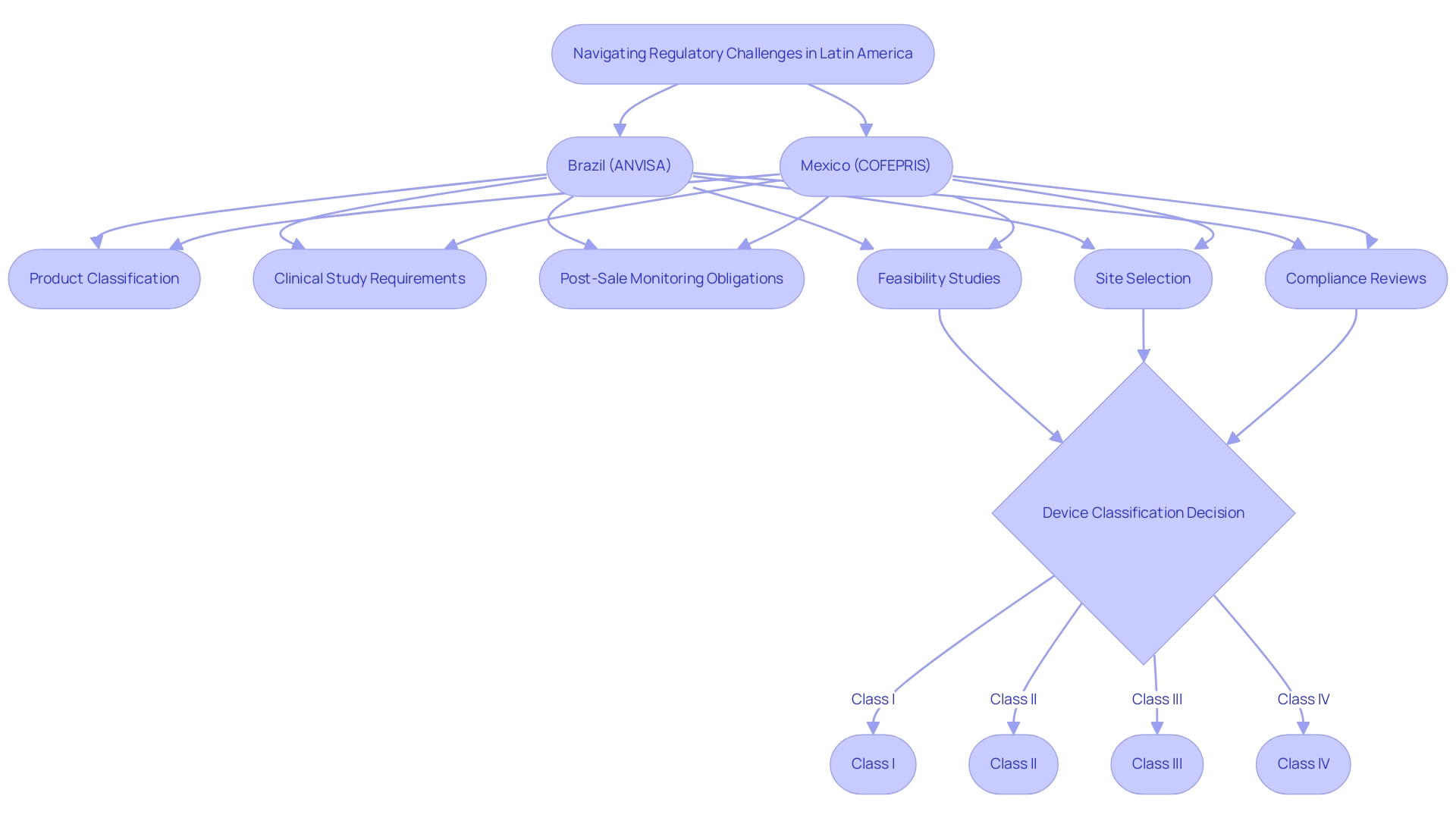
Navigating the regulatory landscape in the Latin America medtech industry presents unique challenges due to the diverse requirements across different countries. In Brazil, the National Health Surveillance Agency (ANVISA) plays a pivotal role, while in Mexico, the Federal Commission for the Protection against Sanitary Risk (COFEPRIS) governs medical device regulations. Businesses seeking to enter these sectors must perform thorough research on each nation's specific regulations, including:
- Product classification
- Clinical study requirements
- Post-sale monitoring obligations
bioaccess® offers comprehensive clinical trial management services that facilitate this process, including:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
Recent regulatory updates, such as RDC 751/2022 and RDC 830/2023, have refined the classification and approval processes for medical devices, aligning them more closely with international standards like the European Medical Device Regulation (EU) 2017/745 and the US Quality System Regulations (21 CFR part 820). Understanding these changes is crucial for manufacturers, as ANVISA has established distinct approval paths based on device classification.
