Overview
This article addresses the critical components and optimal practices for designing clinical trials specifically for medical devices, highlighting the necessity of systematic investigation to guarantee both safety and efficacy. It delineates the fundamental phases of clinical trials, strategic planning steps, regulatory requirements, and emerging trends, demonstrating how these elements play a pivotal role in achieving successful research outcomes and enhancing patient care.
Introduction
In the rapidly evolving world of medical technology, clinical trials are the cornerstone for ensuring the safety and efficacy of new medical devices. These systematic investigations not only inform regulatory decisions but also play a pivotal role in enhancing patient care. From the initial preclinical studies to the intricate phases of human trials and post-market surveillance, understanding the multifaceted nature of clinical trials is crucial for researchers and stakeholders alike.
As the landscape of medical device research continues to transform, innovative strategies and regulatory compliance become paramount in navigating the complexities of clinical trials. This article delves into the essential components of clinical trials, offering insights into effective planning, design considerations, and emerging trends that are shaping the future of medical device research.
Understanding Clinical Trials: A Foundation for Medical Device Research
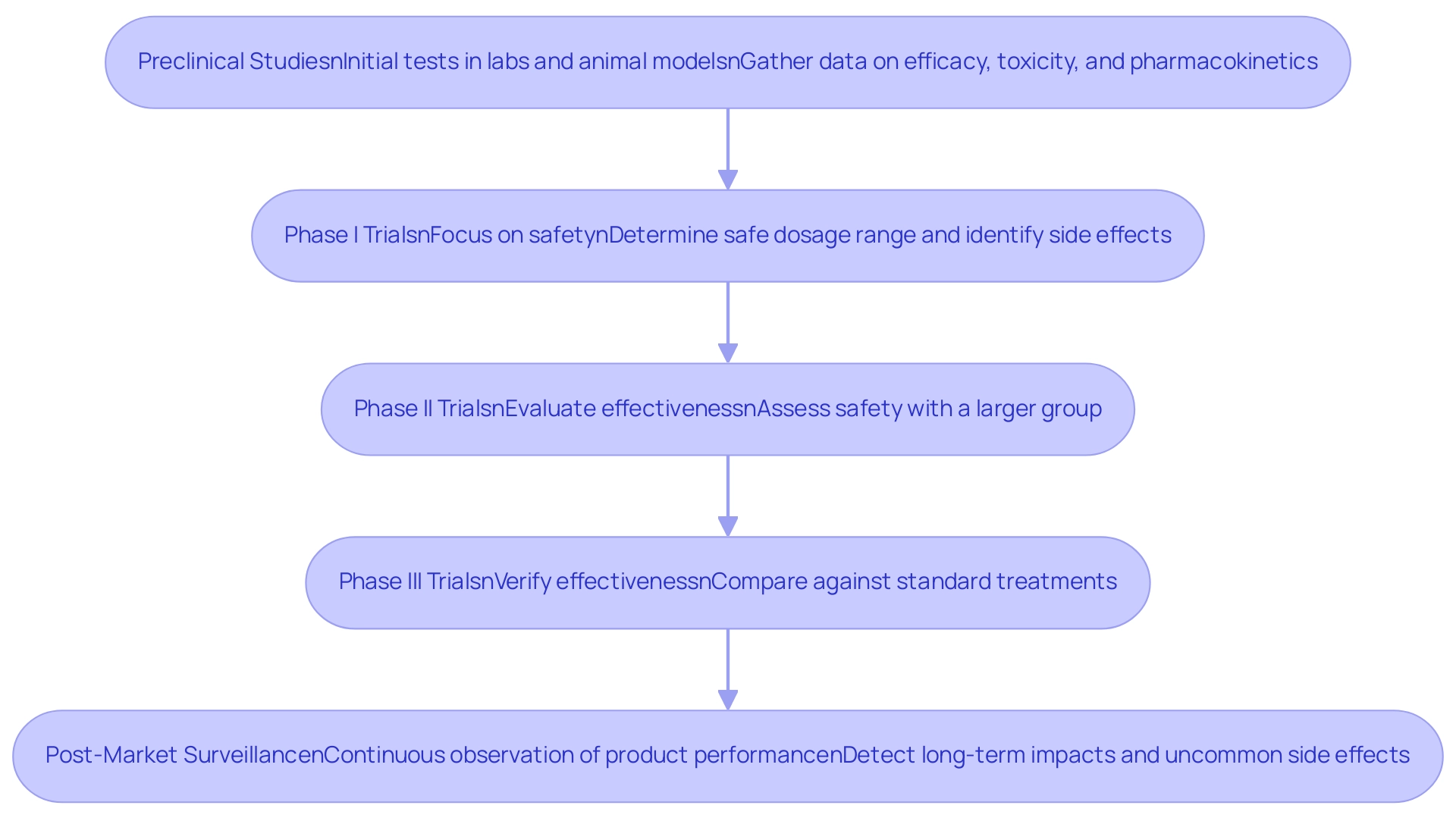
Designing clinical trials for medical devices entails systematic investigations aimed at evaluating their safety and efficacy through human participants. These experiments are crucial for gathering information that informs regulatory decisions and medical practices, ultimately enhancing patient care. The key phases of clinical trials include:
- Preclinical Studies: Initial tests conducted in laboratories and animal models gather preliminary data on efficacy, toxicity, and pharmacokinetics, laying the groundwork for human trials.
- Phase I Trials: This phase primarily focuses on safety, determining the safe dosage range and identifying potential side effects. It typically involves a small group of participants to assess how the equipment interacts with the human body.
- Phase II Trials: Here, the effectiveness of the apparatus is further evaluated, with a larger participant group continuing to assess safety while beginning to gauge therapeutic efficacy.
- Phase III Trials: This essential phase verifies the effectiveness of the apparatus, observes side effects, and compares the apparatus against standard or similar treatments, often involving varied populations to ensure comprehensive information.
- Post-Market Surveillance: Following approval, continuous observation of the product's performance in the general population is conducted to detect any long-term impacts or uncommon side effects that may not have been apparent in previous studies.
