Overview
This article delineates the crucial steps and considerations involved in designing clinical trials for medical devices in Brazil. It underscores the significance of regulatory compliance, meticulous study design, and effective recruitment strategies. By detailing the essential phases of trial design—such as defining objectives, selecting suitable study populations, and adhering to ANVISA regulations—this article collectively enhances the credibility and success of medical device trials within the Brazilian context.
Introduction
In the complex and rapidly evolving landscape of medical device development, clinical trials play a pivotal role in ensuring safety and efficacy. These systematic investigations are not merely regulatory hurdles; they are essential for transforming innovative concepts into viable products that can improve patient outcomes.
Understanding the intricacies of clinical trial phases, from initial feasibility studies to post-market follow-ups, is crucial for researchers and stakeholders alike. As the demand for advanced medical technologies grows, so does the need for effective strategies in trial design, participant recruitment, and regulatory compliance.
This article delves into the essential elements of conducting successful clinical trials for medical devices, highlighting best practices, common challenges, and the importance of patient engagement throughout the process.
Understanding the Basics of Clinical Trials for Medical Devices
Designing clinical trials for medical devices in Brazil involves systematic investigations aimed at assessing the safety and effectiveness of a device. These experiments typically unfold in multiple stages, including pilot studies, pivotal studies, and post-market follow-up studies, each with distinct goals and methodologies. A comprehensive understanding of these stages is essential for crafting a successful trial.
Key components to consider include:
- Objectives: Clearly defining the trial's aims is paramount. Objectives may focus on safety, efficacy, or usability, guiding the overall design and execution of the research. As Berra Yazar-Klosinski emphasizes, establishing clear objectives is fundamental to the success of clinical trials.
- Study Population: Identifying the target demographic is crucial. The research group should accurately represent the intended user base of the product, ensuring that findings are applicable to real-world situations. The statistic that over 80% of participants in specific studies reported a lifetime history of suicidal thoughts underscores the importance of thorough participant screening and monitoring throughout the research phases, ensuring that the study group is well-defined and relevant.
- Endpoints: Establishing primary and secondary endpoints is vital for effectively measuring the outcomes of the research. These endpoints provide a framework for evaluating the performance and safety of the device. Understanding the regulations governing medical studies in Brazil, particularly those established by ANVISA, is critical for designing clinical trials for medical devices in the country. Adherence to these regulations ensures that the study complies with legal and ethical standards, facilitating more efficient approval processes.
Recent statistics reveal that comprehending the stages of medical research can significantly impact the success of healthcare products. The previously mentioned statistic regarding suicidal thoughts highlights the critical nature of participant screening, as it can influence the overall safety and effectiveness results of the trial.
Case analyses, such as Avantec Vascular's first-in-human trial of an innovative vascular tool in Latin America with bioaccess™ support, exemplify the practical application of these principles. Bioaccess™ not only assists in regulatory submissions but also ensures that studies are conducted with a focus on local market requirements and compliance, thereby enhancing the credibility of the results.
Moreover, bioaccess™ is committed to ensuring information security and client trust throughout the research process. Their complaint and data protection protocols are designed to address client concerns with adherence and transparency, emphasizing the importance of ethical practices in research.
In conclusion, designing clinical trials for medical devices in Brazil through a systematic approach that prioritizes objectives, participant demographics, endpoints, and regulatory compliance is vital for advancing medical devices in Latin America. This methodology not only bolsters the credibility of the findings but also accelerates the commercialization pathway. Furthermore, recent advancements in statistical evaluation and assumptions related to medical studies highlight the complexities inherent in this process.