For instance, Class I and II devices are subject to notification, while Class III and IV devices require comprehensive approval and testing procedures. This differentiation underscores the necessity for manufacturers to accurately determine the risk class of their devices to navigate the regulatory landscape effectively. As Margret Seidenfaden, an expert in the field, states, "Manufacturers intending to promote a medical device in Brazil must first determine the risk class of the device."
Engaging with local regulatory consultants can significantly streamline the approval process, helping to mitigate risks associated with non-compliance. These experts provide invaluable insights into the intricacies of ANVISA and COFEPRIS regulations, ensuring that companies remain informed about ongoing changes. Regular updates on regulatory developments are essential for maintaining compliance and adapting strategies in the Latin America medtech industry, ultimately facilitating a smoother entry for innovative medical devices.
The case study titled 'Approval Process Overview' illustrates the distinct approval paths established by ANVISA for different classes of devices, reinforcing the importance of understanding these classifications for timely entry.
Effective Market Access Strategies for Medtech Companies
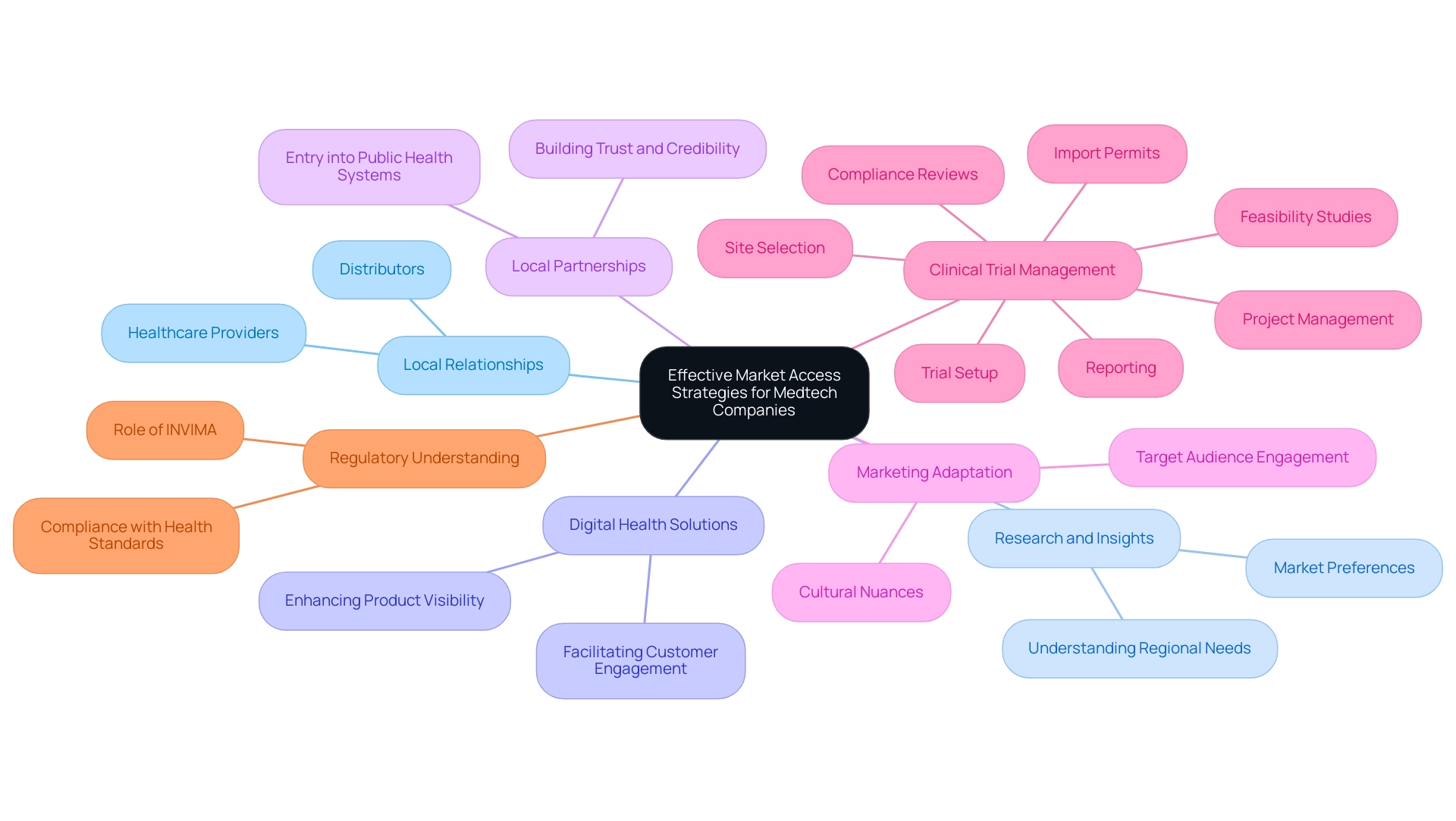
To achieve effective access in the Latin America medtech industry, companies must prioritize the establishment of robust relationships with local distributors and healthcare providers. Thorough research is essential to grasp regional needs and preferences, significantly influencing product acceptance and success. Additionally, leveraging digital health solutions can enhance product visibility and accessibility, facilitating easier engagement for potential customers with offerings.
