Overview
Navigating trial protocols in Brazil necessitates a comprehensive understanding of the regulatory framework established by ANVISA and CONEP, alongside strict adherence to ethical guidelines and submission processes. This article underscores that successful navigation is contingent upon:
- Thorough preparation
- Fostering local partnerships
- Ensuring compliance with regulations
Such diligence not only enhances the integrity of clinical research but also boosts its efficiency within the region.
Introduction
Navigating the intricate landscape of clinical trials in Brazil demands a profound understanding of the regulatory framework governing research involving human subjects. With agencies like ANVISA and CONEP at the forefront, researchers are required to adhere to stringent ethical guidelines and regulatory requirements that safeguard participant welfare and uphold study integrity.
Recent legislative changes, including Law No. 14.874, seek to streamline the approval process, fostering a more efficient environment for clinical research. However, challenges such as protracted approval times and cultural nuances can complicate recruitment efforts.
This article explores the essential components of conducting clinical trials in Brazil, underscoring the critical importance of:
- Compliance
- Ethical considerations
- The pivotal role of local Contract Research Organizations (CROs) in surmounting these obstacles.
By comprehending and leveraging these insights, researchers can significantly enhance their chances of successful trial execution and contribute to the advancement of medical innovation in the region.
Understanding the Regulatory Framework for Clinical Trials in Brazil
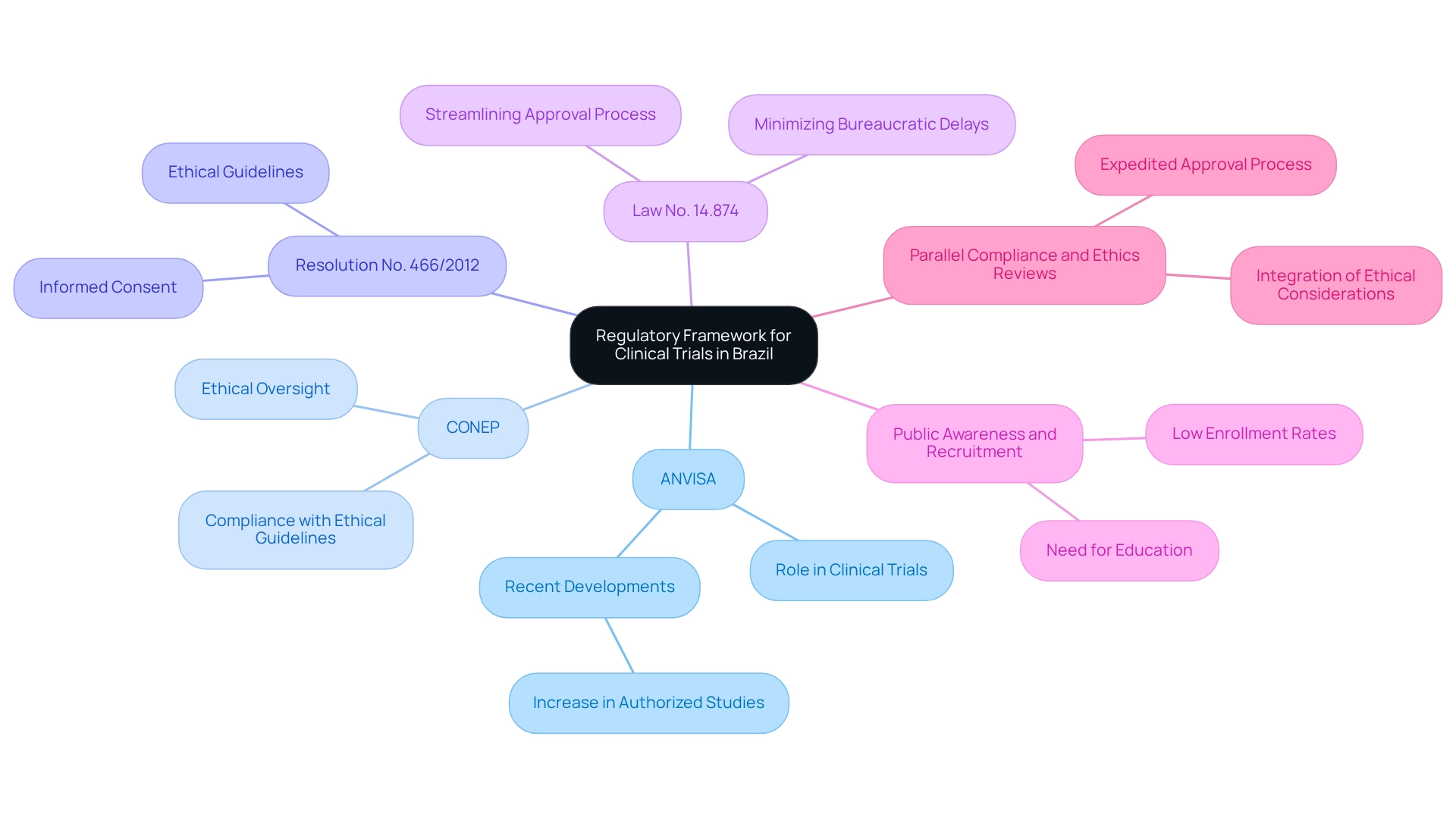
Navigating trial protocols in Brazil is fundamentally shaped by the oversight framework established by the National Health Surveillance Agency (ANVISA) and the National Commission for Ethics in Research (CONEP). Researchers must maneuver through a complex landscape that includes critical regulations such as Resolution No. 466/2012, which lays down essential ethical guidelines for conducting research involving human subjects.
This resolution underlines the necessity of informed consent and the ethical treatment of participants, ensuring their rights and welfare are prioritized throughout the research process. Furthermore, the recent enactment of Law No. 14.874 in May 2024 marks a significant advancement in the governance framework, aiming to streamline the approval process for medical studies. This law is crafted to minimize bureaucratic delays, thereby facilitating quicker access to innovative treatments for patients.