Navigating Brazil's Regulatory Framework for Medical Device Trials
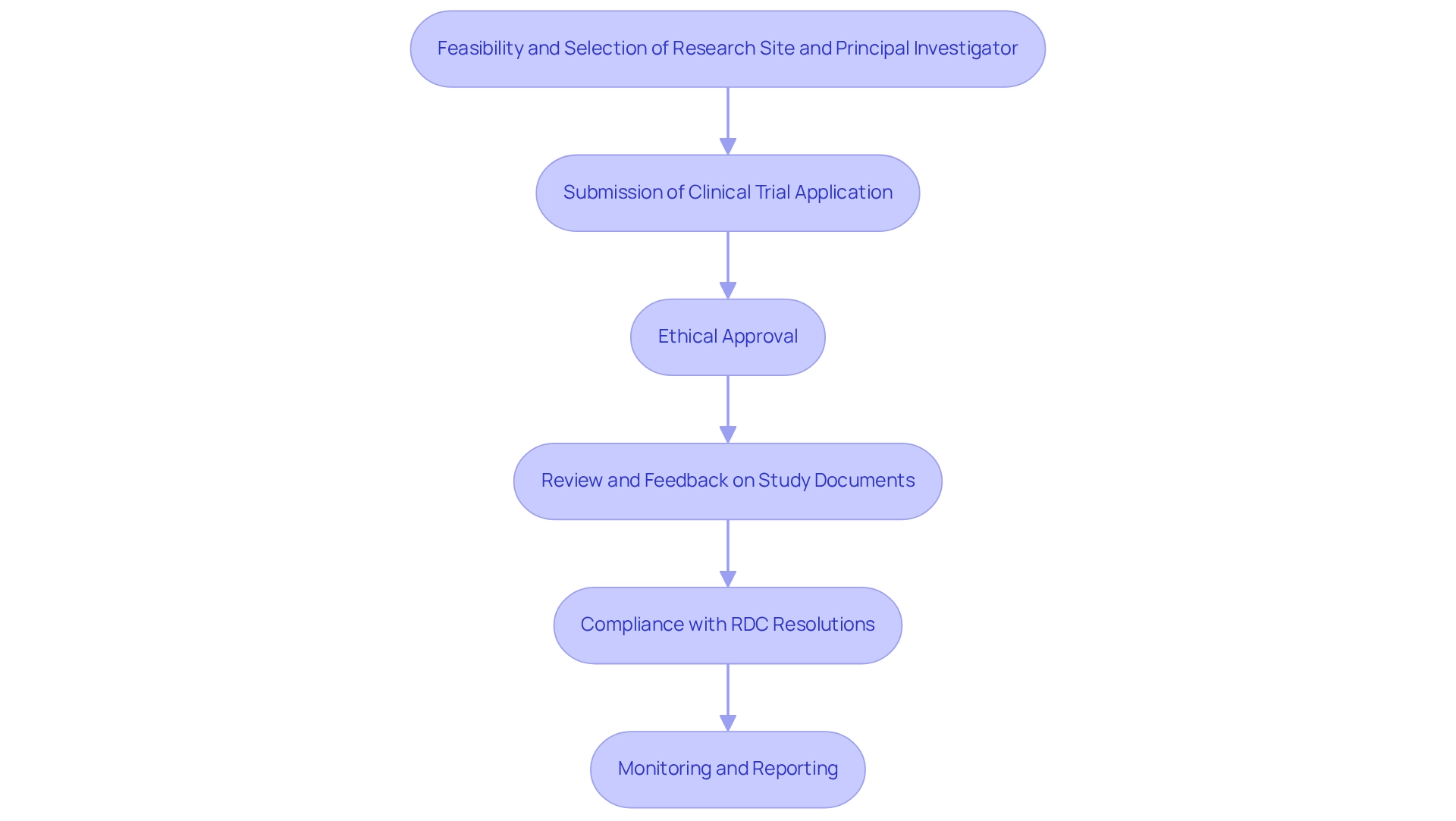
Designing clinical trials for medical devices in Brazil necessitates strict compliance with the regulations set by ANVISA (Agencia Nacional de Vigilância Sanitária). The subsequent key steps delineate the thorough process for progressing medical device studies:
- Feasibility and Selection of Research Site and Principal Investigator (PI): Start by evaluating potential research locations and choosing qualified principal investigators. This step is vital for guaranteeing that the examination is carried out in a suitable setting with skilled personnel.
- Submission of Clinical Trial Application (CTA): Prepare a detailed Clinical Trial Application that includes the research protocol, informed consent forms, and qualifications of the investigators involved. This submission is crucial for initiating the regulatory review process.
- Ethical Approval: Securing approval from local ethics committees is essential. These committees assess the ethical implications of the experiment, ensuring that participant safety and welfare are prioritized throughout the study.
- Review and Feedback on Study Documents: It is vital to review and provide feedback on study documents to comply with country requirements, ensuring that all necessary information is accurately presented.
- Compliance with RDC Resolutions: Familiarize yourself with pertinent RDC resolutions, particularly RDC No. 10/2015, which outlines the specifications for research involving medical devices. Grasping these regulations will aid in simplifying the approval procedure and guaranteeing adherence.
- Monitoring and Reporting: Create a strong system for tracking the progress of the test. This encompasses prompt reporting of any negative occurrences to ANVISA, which is required to ensure participant safety and preserve the integrity of the study. bioaccess® provides extensive management services for studies, including project oversight and reporting, to guarantee that all elements of the study are carefully managed.