Engaging in local partnerships not only facilitates entry into public health systems but also fosters trust and credibility within the community. Participation in industry events represents another strategic avenue, allowing companies to establish a strong brand presence and network with key stakeholders. Furthermore, adapting marketing strategies to resonate with cultural nuances is vital for enhancing engagement with target audiences.
As highlighted by industry experts, understanding the local landscape and building relationships with distributors can mitigate challenges such as logistics and regulatory hurdles. George Frazer Moreira, Founder of FRAGIA CORP., emphasizes that "with challenges ranging from logistics to import regulations, creating an efficient supply chain in Latin America can seem daunting. But with the right knowledge and strategies, these challenges can be overcome."
This insight underscores the significance of local partnerships in navigating economic dynamics effectively.
Moreover, comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting, are crucial for medical technology companies seeking success in this region. Understanding the role of INVIMA (Colombia National Food and Drug Surveillance Institute) is also essential, as it oversees the regulatory framework for medical devices, ensuring compliance with health standards and facilitating the import and export of products. INVIMA's classification as a Level 4 health authority by PAHO/WHO further emphasizes its importance in maintaining the safety, efficacy, and quality of medical devices in Colombia.
The competitive ventilators sector illustrates this point, with Draeger leading with a 17% share, followed closely by Neumovent at 16% and Medtronic at 12%. By concentrating on these strategies and comprehending the competitive landscape, medical technology firms can position themselves for success in the vibrant Latin America medtech industry. The scale of the medical technology industry is significant, as demonstrated by Abbott Laboratories' global revenue of over US$31.2 billion in 2022, highlighting the growth potential in the Latin America medtech industry.