Understanding these phases is vital for researchers involved in designing clinical trials for medical devices that comply with regulatory requirements and ultimately improve patient outcomes. Recent statistics reveal that automated information mining has effectively matched around 84% of medical studies to specific diseases or conditions, underscoring the significance of focused research in the medical device sector. Furthermore, innovations aimed at enhancing site experiences in research trials have demonstrated that addressing pain points, such as prolonged entry processes, can significantly improve operational efficiency and patient care.
As noted by a Senior Director within a global biopharma’s research sciences and study management group, "Eliminating one 20-minute task per visit across 130,000 visits avoids 43,000 hours of work." This allows researchers to focus on what truly matters. A notable case study highlighted how companies like Alcon have streamlined site performance monitoring, leading to more user-friendly systems and better data entry efficiency.
In Latin America, bioaccess® provides accelerated medical device research study services, prominently featuring the design of clinical trials for medical devices, while leveraging over 20 years of expertise in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF). Their extensive research management services encompass feasibility studies, site selection, compliance evaluations, setup, import permits, project oversight, and reporting. This customized approach not only enhances the effectiveness of medical studies but also supports local economies through job creation and healthcare improvements.
As the landscape of medical studies evolves, remaining knowledgeable about these stages and their implications is essential for promoting medical equipment research and development.
Strategic Planning in Clinical Trials: From Concept to Execution
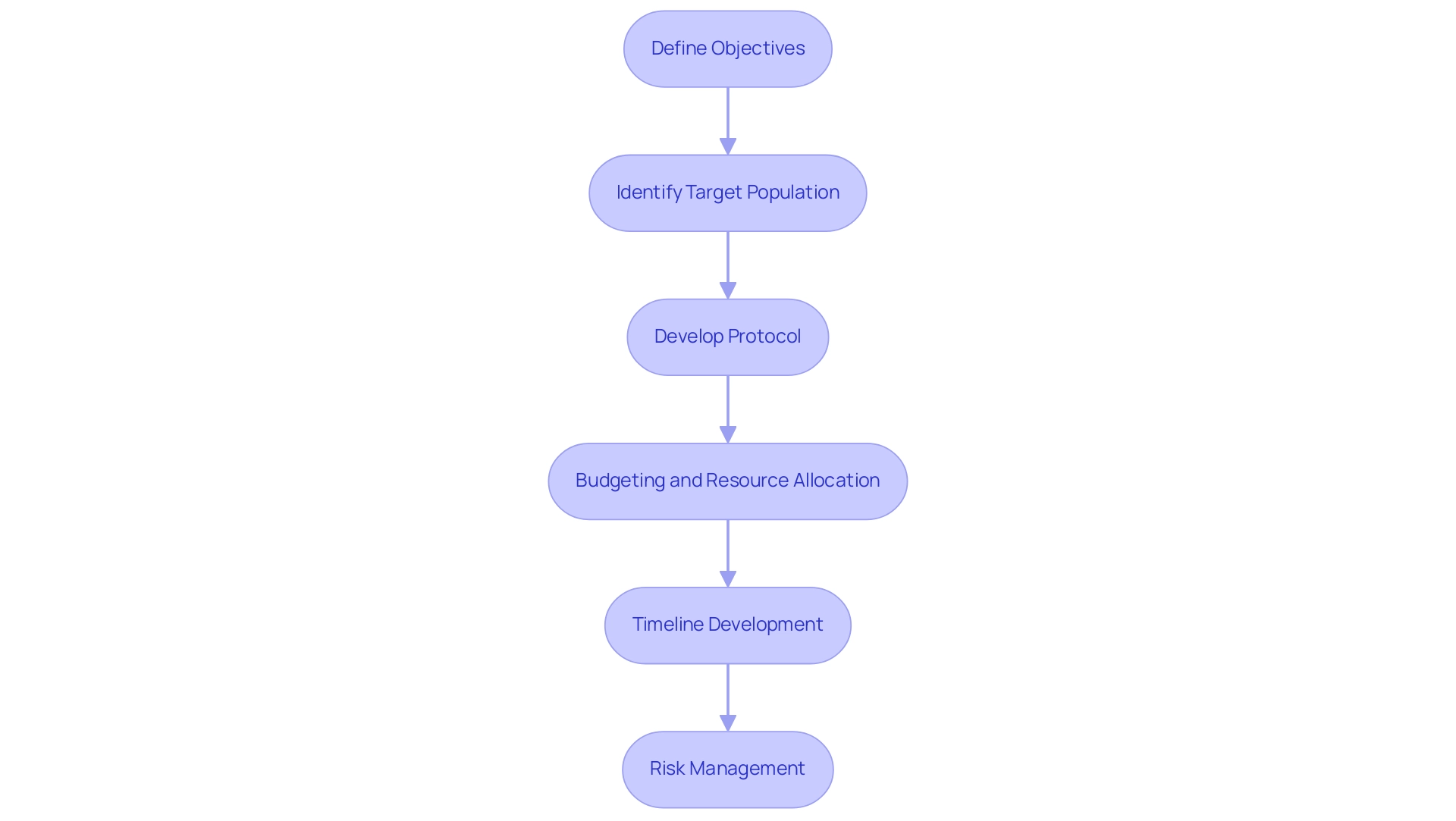
Effective strategic planning is essential for the success of designing clinical trials for medical devices, particularly in the medical device sector. The subsequent key steps outline best practices for optimizing study design in 2025:
- Define Objectives: Clearly articulate both primary and secondary goals of the experiment. This clarity not only guides the study but also aligns stakeholders on expected outcomes.
- Identify Target Population: Determine the specific characteristics of participants to be included in the study. A well-defined target population enhances the relevance and applicability of study results.
- Develop Protocol: Create a comprehensive study protocol that details the methodology, including inclusion and exclusion criteria, endpoints, and statistical analysis plans. A robust protocol acts as a roadmap for the study, ensuring consistency and adherence to regulatory standards.
- Budgeting and Resource Allocation: Accurately estimate costs and allocate resources to ensure the project remains within budget. Statistics indicate that effective budgeting can significantly influence feasibility and success rates, with strategic planning being a key factor in managing financial resources efficiently. Notably, long-term agreements with provider partners typically last 20 years, highlighting the stability and reliability of partnerships in clinical research. Companies like bioaccess® utilize their 20+ years of expertise in Medtech to offer personalized solutions that improve budgeting and resource management, ensuring that each study is customized to meet specific needs.