In 2025, ANVISA continues refining its regulations for studies, highlighting the significance of transparency and participant representation. For instance, the inclusion of Research Participant Representatives (RPPs) in ethics committees has been shown to enhance the ethical review process by ensuring that the perspectives of diverse participant groups are considered. RPPs are necessary to possess a background of community engagement, which enhances their function in promoting trust and accountability in research.
Moreover, recent data shows that the yearly monitoring report for protocols must be submitted within 60 days of the study's anniversary date, emphasizing the significance of prompt adherence. Payments for the TFVS fee can be processed through different methods, including bank transfers and online platforms, which are essential logistical elements of carrying out research activities in Brazil. Grasping these rules and procedures is essential for effectively navigating the research landscape, particularly in the context of designing clinical trials for medical devices in Brazil.
Furthermore, ANVISA has set particular standards for Equivalent Foreign Regulatory Authorities (AREEs), which further clarifies the regulatory environment for medical product evaluations. By utilizing bioaccess®'s knowledge in overseeing research studies, you can effectively tackle the obstacles encountered by medical technology startups, particularly in designing clinical trials for medical devices in Brazil, ensuring a more streamlined route to positive study results. bioaccess® is dedicated to data protection and has set up grievance procedures to handle client issues, ensuring adherence and openness throughout the research process.
Choosing the Right Study Design for Your Clinical Trial
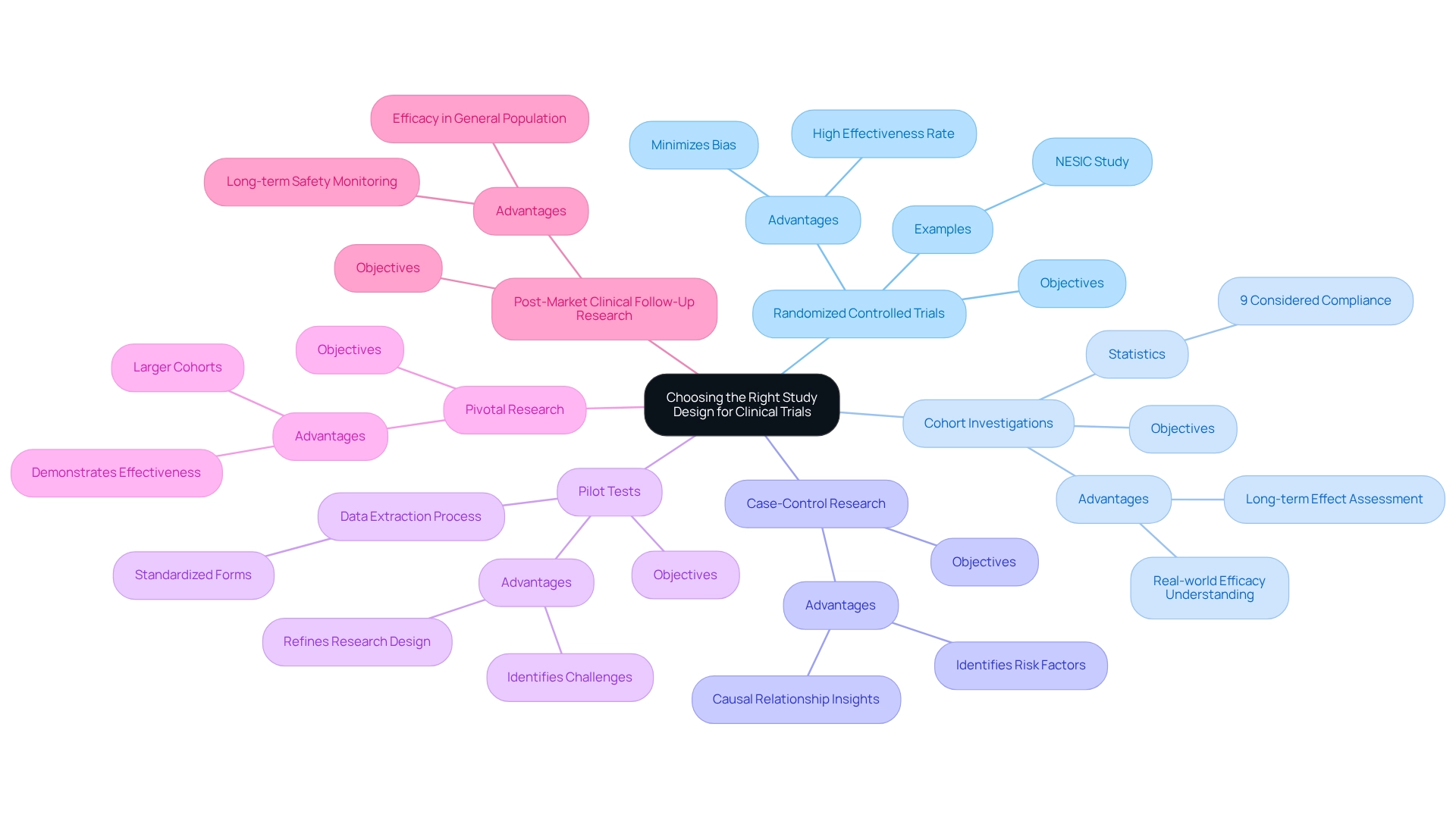
Selecting the appropriate research framework is crucial for successfully designing clinical trials for medical devices in Brazil, particularly in medical evaluations involving health instruments. This decision should be guided by the specific objectives of the trial and the characteristics of the item being assessed. Below are some common research designs utilized in this domain:
- Randomized Controlled Trials (RCTs): RCTs are widely recognized as the gold standard in medical research. By randomly assigning participants to either treatment or control groups, RCTs effectively minimize bias, ensuring that the results are attributable to the intervention rather than confounding variables. Recent statistics indicate that RCTs demonstrate a high effectiveness rate in medical equipment research, with a significant portion yielding reliable outcomes. As noted by F.F., the idea of conducting a review on this topic emerged while drafting the Statistical Analysis Plan for the NESIC research, underscoring the importance of robust research designs in clinical trials.