The Role of Clinical Trials in Medtech Success
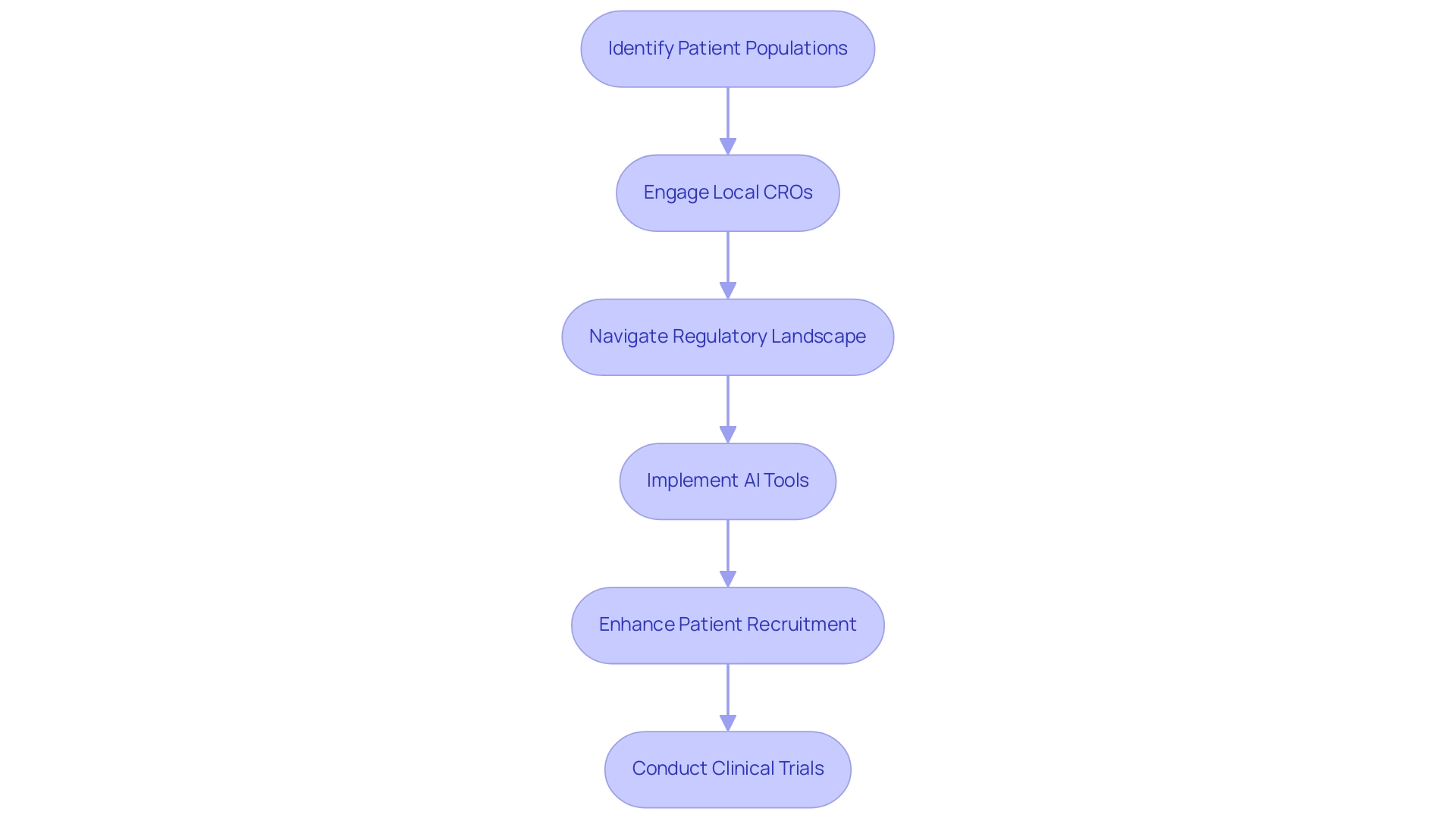
Clinical studies are crucial to the success of medical technology products, providing essential insights into safety and effectiveness. As we look to 2025, the Latin America medtech industry emerges as an optimal setting for these studies, characterized by its diverse patient populations and significantly lower operational costs compared to other regions. This distinctive environment not only enhances the representativeness of clinical data but also accelerates the recruitment process, making it an attractive choice for stakeholders in the Latin America medtech sector.
Engaging with local clinical research organizations (CROs) like bioaccess® is vital for navigating the intricate regulatory landscape and optimizing study processes. With over 20 years of experience in Medtech, bioaccess® possesses a deep understanding of regional regulations, facilitating smoother interactions with regulatory bodies such as INVIMA, Colombia's National Food and Drug Surveillance Institute. Additionally, collaborations with local healthcare organizations can substantially enhance patient recruitment initiatives and streamline data gathering, ultimately leading to more effective study execution.
The importance of adhering to ethical standards and regulatory guidelines cannot be overstated, as the integrity of clinical studies is paramount. Recent innovations in the clinical research sector, particularly AI-powered tools for management and patient recruitment, are poised to further enhance operational efficiency and participant retention. These advancements reflect a broader trend towards digital and decentralized clinical studies, which are gaining momentum in the region.