- Timeline Development: Establish a realistic timeline for each phase of the study, encompassing recruitment, data collection, and analysis. Timelines should be flexible yet structured to accommodate potential delays, thereby enhancing overall project management. bioaccess® specializes in accelerated medical device research study services, ensuring timely execution of Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Research Follow-Up Studies (PMCF).
- Risk Management: Identify potential risks early in the planning process and develop proactive mitigation strategies. This foresight is essential for minimizing disruptions and ensuring patient safety throughout the study. Grasping the regulatory environment, including the role of INVIMA as a Level 4 health authority by PAHO/WHO, is essential for adherence and supervision in research in Latin America.
By following these best practices in designing clinical trials for medical devices, researchers can greatly enhance the chances of success while ensuring compliance with regulatory requirements. Moreover, bioaccess® applies these best practices in its research services, guaranteeing that each study is structured with an emphasis on efficiency and patient care. Innovations in research studies are aimed at simplifying the site experience, with quicker queries and platform-based methods helping to reduce the time and effort spent on data entry.
Key Design Considerations for Medical Device Clinical Trials
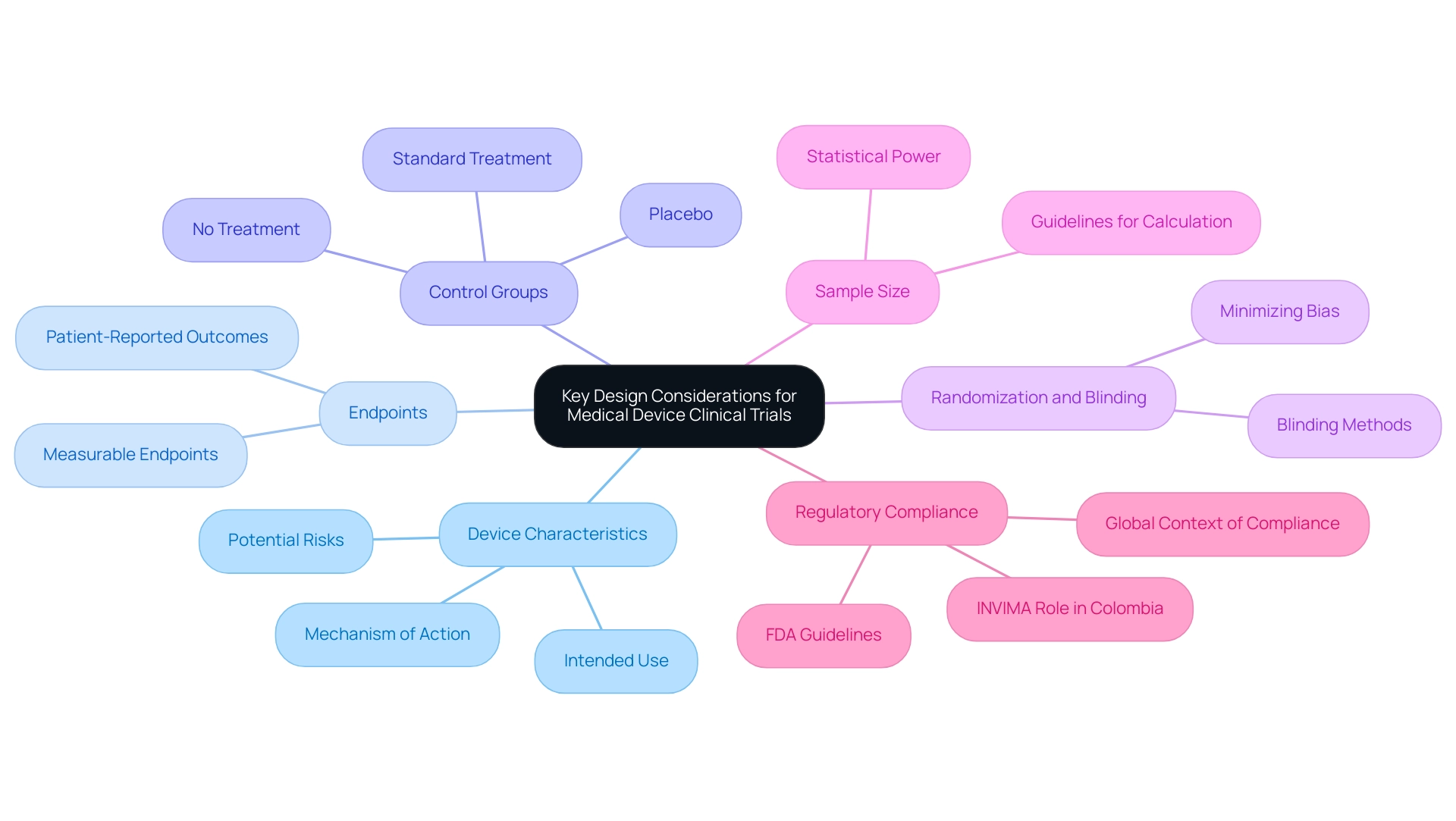
Designing clinical trials for medical devices necessitates a meticulous approach, where several critical factors must be taken into account:
- Device Characteristics: A thorough understanding of the device's specific features is paramount. This includes its intended use, mechanism of action, and potential risks associated with its application. Such insights assist in customizing the study framework to the distinct features of the apparatus.
- Endpoints: Clearly defined and measurable endpoints are essential to accurately reflect the apparatus's performance and safety. Successful endpoint definitions have been demonstrated in various case studies, showcasing how precise metrics can lead to more reliable outcomes and facilitate regulatory approval. Significantly, results from Nicole Latimer highlight the role of patient-reported outcomes in influencing reimbursement approaches for medical equipment studies, further emphasizing the significance of clearly defined endpoints.
- Control Groups: The choice of control group is crucial. Researchers must decide whether to utilize a placebo, standard treatment, or no treatment at all. This choice affects the experiment's capacity to show the tool's effectiveness in relation to current alternatives.
- Randomization and Blinding: Implementing randomization is vital to minimize bias in the study results. Additionally, considering blinding methods can significantly enhance the validity of the findings, ensuring that the outcomes are not influenced by participants' or researchers' expectations.