Understanding these regulations is imperative for researchers navigating trial protocols in Brazil, as it enables them to align their study protocols with national standards and ethical guidelines, ultimately enhancing the integrity of their investigations.
Moreover, the simultaneous execution of compliance and ethics evaluations for research proposals can greatly expedite the approval process. This integrated approach not only fosters efficiency but also ensures that ethical considerations are embedded within the regulatory framework from the outset. The significance of this method is underscored by case analyses demonstrating that navigating trial protocols in Brazil often closely adheres to ANVISA regulations, resulting in higher approval rates and more efficient operational processes.
bioaccess® offers extensive management services for research, including feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting. With over 20 years of experience in Medtech, bioaccess® specializes in various study types, such as Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF). This expertise is crucial for navigating trial protocols in Brazil and effectively maneuvering through the intricacies of the compliance framework.
Additionally, a survey revealed a deficiency in public awareness about medical research, leading to low enrollment rates for studies. Numerous oncologists indicated that patients are often unaware of the benefits of participating in research studies. Enhancing public knowledge and education regarding research studies is vital for increasing patient involvement and addressing recruitment challenges faced by oncologists.
As of 2024, ANVISA has reported a rise in the number of authorized research studies, reflecting the positive impact of these oversight advancements. This increase not only illustrates the effectiveness of the new legislation but also highlights the necessity for adherence and ethical consideration of participants in research studies.
In conclusion, navigating trial protocols in Brazil, along with having a comprehensive understanding of the country's research regulations and the roles of ANVISA and CONEP, is essential for researchers aiming to conduct ethical and compliant investigations. By staying informed about the evolving compliance landscape and leveraging the expertise of bioaccess®, researchers can more effectively manage the complexities of studies in Brazil, ultimately advancing medical technology and enhancing patient care.