- Cohort Investigations: These observational studies track a group of participants over time, allowing researchers to assess the long-term effects associated with the use of a medical instrument. Cohort analyses are particularly beneficial for understanding the real-world efficacy of instruments, as they can compile data on diverse patient demographics and instrument applications. Notably, a review found that only 9% of studies considered compliance as a factor in their analysis, highlighting the need for more comprehensive data collection in cohort research to ensure strong findings.
- Case-Control Research: Ideal for investigating rare outcomes, case-control studies compare individuals who have experienced a specific outcome with those who have not, examining their prior exposure to the medical instrument. This design is particularly useful in identifying potential risk factors and understanding the causal relationships between device usage and outcomes.
- Pilot Tests: Conducting a pilot test serves as an initial phase to refine the research design and identify potential challenges before embarking on a larger-scale investigation. These smaller studies can provide critical insights into feasibility, recruitment strategies, and data collection processes, ultimately enhancing the robustness of the main study. Incorporating a structured data extraction procedure, as demonstrated in a recent case analysis, can further bolster the reliability of results. This process involved creating standardized forms to gather essential characteristics of the experiment, including equipment types and statistical methods employed, ensuring a comprehensive understanding of compliance management in equipment evaluations.
- Pivotal Research: These evaluations are essential for demonstrating the effectiveness and safety of a medical device prior to its market approval. They typically involve larger cohorts and aim to provide conclusive evidence of the instrument's performance.
- Post-Market Clinical Follow-Up Research (PMCF): These studies are conducted after a device has been authorized and are crucial for monitoring long-term safety and efficacy in the general population.
At bioaccess®, we excel in managing these types of research, leveraging over 20 years of experience in the Medtech sector. Our comprehensive management services for research encompass feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting. The FDA's recent emphasis on the use of real-world evidence to support regulatory decision-making for medical devices further underscores the importance of selecting appropriate research designs.
By thoroughly evaluating these research frameworks and their respective advantages, investigators can adeptly navigate the complexities involved in designing clinical trials for medical devices in Brazil.