- Sample Size: Calculating an appropriate sample size is fundamental to achieving statistical power and ensuring the reliability of results. In 2025, the focus on strong sample size calculations has grown more significant, with guidelines adapting to represent the complexities of medical device studies.
- Regulatory Compliance: Adhering to relevant regulatory guidelines, such as those established by the FDA or EMA, is non-negotiable. The FDA Adverse Event Reporting System (FAERS) is backed by more than 150 nations, emphasizing the global context of regulatory adherence in clinical studies. Compliance not only ensures the integrity of the study but also facilitates smoother interactions with regulatory bodies during the approval process. In Colombia, the INVIMA plays a crucial role as a Level 4 health authority, overseeing medical regulations and ensuring compliance with national standards.
These factors are not simply procedural; they are crucial for creating an examination that can endure scrutiny from regulatory bodies and produce significant information regarding the device's safety and efficacy. By concentrating on these essential design aspects, researchers can enhance the effectiveness of designing clinical trials for medical devices and improve the chances of favorable results in their studies. Moreover, the trend towards enhanced data ownership and transparency among sponsors, as demonstrated in the case study titled 'Increased Data Ownership and Transparency by Sponsors,' signifies a shift in data management practices that can significantly influence study design considerations.
As Vivienne van der Walle aptly stated, "Anything that takes away time from patients is a pain point for a site, and anyone who resolves that is helping patient care." This viewpoint corresponds with the emphasis on reducing patient load in research studies.
Navigating Regulatory Requirements in Clinical Trials
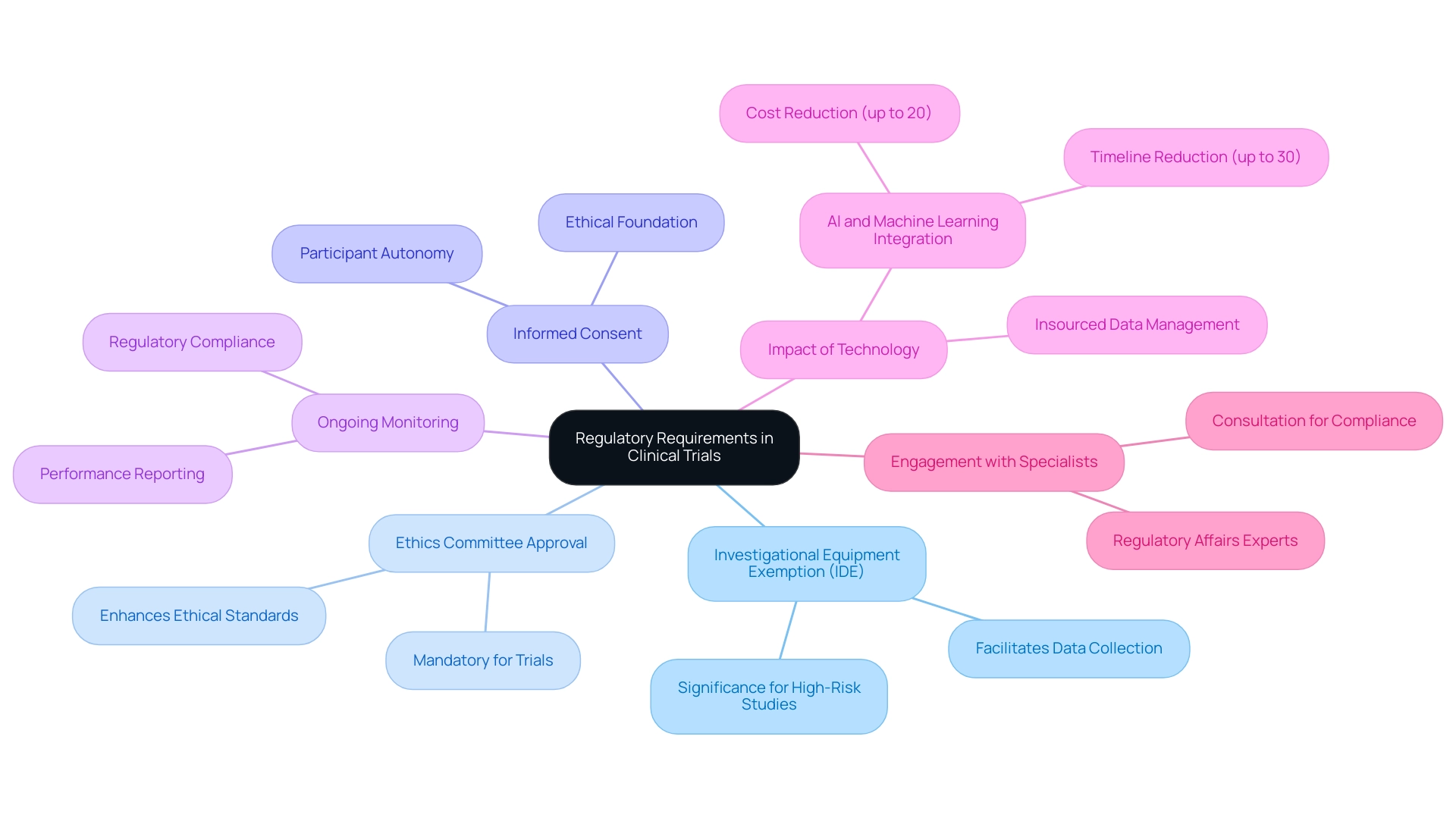
Regulatory standards for medical products are essential for guaranteeing participant safety and the integrity of the research process in designing clinical trials for medical devices. These requirements can differ greatly by area, but several key elements are universally acknowledged:
- Investigational Equipment Exemption (IDE): In the United States, an IDE is crucial for studies involving significant risk apparatus. This exemption allows the investigational instrument to be utilized in designing clinical trials for medical devices, facilitating the collection of necessary data to support safety and efficacy claims.
Securing approval from an Institutional Review Board (IRB) or ethics committee is mandatory when designing clinical trials for medical devices. This step is vital to ensure that designing clinical trials for medical devices adheres to ethical standards and prioritizes participant safety throughout the trial.
- Informed Consent: It is imperative that all participants provide informed consent, fully understanding the risks and benefits associated with their involvement in the study. This process not only respects participant autonomy but also enhances the ethical foundation of the trial, which is essential when designing clinical trials for medical devices, alongside compliance with guidelines for reporting trial results. This encompasses the duty to inform regulatory authorities about adverse events and outcomes, ensuring transparency and accountability in the research process.