Key Requirements for Conducting Clinical Trials in Brazil
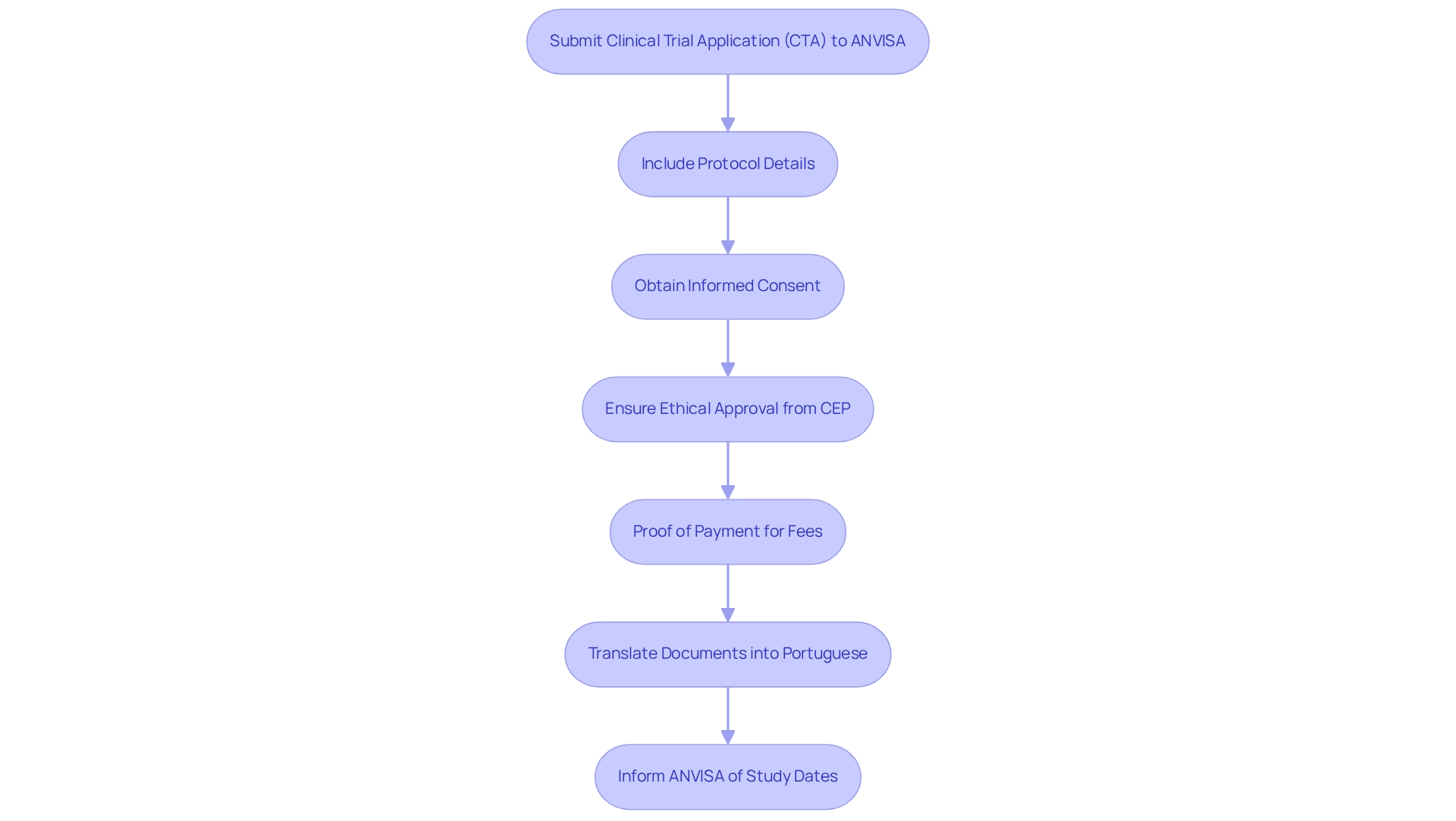
Conducting clinical trials in Brazil requires the submission of a Clinical Trial Application (CTA) to ANVISA, the national health regulatory agency. This application must include comprehensive details about the protocol, covering the objectives, methodology, and expected outcomes. Additionally, informed consent documents are essential, ensuring that participants are fully aware of their rights and the nature of the research.
Proof of payment for any applicable fees associated with the application process is also necessary. Ethical approval from a Research Ethics Committee (CEP) is a critical requirement that cannot be overlooked; this committee evaluates the ethical implications of the study, ensuring that participant welfare is prioritized. Researchers must ensure that all documentation is translated into Portuguese and complies with local regulations, as this is vital for a smooth approval process.