Effective Recruitment Strategies for Clinical Trials in Brazil
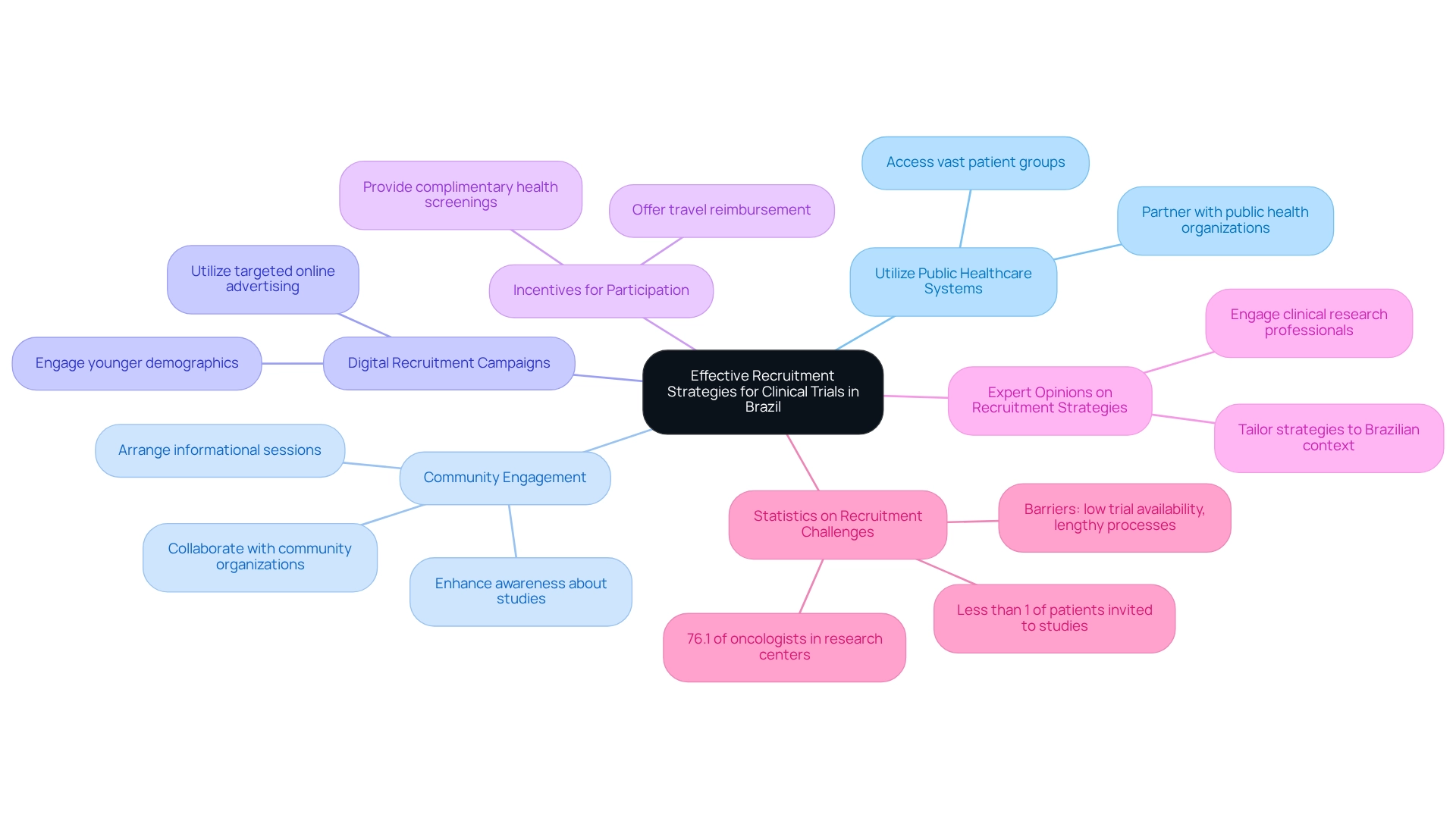
To successfully recruit individuals for medical studies in Brazil, several strategic methods can be utilized:
-
Utilize Public Healthcare Systems: Brazil's extensive public healthcare network serves as a crucial resource for reaching potential participants. By partnering with public health organizations, researchers can access a vast group of patients who may gain from clinical studies, especially those overseen by bioaccess®, which focuses on managing the intricacies of clinical investigations, including Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH).
-
Community Engagement: Establishing connections with local communities is essential. Arranging informational sessions and collaborating with community organizations can greatly enhance awareness about the proceedings, building trust and promoting involvement. A recent survey among Brazilian oncologists emphasized that despite a high occurrence of cancer, many patients were unaware of available studies, indicating a need for improved community outreach. Engaging with bioaccess® can also offer insights into how research contributes to local economies, including job creation and healthcare advancements.
-
Digital Recruitment Campaigns: Utilizing targeted online advertising and social media platforms can broaden the reach of recruitment efforts. These campaigns can effectively engage younger demographics and those who may not be reached through traditional methods, aligning with bioaccess®'s innovative strategies for managing research studies.
-
Incentives for Participation: Offering incentives, such as travel reimbursement or complimentary health screenings, can motivate individuals to take part in studies. This method not only improves recruitment rates but also shows a dedication to participant well-being, a principle that bioaccess® maintains in its clinical research services.
-
Expert Opinions on Recruitment Strategies: Engaging with clinical research professionals can provide valuable insights into effective recruitment strategies tailored to the Brazilian context. Their experience can steer the creation of focused strategies that connect with potential participants, particularly in studies conducted by bioaccess®.