Significantly, Medable Inc. introduced Medable AI in November 2024, a generative AI solution designed to expedite digital and decentralized clinical studies, thereby improving efficiency and transparency.
As the prevalence of chronic diseases continues to rise, contributing to the expansion of the phase III segment, the demand for effective medical technology solutions becomes increasingly urgent. Long-acting formulations, such as Cabotegravir and Lenacapavir, illustrate how innovative approaches can enhance adherence and alleviate the burden of daily medication for at-risk populations. As Vivienne van der Walle, Founder and Medical Director, aptly noted, "Anything that takes away time from patients is a pain point for a site, and anyone who resolves that is helping patient care."
By leveraging the advantages of conducting clinical studies within the Latin America medtech industry, including successful case examples like ReGelTec's early feasibility analysis on HYDRAFIL™ for chronic low back pain, medical technology firms can not only expedite their product development timelines but also contribute to the advancement of medical solutions that significantly impact patient care.
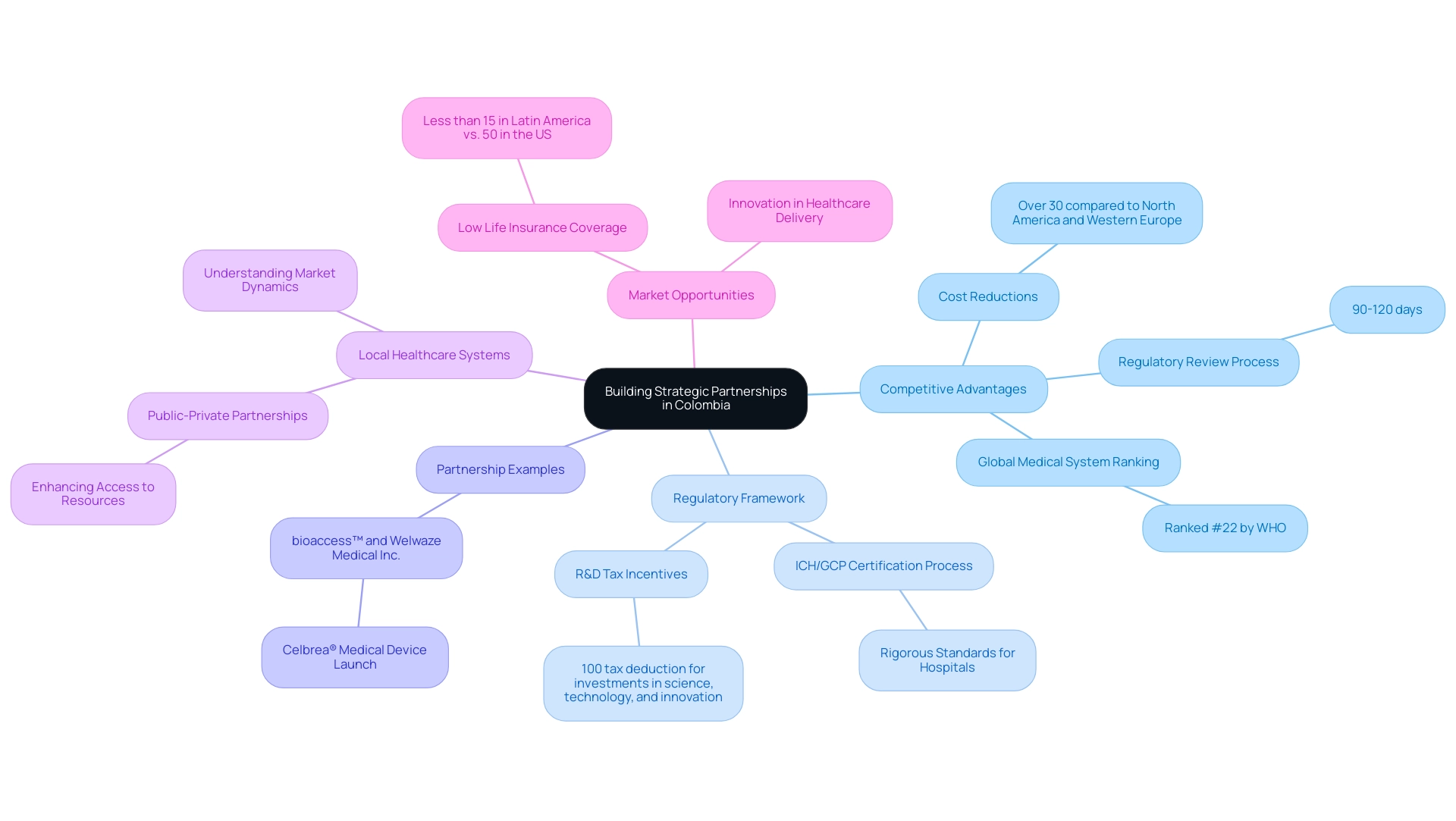
Building Strategic Partnerships and Understanding Local Healthcare Systems
Forming strategic alliances with local stakeholders—such as medical providers, distributors, and regulatory consultants—is crucial for Medtech firms seeking to enter the Latin American sector, particularly in Colombia. This nation offers notable competitive advantages for first-in-human clinical trials, including:
- Cost reductions exceeding 30% compared to North America and Western Europe
- A regulatory review process that typically requires only 90-120 days
- A medical system ranked #22 globally by the World Health Organization
Hospitals in Colombia must undergo a rigorous ICH/GCP certification process to conduct clinical research, further enhancing the country's reputation.