Statistics indicate that sponsors must inform ANVISA of study start and end dates within 30 calendar days after each date, reflecting the regulatory requirements that must be adhered to. Furthermore, a significant percentage of clinical trials submitted to ANVISA receive approval, underscoring the agency's commitment to facilitating research while upholding rigorous safety standards. In recent years, the approval rate has been notably high, highlighting the importance of thorough preparation and adherence to submission requirements.
In emergency research scenarios, investigations may proceed without consent if participants are in life-threatening conditions and obtaining consent is not feasible, provided that specific criteria are met and sanctioned by an ethics committee. This aspect of the oversight landscape is crucial for researchers to understand.
Case studies highlight the necessity of informed consent, particularly in sensitive situations involving participants who may have impaired autonomy. For instance, research involving mentally impaired individuals mandates that consent be obtained through legal representatives, ensuring that ethical standards are upheld. This framework not only safeguards the rights of at-risk groups but also emphasizes the significance of transparent communication and the necessity for agreement without pressure.
By comprehending and navigating these regulatory obstacles, researchers can enhance their opportunities for successful study initiation and facilitate trial protocols in Brazil, thereby supporting the advancement of medical technology. bioaccess® provides extensive assistance for conducting research studies in Brazil, connecting clients with skilled service providers and specialists, thus strengthening their knowledge in this essential field. Our services encompass feasibility studies, site selection, compliance reviews, testing setup, import permits, project management, and reporting, ensuring a streamlined process for medical device research in the region.
Moreover, bioaccess® focuses on overseeing Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH), offering tailored solutions that address the distinct challenges of each study. With a proven history in overcoming regulatory hurdles, bioaccess® is dedicated to enabling successful research outcomes in Brazil.
Navigating the Clinical Trial Approval Process in Brazil
Navigating the clinical trial approval process in Brazil necessitates a comprehensive understanding of several critical steps:
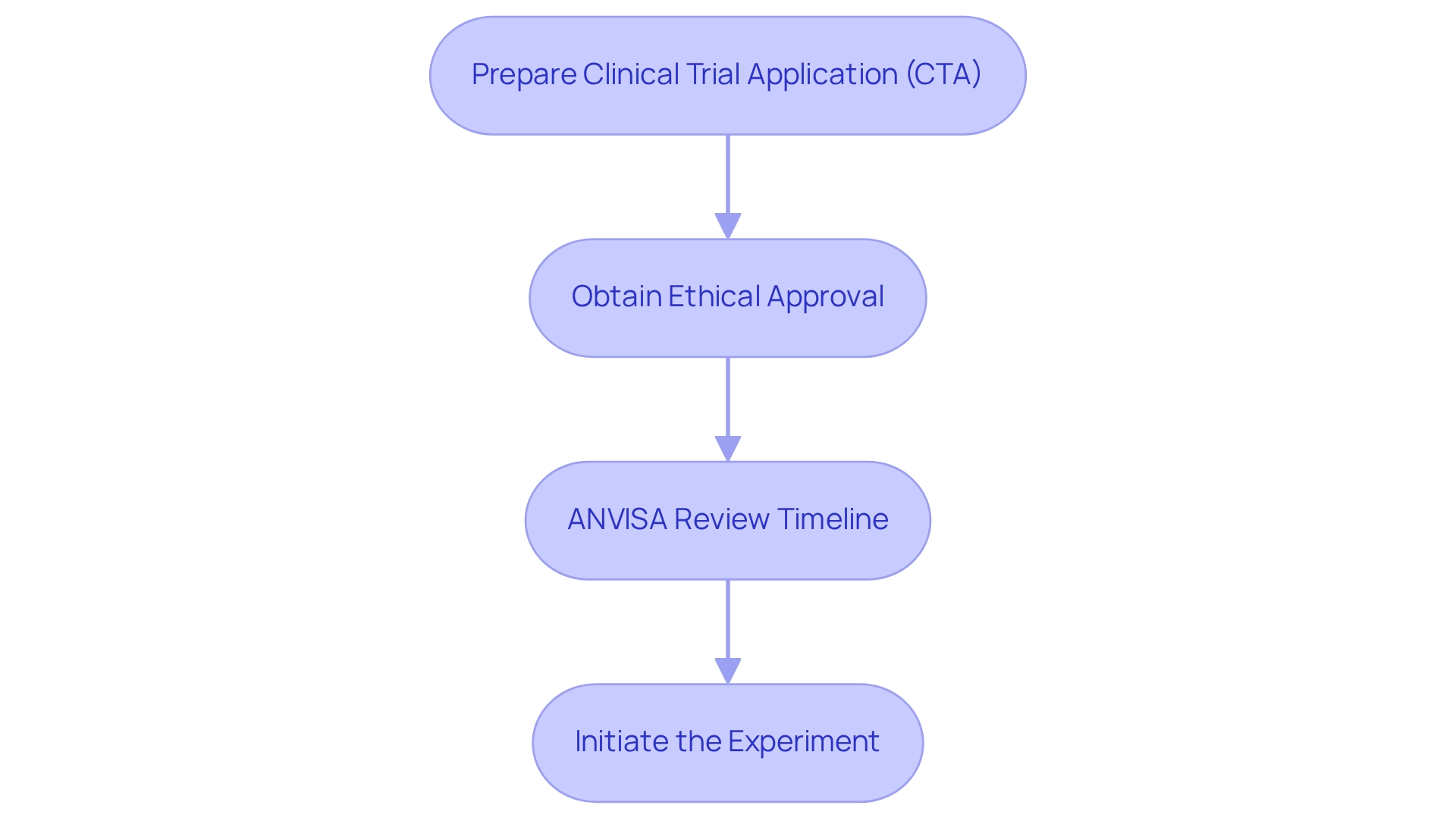