-
Statistics on Recruitment Challenges: Data shows that while 76.1% of oncologists in Brazil operate in research centers, many invite less than 1% of their patients to take part in studies. This statistic emphasizes the significance of tackling obstacles like limited study availability and prolonged regulatory procedures to enhance recruitment results. Additionally, the results from a survey of Brazilian oncologists indicate that better physician involvement and patient education are essential elements in boosting recruitment rates, as many oncologists at present do not actively encourage their patients to take part in studies. By utilizing the knowledge of bioaccess®, which has a proven history in Medtech, researchers can more effectively tackle these challenges and improve recruitment initiatives. By applying these strategies, researchers can boost participant enrollment for studies in Brazil, ultimately promoting the advancement of medical devices and enhancing patient outcomes.
Incorporating Patient-Reported Measures in Trial Design
Incorporating patient-reported outcomes (PROs) into clinical study design is essential for gaining valuable insights into the patient's experience. Key considerations for effective integration include the following:
- Defining PROs: Clearly defining the specific patient-reported outcomes to be measured, such as quality of life, symptom relief, or treatment satisfaction, is crucial. This clarity aligns the study's objectives with patient needs.
- Selecting Appropriate Instruments: Choosing validated instruments for measuring PROs is vital to ensure the reliability and validity of the data collected. Utilizing established tools enhances the credibility of the findings and supports regulatory submissions.
- Timing of Assessments: Determining the optimal timing for collecting PRO data during the trial is important to capture relevant changes in patient status. Evaluations should be strategically arranged to demonstrate the influence of the medical instrument over time.
- Data Analysis: Organizing the examination of PRO data alongside health outcomes offers a thorough perspective on the impact of the product. This dual analysis allows for a more nuanced understanding of how the device affects patients, beyond traditional clinical metrics.
A recent case study titled 'Visualization of PRO Data' investigated how PRO data was presented in research reports, revealing that 80% included figures or tables to effectively convey results. However, inconsistencies in labeling and clarity highlighted the need for standardized practices in future publications. Moreover, statistics show that 65.7% of study endpoints were categorized as secondary endpoints, underscoring the significance of incorporating PROs into the overall study framework.
As researcher AM noted, defining PROs in study design is crucial for capturing the patient experience accurately. Furthermore, nine tables displayed descriptive summaries of PRO scores, demonstrating the practical use of PRO data in medical studies. By concentrating on these factors, medical studies can more accurately represent the patient experience, ultimately resulting in enhanced medical product development and improved patient outcomes.
Data Management and Analysis: Ensuring Compliance and Accuracy
To ensure compliance and accuracy in data management and analysis during clinical trials for medical devices, it is essential to adhere to the following best practices:
- Implement a Clinical Data Management System (CDMS): A robust CDMS is crucial for streamlining the processes of data collection, storage, and analysis. This system enhances efficiency and minimizes the risk of errors, ultimately leading to more reliable outcomes. Organizations overseeing Phase 3-4 research emphasize drug safety and real-world evidence through registries and observational studies, highlighting the significance of effective data management practices. bioaccess® offers comprehensive support in this area, facilitating the setup and management of CDMS tailored to the specific needs of Latin American markets.
- Establish Data Validation Protocols: Developing comprehensive protocols for data validation is vital to ensure the accuracy and completeness of the data collected. These protocols should include systematic checks and balances to verify that the data meets predefined quality standards, which is particularly important in navigating the regulatory landscape of Latin America.
- Conduct Regular Audits: Regular audits of data management processes are necessary to identify and rectify discrepancies. These audits help maintain the integrity of the data and ensure compliance with established protocols, addressing challenges faced by medical device startups in Brazil when designing clinical trials for medical devices and ensuring data reliability and regulatory adherence. Designing clinical trials for medical devices in Brazil requires that all data management practices ensure compliance with regulatory standards, including those set forth by ANVISA. Following these standards not only guarantees legal compliance but also boosts the reliability of the research outcomes. As Peter Benton, President and CEO, noted, "After working with Worldwide Clinical Trials for many years, it’s easy to see why they consistently perform so well in the ISR Phase II/III benchmarking report, especially in the areas of Overall Performance and Project Manager Quality."
- Feasibility and Selection of Research Locations: bioaccess® aids in the feasibility and selection of research sites and principal investigators (PIs), ensuring that studies are conducted in optimal environments for success.
- Study Arrangement and Project Oversight: bioaccess® offers extensive assistance for study setup, initiation, and project management, guaranteeing that all elements of the research process are effectively managed.