A nuanced understanding of these local healthcare systems, encompassing both public and private sector dynamics, enables companies to tailor their strategies effectively. Engaging in public-private partnerships can significantly enhance access to vital resources, thereby facilitating a smoother entry process. For instance, collaborations between bioaccess™ and organizations like Welwaze Medical Inc. for the Celbrea® medical device launch exemplify how strategic alliances can streamline regulatory access and entry in Colombia.
Moreover, active participation in local industry associations is essential for networking and staying abreast of market trends and regulatory developments. This engagement not only promotes teamwork but also fosters an understanding of the evolving landscape of medical services in the region.
As the Latin America medtech industry continues to expand, comprehending the complexities of local medical systems and establishing strategic partnerships will be crucial for success in 2025 and beyond. With less than 15% of the population possessing life insurance policies compared to roughly 50% in the United States, there exists a substantial opportunity for healthcare companies to address unmet needs and improve healthcare delivery through innovative solutions. As noted by a US TPS Business Development Manager, 'The response was good, and I got what I was looking for as far as the report. Thank you for that,' highlighting the importance of tailored insights in navigating this complex market.
Additionally, the region's commitment to enhancing research infrastructure and regulatory frameworks, as illustrated in the case studies of bioaccess™'s collaborations with various partners, underscores how these advancements are fostering clinical trial methodologies. This context further supports the argument for strategic partnerships and understanding local systems, reinforcing the notion that collaboration is key to success in the region. Furthermore, the R&D tax incentives available in Colombia, including a 100% tax deduction for investments in science, technology, and innovation projects, provide additional financial benefits that can enhance the market's appeal for medical technology firms.
Julio Martinez-Clark, CEO of bioaccess™, underscores these opportunities, highlighting the potential for growth in the clinical research landscape of medical technology.
Emerging Trends and Innovations in Latin American Medtech
The Latin America medtech industry is undergoing a significant transformation, propelled by the integration of digital health technologies such as telemedicine and remote monitoring solutions. These innovations not only enhance patient care but also address pressing local health challenges, including the management of chronic diseases and the needs of aging populations. As companies in the Latin America medtech sector pivot towards developing cutting-edge medical devices tailored to these specific issues, they increasingly prioritize sustainability and eco-friendly practices in their operations. By 2025, the emphasis on sustainability is expected to shape product development and business strategies within the Latin America medtech industry, compelling organizations to align their offerings with the growing consumer demand for environmentally responsible solutions.