Following approval, ongoing monitoring of the product's performance is essential for designing clinical trials for medical devices. This includes reporting any issues to regulatory bodies, which is essential for designing clinical trials for medical devices to ensure their safety and efficacy in the market. In 2025, the landscape of regulatory requirements continues to evolve, with a notable emphasis on the integration of advanced technologies such as artificial intelligence and machine learning. These innovations are streamlining operations and lowering expenses, with studies indicating that they can shorten timelines by up to 30% and costs by as much as 20%. Furthermore, sponsors are increasingly adopting insourced models for data management, seeking enhanced control and transparency over their data, as they aim to optimize the development journeys of assets to achieve not only an approval-enabling endpoint but to qualify for commercial success, as noted by Max Baumann, Head of Execution at Treehill Partners.
The significance of informed consent remains paramount, as it not only protects participants but also fosters trust in the research process. Recent statistics suggest that ethics committee approval rates are enhancing, demonstrating an increasing dedication to ethical standards in research studies. Specific statistics show that these approval rates are on the rise, indicating a positive trend in the regulatory landscape, which is important for designing clinical trials for medical devices. As regulatory frameworks adapt to the changing landscape, including the increasing attractiveness of the Japanese market to Western biopharmaceuticals due to regulatory changes, staying informed about updates to the Investigational Device Exemption (IDE) processes and other regulatory requirements is essential for researchers.
Engaging with Regulatory Affairs specialists, such as Ana Criado, Director of Regulatory Affairs, and Katherine Ruiz, an expert in designing clinical trials for medical devices, can provide valuable insights into navigating these complexities. bioaccess provides extensive management services for research studies, including feasibility assessments, site selection, compliance evaluations, study setup, import permits and nationalization of investigational tools, project management and oversight, and reporting (study status, inventory, serious and non-serious adverse events), ensuring that research studies are structured and carried out efficiently.
Data Management and Statistical Planning: Ensuring Valid Results
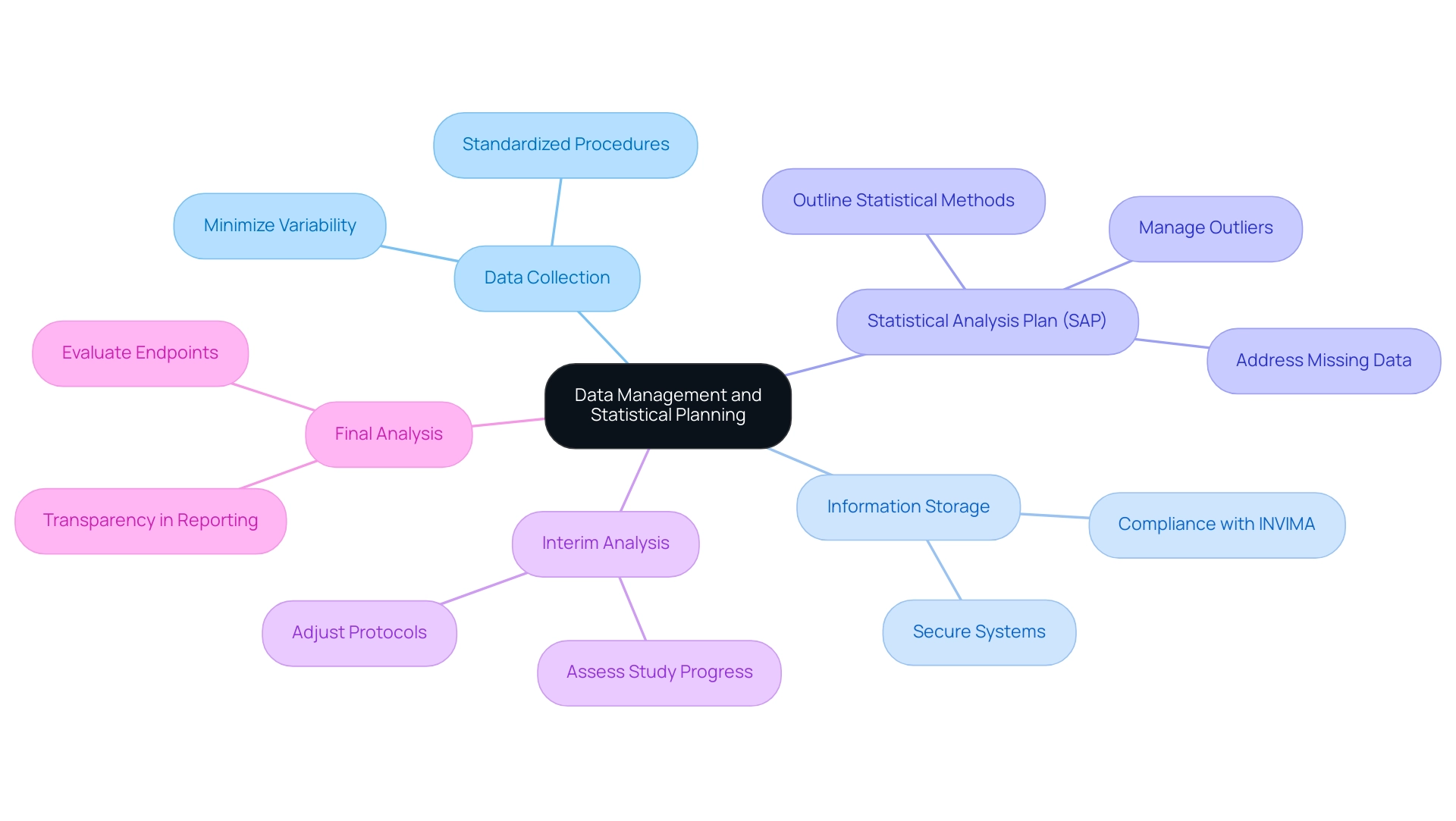
Key components of effective data management and statistical planning in clinical trials for medical devices are crucial for ensuring reliable outcomes:
- Data Collection: Establishing standardized procedures for data collection minimizes variability and bias, ensuring that the information gathered is trustworthy and can be efficiently analyzed. This principle aligns with bioaccess's commitment to high-quality clinical study management services.