- Grievance and Data Protection Procedures: bioaccess® is committed to ensuring information security and client trust, with established grievance and data protection procedures to address any concerns with compliance and transparency.
By adopting these optimal strategies, organizations can greatly enhance the quality of their healthcare data management, which is crucial when designing clinical trials for medical devices in Brazil, resulting in more favorable outcomes. Moreover, utilizing technological assistance, like the 1,500+ applications provided by ServiceNow, can further improve the efficiency and effectiveness of data management systems.
Conducting Post-Market Clinical Follow-Up Studies
Post-market clinical follow-up investigations (PMCF) are vital for gathering information on the long-term efficacy and safety of medical instruments. These investigations not only assist in ensuring compliance with regulatory standards but also play a significant role in enhancing patient safety and device efficiency. Key steps in designing effective PMCF studies include:
-
Define Objectives: Establish clear and measurable objectives for the post-market study, emphasizing both safety and effectiveness. This clarity aligns the study with regulatory expectations and market needs.
-
Data Collection Methods: Employ diverse data collection methods to ensure comprehensive data gathering. Effective techniques include:
- Surveys: Utilize structured questionnaires to gather patient feedback on device performance and satisfaction.
- Patient Registries: Establish registries to monitor long-term results and complications related to the apparatus.
- Electronic Health Records (EHRs): Utilize EHRs to obtain real-world information on patient outcomes and equipment usage trends.
-
Regulatory Reporting: Adhere to regulatory reporting requirements by ensuring timely submission of findings to ANVISA. This includes documenting the analysis of PMCF studies in the PMCF evaluation report, integral to the Clinical Evaluation Report (CER). The necessity of this documentation underscores the regulatory requirements and the importance of thorough record-keeping.
-
Continuous Monitoring: Establish a robust system for ongoing oversight of equipment performance and patient outcomes. This proactive strategy enables the early detection of any emerging safety issues, thereby improving patient safety and equipment reliability.
The goals of PMCF studies are diverse, focusing on collecting evidence to support safety and performance assertions. A careful selection of PMCF activities tailored to the specific context of each medical product is crucial. For example, elements such as classification of equipment, sales levels, and intended use should guide the selection of activities, ensuring that they effectively gather the necessary clinical evidence.
This approach is emphasized in the case analysis titled 'Selecting the Right PMCF Activities,' which highlights the importance of tailoring PMCF activities to the unique context of each medical device.
Expert insights, including those from Jasper, who possesses 20 years of experience in the MedTech sector, underscore the importance of these factors in designing clinical trials for medical devices in Brazil. Additionally, one of the essential quality requirements for both research investigations and PMCF under the Medical Device Regulation (MDR) is adherence to Good Clinical Practice (GCP), as noted by Jon Bergsteinsson. This compliance not only ensures the integrity of the data collected but also fosters trust among stakeholders in the MedTech industry.
Furthermore, entities such as bioaccess® provide extensive assistance in the design, execution, recruitment, and evaluation of PMCF surveys, highlighting their significance in upholding regulatory compliance and enhancing the practical relevance of PMCF projects. Bioaccess® specializes in managing various types of studies, including Early-Feasibility Studies, First-In-Human Studies, and more, ensuring that clients receive customized solutions that address their specific needs.
Addressing Common Challenges in Clinical Trials for Medical Devices
Common challenges in medical device studies encompass several critical areas:
- Patient Recruitment: Low recruitment rates pose a significant barrier to study progress. Research indicates that single-site randomized controlled trials (RCTs) often achieve higher recruitment success compared to multicenter trials. Understanding the factors influencing recruitment success is crucial for developing effective solutions to participant accrual challenges in RCTs. Strategies such as community engagement and digital outreach are essential. Utilizing diverse methods, including partnerships with local organizations and targeted digital advertising, can effectively identify and attract suitable participants. Notably, GlobalCare Clinical Trials, in partnership with bioaccess™, has achieved over a 50% reduction in recruitment time and a retention rate exceeding 95% in their ambulatory services in Colombia, showcasing the effectiveness of strategic recruitment efforts. Furthermore, practical measures for enhancing recruitment strategies can be obtained from recent studies, highlighting the significance of government funding and compensation in boosting recruitment efforts.
- Regulatory Compliance: Designing clinical trials for medical devices in Brazil requires navigating the intricate landscape of regulations, particularly those established by ANVISA, which can be daunting for researchers. Staying abreast of regulatory changes is crucial for ensuring compliance and avoiding delays. Engaging with regulatory experts, such as Katherine Ruiz, who specializes in regulatory affairs for medical devices and in vitro diagnostics in Colombia, can facilitate smoother navigation through the compliance process. As noted by T. Walpole, collaboration with experienced professionals can significantly improve the quality of compliance strategies. Bioaccess™ provides extensive management services for studies that include regulatory assistance, ensuring that they meet all essential compliance standards.