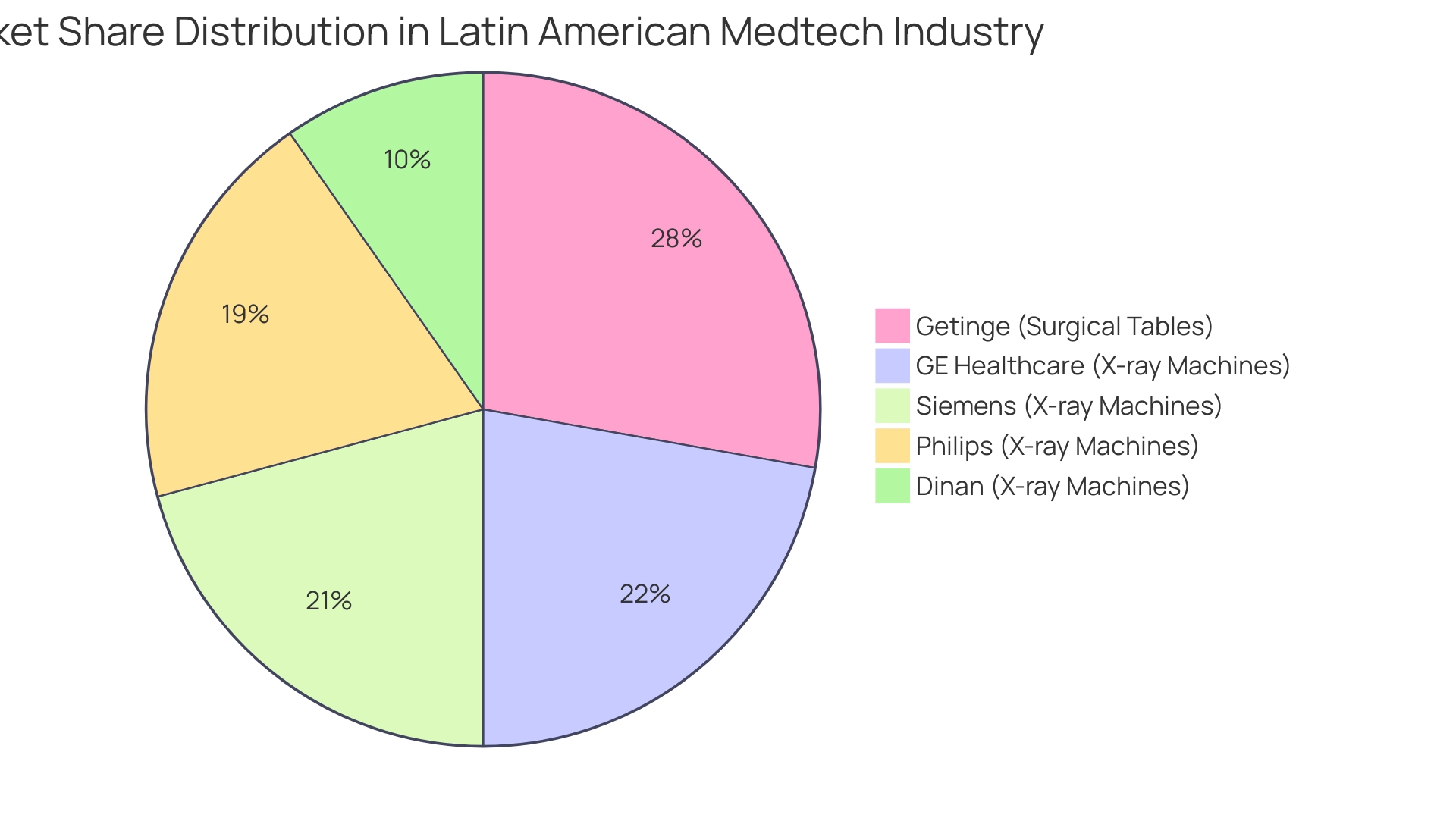
For instance, the surgical tables sector in Latin America is currently dominated by key players like Getinge, which holds a 20% market share, reflecting the competitive landscape that companies must navigate in this area. Furthermore, the fragmented X-ray machine industry, with over 70 players, showcases the potential for innovation and differentiation. GE Healthcare leads this sector with a 16% market share, followed closely by Siemens at 15%, Philips at 14%, and Dinan at 7%.
This landscape suggests ample opportunity for new participants to carve out their niche in the Latin America medtech industry by emphasizing innovative solutions that address local needs.
The influence of Medtech clinical studies extends beyond mere product development; they generate significant economic impacts by creating jobs, fostering economic growth, and enhancing health outcomes. These studies also elevate international recognition and promote increased research and development, contributing to the advancement of medical technology. As companies engage in clinical trials, they not only drive the advancement of medical technology within the Latin America medtech industry but also foster international collaboration, enhancing the overall healthcare landscape in the region. To thrive in this dynamic environment, companies must engage in continuous innovation and closely monitor emerging trends.