- Information Storage: The utilization of secure and compliant systems for information storage is vital. These systems must guarantee confidentiality and integrity, safeguarding sensitive information throughout the process, particularly in accordance with INVIMA regulations, which oversee medical device oversight in Colombia.
- Statistical Analysis Plan (SAP): A well-organized SAP is essential, outlining the statistical methods to be employed for analysis. This includes strategies for addressing missing data and outliers, which can significantly impact the validity of study results. Bioaccess's expertise in designing clinical trials for medical devices—including Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies—ensures that these plans are meticulously crafted to meet regulatory standards.
- Interim Analysis: Planning for interim analysis allows researchers to assess the study's progress at predetermined intervals. This proactive approach enables timely adjustments to the study protocol, enhancing the overall efficiency of the trial, a practice that bioaccess incorporates into its project management methodologies.
- Final Analysis: Conducting a thorough final analysis is necessary to evaluate both primary and secondary endpoints. Transparency in reporting results is paramount, as it fosters trust and credibility in the findings—values that bioaccess upholds in its clinical study services.
Implementing these robust information management and statistical planning practices is essential in designing clinical trials for medical devices, as it not only enhances the credibility of research findings but also supports informed decision-making. For instance, sponsors are increasingly insourcing information management to maintain direct access to live information, facilitating better control and quality. As noted by the Head of Clinical Data Engineering, "Part of the initiative is to bring all our studies in-house so that our internal teams can start working on it. They can be more involved, and we implement studies internally, allowing us to take command of our information and deliver high quality for our patients."
A notable example is Alcon, where 45% of information is entered on the same day as the visit date, reflecting a commitment to accuracy and timeliness.
Furthermore, case studies illustrate the effectiveness of proactive issue management utilizing historical information. By establishing thresholds and sharing insights across departments, sponsors can monitor trends and address anomalies quickly, resulting in enhanced information quality and resource efficiency. This collaborative approach is essential for successful implementation and ultimately contributes to shorter study timelines.
Innovations in research studies are also focused on simplifying the site experience, with efforts to lessen the burden on locations and improve data entry efficiency, further enhancing the overall quality of medical research. With bioaccess as a vetted CRO and consulting partner, U.S. medical equipment companies can navigate these complexities effectively in Colombia.
Overcoming Challenges in Clinical Trial Execution
Common challenges in designing clinical trials for medical devices can significantly impact the success of medical device research. Key areas of concern include:
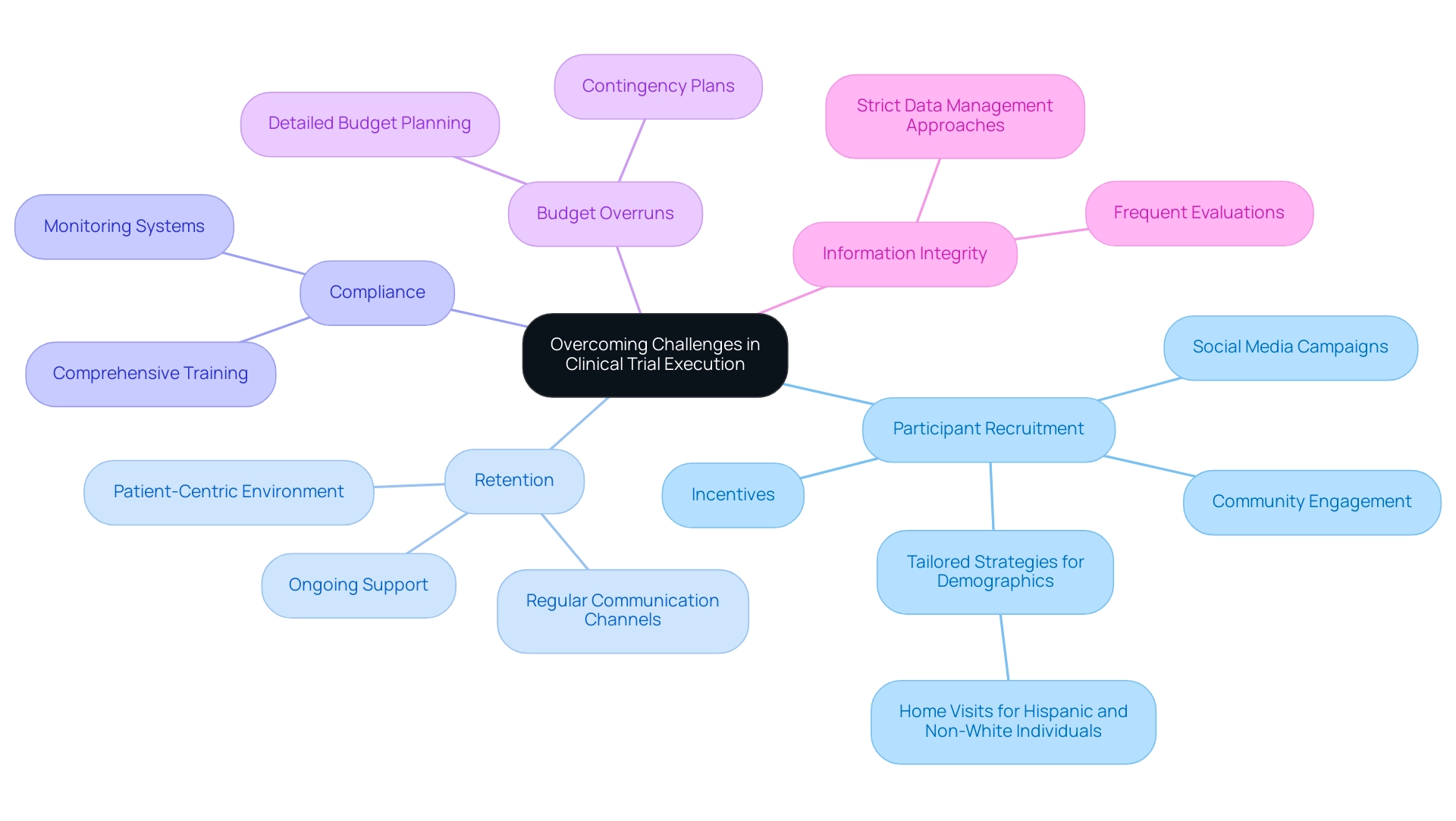
- Participant Recruitment: Identifying eligible participants remains a major hurdle, often leading to delays in study timelines. With over 6000 different rare diseases, the need for diverse participant recruitment is critical. To enhance recruitment efforts, strategies such as community engagement initiatives, targeted social media campaigns, and the provision of incentives can be effective. Engaging patients early in the process not only increases recruitment rates but also fosters a sense of ownership and advocacy for the research. Notably, a study by Antidote found that home visits appealed more to Hispanic and non-white individuals, highlighting the importance of tailored recruitment strategies that consider demographic factors.