- Data Management: Preserving data integrity and adherence throughout the study is a complicated task. Implementing robust data management systems, coupled with regular audits, can significantly mitigate risks associated with data handling. This method not only guarantees adherence but also improves the overall quality of the study data.
- Budget Constraints: Financial limitations can significantly affect the implementation of research. Careful budgeting is essential, and seeking additional funding sources can alleviate financial pressures. Exploring partnerships with academic institutions or government grants may provide the necessary financial support for designing clinical trials for medical devices in Brazil, which is essential to ensure successful trial completion. Addressing these challenges proactively in designing clinical trials for medical devices in Brazil can lead to more efficient and effective outcomes, ultimately accelerating the development and commercialization of innovative medical devices. Notably, statistics indicate that patient satisfaction can be adversely affected by wait times, with an average wait time of just over 34 minutes leading to lower satisfaction levels. This highlights the importance of optimizing recruitment processes to enhance participant experience and retention.
Conclusion
A comprehensive understanding of clinical trials is essential for advancing medical device development and ensuring that innovative solutions reach the market safely and effectively. Key elements such as clearly defined objectives, appropriate study designs, and robust participant recruitment strategies are vital for conducting successful trials. Moreover, navigating Brazil’s regulatory landscape requires meticulous attention to compliance with ANVISA guidelines, emphasizing the importance of ethical practices and patient safety throughout the process.
Incorporating patient-reported outcomes into trial designs enriches the data collected, providing valuable insights into the patient experience that traditional metrics may overlook. Additionally, effective data management and analysis are crucial for maintaining the integrity of trial results, ensuring that findings are both reliable and actionable.
By addressing common challenges such as patient recruitment, regulatory compliance, and budget constraints proactively, researchers can enhance the efficiency and effectiveness of clinical trials. Ultimately, the commitment to ethical practices, transparency, and patient engagement not only fosters trust in the research process but also enhances the potential for medical devices to improve patient outcomes. As the landscape of medical technology continues to evolve, the importance of rigorous clinical trials cannot be overstated; they remain the cornerstone of innovation in healthcare.
Frequently Asked Questions
What is the purpose of designing clinical trials for medical devices in Brazil?
The purpose is to systematically investigate the safety and effectiveness of a medical device through various stages, including pilot studies, pivotal studies, and post-market follow-up studies.
What are the key components to consider when designing clinical trials?
Key components include defining clear objectives, identifying the study population, and establishing primary and secondary endpoints, all of which guide the trial's design and execution.
Why is it important to define clear objectives for a clinical trial?
Clear objectives are fundamental to the success of clinical trials as they focus the research on safety, efficacy, or usability, guiding the overall design and execution.
How does the study population impact clinical trials?
The study population must accurately represent the intended user base of the product to ensure that findings are applicable to real-world situations, emphasizing the importance of thorough participant screening.
What role do endpoints play in clinical trials?
Endpoints provide a framework for measuring the outcomes of the research, allowing for effective evaluation of the device’s performance and safety.
What regulations must be followed when designing clinical trials in Brazil?
Trials must comply with regulations set by ANVISA (Agencia Nacional de Vigilância Sanitária), which ensures legal and ethical standards are met.
What are the key steps in the clinical trial process for medical devices in Brazil?
Key steps include feasibility assessments, submission of Clinical Trial Applications (CTA), obtaining ethical approval, reviewing study documents, ensuring compliance with RDC resolutions, and monitoring and reporting progress.
What is the significance of ethical approval in clinical trials?
Ethical approval from local committees is essential to ensure participant safety and welfare throughout the study.
What is the role of Research Participant Representatives (RPPs) in ethics committees?
RPPs enhance the ethical review process by representing diverse participant perspectives, promoting trust and accountability in research.
How does bioaccess™ support the clinical trial process?
Bioaccess™ provides management services that include project oversight, reporting, and ensuring compliance with local market requirements and regulations, thereby enhancing the credibility of the results.
What is the importance of monitoring and reporting in clinical trials?
Monitoring and reporting are crucial for tracking the trial's progress and ensuring participant safety, including prompt reporting of any adverse events to ANVISA.
How do recent advancements in statistical evaluation impact clinical trials?
Advancements highlight the complexities in medical research, emphasizing the need for a systematic approach to improve the success of healthcare products.