Participation in industry events, such as the 'Latin America is Open for Business' workshop at the Florida International Medical Expo 2023, can provide invaluable insights and networking opportunities. As George Frazer Moreira noted, such workshops offer a platform for knowledge sharing and collaboration among industry veterans, which is essential for staying ahead in this rapidly evolving market. Additionally, understanding the regulatory landscape is crucial for success. INVIMA, the Colombia National Food and Drug Surveillance Institute, plays a pivotal role in overseeing medical devices and ensuring compliance with health standards.
As a Level 4 health authority recognized by PAHO/WHO, INVIMA's functions are vital for maintaining the safety, efficacy, and quality of medical products in the region.
Conclusion
The Latin American Medtech sector is poised for remarkable growth, projected to reach USD 30 billion by 2025. This surge is propelled by increasing healthcare expenditures, an aging population, and valuable lessons learned from the COVID-19 pandemic, which have unveiled both challenges and opportunities. Major players such as Medtronic and Abbott Laboratories are strategically targeting Brazil and Mexico as prime markets for investment and innovation.
Success in this dynamic environment necessitates a profound understanding of local regulations and healthcare systems. Companies must adeptly navigate the requirements of regulatory bodies like ANVISA in Brazil and COFEPRIS in Mexico. Establishing strategic partnerships with local stakeholders, including clinical research organizations, is essential for ensuring compliance and facilitating seamless market entry.
Moreover, embracing innovative technologies and prioritizing sustainability will be crucial for product development. Companies that customize solutions to address regional health challenges will uncover significant growth opportunities.
In conclusion, the transformation of the Latin American Medtech landscape offers immense potential for those prepared to navigate its complexities. By leveraging strategic partnerships and fostering innovation, Medtech companies can thrive in this vibrant market and contribute to improved healthcare outcomes for diverse populations. The future shines brightly for those ready to seize the opportunities that lie ahead.
Frequently Asked Questions
What is the projected growth of the Latin America medtech industry by 2025?
The Latin America medtech industry is projected to reach approximately USD 30 billion by 2025.
What factors are driving the growth of the medtech industry in Latin America?
The growth is driven by rising medical expenditures, an aging population, and the prevalence of chronic diseases. The COVID-19 pandemic has also highlighted medical challenges and opportunities in the sector.
Who are the key players in the Latin America medtech industry?
Key players include Medtronic, Stryker, and Abbott Laboratories.
Which countries are leading the growth in the Latin America medtech sector?
Brazil and Mexico are at the forefront of this growth.
How has the economic recovery post-pandemic affected the medtech industry in Latin America?
The economic recovery has led to increased investor interest in medical services, creating a favorable environment for the medtech sector.
What percentage of the global medical device contract manufacturing market does the Latin America medtech industry account for?
The Latin America medtech industry accounted for 5.4% of the global medical device contract manufacturing market in 2024.
What role does bioaccess™ play in the Latin America medtech industry?
Bioaccess™ is a leading contract research organization that provides comprehensive clinical trial management services, which are essential for medtech startups navigating the regulatory environment.
What services does bioaccess™ offer for clinical trial management?
Bioaccess™ offers services including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting.
What challenges do businesses face when entering the Latin America medtech market?
Businesses must navigate diverse regulatory requirements across countries, including product classification, clinical study requirements, and post-sale monitoring obligations.
What are the key regulatory bodies in Brazil and Mexico for medical devices?
In Brazil, the National Health Surveillance Agency (ANVISA) governs medical device regulations, while in Mexico, the Federal Commission for the Protection against Sanitary Risk (COFEPRIS) is responsible.
What recent regulatory updates have impacted the approval processes for medical devices in Latin America?
Recent updates include RDC 751/2022 and RDC 830/2023, which have refined classification and approval processes to align more closely with international standards.
How do device classifications affect the regulatory approval process?
Class I and II devices require notification, while Class III and IV devices need comprehensive approval and testing procedures, making it crucial for manufacturers to accurately determine the risk class of their devices.
Why is it important for manufacturers to engage with local regulatory consultants?
Local regulatory consultants can help streamline the approval process, mitigate risks associated with non-compliance, and provide insights into ANVISA and COFEPRIS regulations.