- Retention: Maintaining participant engagement throughout the research is vital for ensuring robust data collection and study integrity. Implementing regular communication channels, providing ongoing support, and creating a patient-centric environment can significantly enhance retention rates. For instance, case studies from organizations like MD group demonstrate that prioritizing participant needs leads to improved retention and advocacy, ultimately resulting in more successful studies. Engaged patients are more likely to complete clinical studies and become advocates for future medical research, reinforcing the significance of participant engagement strategies.
- Compliance: Adherence to study protocols is crucial for the validity of study outcomes. To mitigate compliance challenges, comprehensive training for both staff and participants is essential. Additionally, establishing monitoring systems can help track adherence and identify potential issues early on.
- Budget Overruns: Clinical studies often face unexpected costs that can jeopardize their feasibility. Developing a detailed budget that includes contingency plans is critical for managing financial risks effectively. This proactive method can assist researchers in managing unexpected costs without jeopardizing the study's integrity.
- Information Integrity: Ensuring the quality and accuracy of information gathered during experiments is paramount. Applying strict data management approaches, together with frequent evaluations, can protect against mistakes and uphold the integrity of the research results.
By proactively tackling these issues, researchers can boost the chances of successful outcomes when designing clinical trials for medical devices, ultimately promoting the advancement of innovative medical solutions and enhancing patient care. At bioaccess®, with over 20 years of experience in Medtech, we aim to advance medical instruments sooner through our expertise and customized approach, ensuring that these challenges are met with effective solutions. Our extensive management services for research studies, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF), are intended to assist U.S. medical equipment firms in navigating the complexities of research in Latin America, contributing to local economies through job creation and healthcare enhancements.
Future Trends in Clinical Trial Design for Medical Devices
New developments in research design are transforming the environment for medical devices, particularly through the design of clinical trials, which provide creative strategies that enhance efficiency and participant involvement. At bioaccess®, we leverage our extensive experience in managing clinical studies across Latin America to implement these trends effectively:
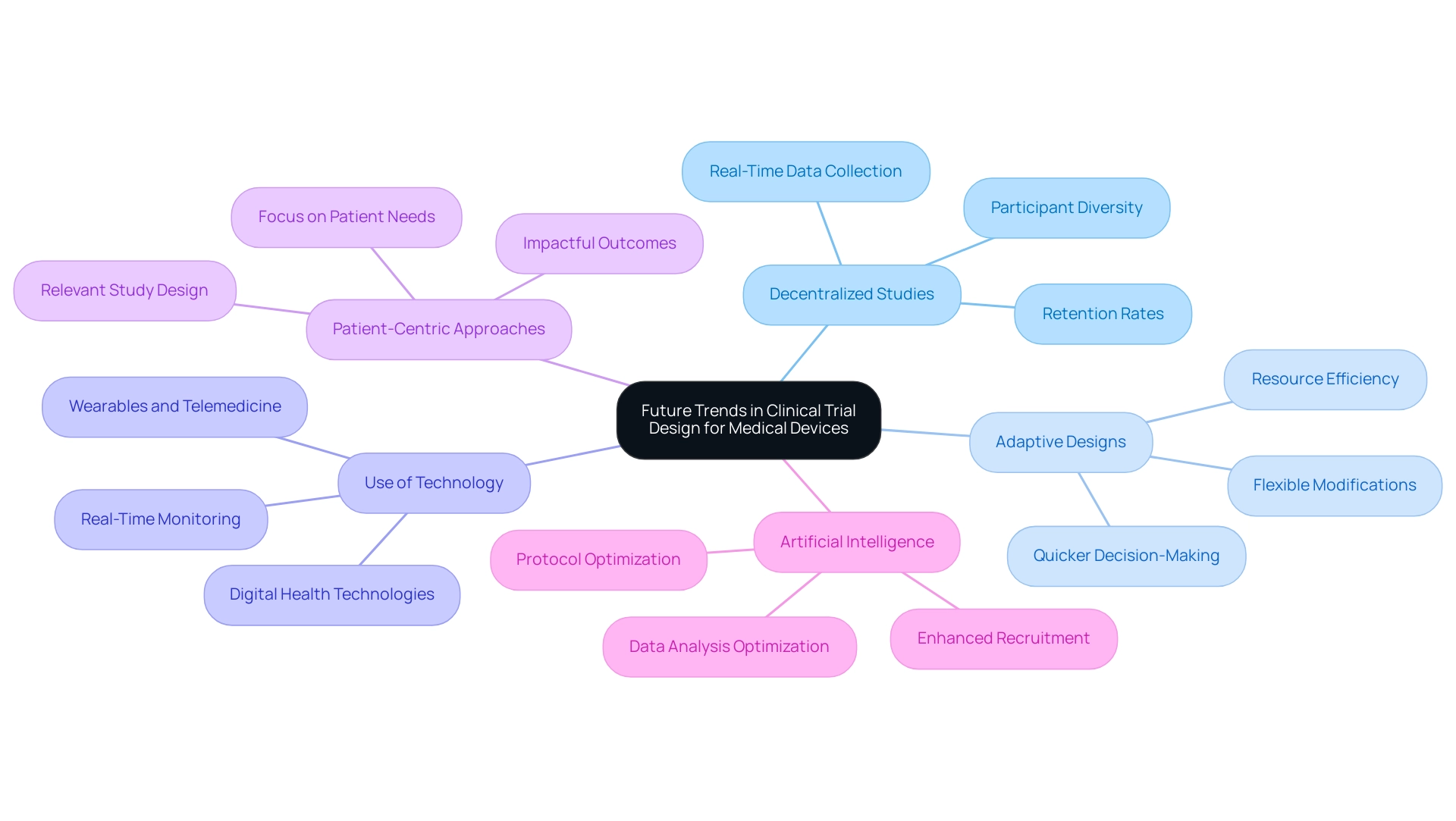
- Decentralized Studies: The shift towards remote studies is gaining momentum, allowing for increased participant diversity and convenience. This model not only broadens recruitment efforts but also improves retention rates, making studies more representative of the general population. Significantly, 45% of Alcon's information is recorded on the same day as the visit date, underscoring the effectiveness of collection methods in decentralized studies. Bioaccess® excels in facilitating these studies, ensuring seamless integration of remote monitoring technologies.
- Adaptive Designs: These flexible study designs enable modifications based on interim results, leading to more efficient resource use and quicker decision-making. By permitting modifications during the experiment, researchers can react to new information, ultimately improving the study's significance and efficiency. Our team at bioaccess® is skilled at applying adaptive designs in Early-Feasibility and Pivotal Studies, ensuring that research remains aligned with evolving objectives.
- Use of Technology: The integration of digital health technologies, including wearables and telemedicine, is transforming information collection methods. These tools facilitate real-time monitoring and enhance participant engagement, ensuring that information is both timely and accurate. Bioaccess® employs innovative technology to enhance information gathering and participant engagement in all our research studies.
- Patient-Centric Approaches: Focusing on patient needs and preferences is becoming vital in study design. By centering on what matters most to participants, researchers can create studies that are not only more relevant but also yield impactful outcomes that resonate with the target population. At bioaccess®, we emphasize patient-centric methodologies in our First-In-Human and Post-Market Research Follow-Up Studies.
- Artificial Intelligence: The application of AI in research studies is transforming data analysis, patient recruitment, and protocol optimization. By utilizing AI capabilities, researchers can optimize processes and improve the overall efficiency of medical studies. Bioaccess® is leading the way in integrating AI into our study management processes, ensuring that our clients benefit from the latest innovations in technology.
As the research environment evolves, adopting these trends is essential for researchers designing clinical trials for medical devices to enhance study efficiency and advance innovation. Max Baumann from Treehill Partners has noted that as 2025 approaches, biotech encounters significant business model challenges as end-markets become increasingly crowded. This highlights the necessity for early-stage developers to concentrate on commercial outcomes in their strategies. Moreover, the FDA's recent push for mandatory single IRB reviews exemplifies the regulatory shift towards more efficient research processes, aligning with the growing complexity of medical studies.
This modernization aims to reduce administrative burdens, facilitating quicker study initiation across multiple institutions, which is vital for the timely advancement of medical technologies. The focus on designing clinical trials for medical devices underscores the importance of optimizing development journeys for commercial success in achieving favorable outcomes, a commitment that bioaccess® firmly upholds.
Conclusion
The exploration of clinical trials in medical device research underscores their indispensable role in ensuring safety and efficacy. From foundational preclinical studies to the rigorous phases of human trials and critical post-market surveillance, each stage is vital to the overall success of medical innovations. A comprehensive understanding of these phases empowers researchers and stakeholders to design effective studies that adhere to regulatory requirements, ultimately enhancing patient care.
Strategic planning and thoughtful design considerations are essential for navigating the complexities of clinical trials. By defining clear objectives, identifying the target population, and establishing robust protocols, researchers can significantly enhance the likelihood of trial success. Furthermore, addressing challenges such as participant recruitment, retention, compliance, and data integrity is crucial for maintaining the integrity of the research process.
As the landscape of clinical trials continues to evolve, embracing emerging trends—such as decentralized trials, adaptive designs, and the integration of advanced technologies—will be key to driving innovation in medical device research. The emphasis on patient-centric approaches and the application of artificial intelligence further highlight a commitment to enhancing trial efficiency and participant engagement.
In conclusion, the future of medical device research hinges on a comprehensive understanding of clinical trials and a proactive approach to overcoming challenges. By staying informed about regulatory requirements and leveraging innovative strategies, researchers can significantly impact the advancement of medical technologies, ultimately leading to improved patient outcomes and healthcare solutions.
Frequently Asked Questions
What is the purpose of designing clinical trials for medical devices?
The purpose is to systematically evaluate the safety and efficacy of medical devices through human participants, gathering information that informs regulatory decisions and medical practices to enhance patient care.
What are the key phases of clinical trials for medical devices?
The key phases include: Preclinical Studies: Initial tests in laboratories and animal models to gather preliminary data. Phase I Trials: Focus on safety and determining safe dosage ranges with a small group of participants. Phase II Trials: Evaluate effectiveness and continue safety assessments with a larger group. Phase III Trials: Verify effectiveness, observe side effects, and compare against standard treatments with diverse populations. Post-Market Surveillance: Continuous observation of the product's performance in the general population after approval.
Why is understanding the phases of clinical trials important?
Understanding these phases is vital for researchers to design compliant clinical trials that ultimately improve patient outcomes.
How has automated information mining impacted medical studies?
Automated information mining has effectively matched around 84% of medical studies to specific diseases or conditions, highlighting the importance of focused research.
What innovations have been made to enhance site experiences in research trials?
Innovations include addressing pain points such as prolonged entry processes, which significantly improve operational efficiency and patient care.
What is the role of bioaccess® in medical device research studies in Latin America?
Bioaccess® provides accelerated research study services, leveraging over 20 years of expertise in managing various types of studies, including Early-Feasibility Studies and Post-Market Follow-Up Studies.
What are the best practices for optimizing study design in clinical trials?
Best practices include: Defining objectives clearly. Identifying the target population. Developing a comprehensive study protocol. Budgeting and resource allocation. Timeline development. Risk management.
How can effective budgeting influence clinical trial success?
Effective budgeting can significantly influence feasibility and success rates, ensuring that the project remains within budget and resources are allocated efficiently.
What is the importance of risk management in clinical trials?
Identifying potential risks early and developing proactive mitigation strategies is essential for minimizing disruptions and ensuring patient safety throughout the study.
How does bioaccess® ensure compliance with regulatory requirements?
Bioaccess® applies best practices in its research services, ensuring that each study is structured for efficiency and patient care while adhering to regulatory standards.