- Prepare the Clinical Trial Application (CTA): Researchers must compile and submit the CTA to ANVISA, ensuring that all required documentation is included to avoid delays. This step is crucial for leveraging the expertise of bioaccess®, a leading Contract Research Organization (CRO) specializing in managing clinical trials in Latin America, ensuring meticulous preparation of all necessary documentation.
- Obtain Ethical Approval: After submission to ANVISA, securing ethical approval from a local Research Ethics Committee (CEP) is essential. Depending on the research, this may also require submission to the National Commission for Ethics in Research (CONEP). Bioaccess® can assist in navigating these ethical requirements, streamlining the approval process for researchers.
- ANVISA Review Timeline: ANVISA is mandated to review the application within a 90-business-day timeframe. However, challenges have emerged, as the agency has historically averaged nearly 8 months for protocol reviews. This discrepancy raises concerns among stakeholders regarding the feasibility of meeting the new deadlines established by Law Number 13,411, effective March 2017. Legal specialists have observed that these prolonged timelines may disrupt research and endanger sponsor investments due to uncertainties in protocol approval. A case analysis titled 'ANVISA's Challenges with New Clinical Trials Law' highlights these difficulties, emphasizing potential complications arising from the agency's struggle to comply with new deadlines.
- Initiate the Experiment: Once the application is approved, researchers can commence the study. Upholding adherence to all regulatory requirements throughout the research is essential to ensure its integrity and success. Bioaccess® provides extensive management services for research projects, encompassing feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting—crucial for successful project initiation. Additionally, bioaccess® focuses on various study types, including Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH), ensuring researchers receive the necessary assistance at every phase of the process.
Grasping these steps is essential for researchers navigating trial protocols in Brazil for prompt study initiation. By effectively navigating trial protocols in Brazil, manufacturers can secure higher prices and develop more efficient launch strategies for their products in this burgeoning market. As Dr. Barbosa da Silva, former ANVISA Director, emphasized, "ANVISA has conducted a comprehensive review of its process, with the objective to strengthen the registration process, and to have a more transparent and predictable ecosystem for all stakeholders."
This commitment is vital for fostering confidence among researchers and sponsors alike. For additional details on how bioaccess® can support your research requirements, please reach out to us today.
Ethical Considerations and Approvals in Clinical Research
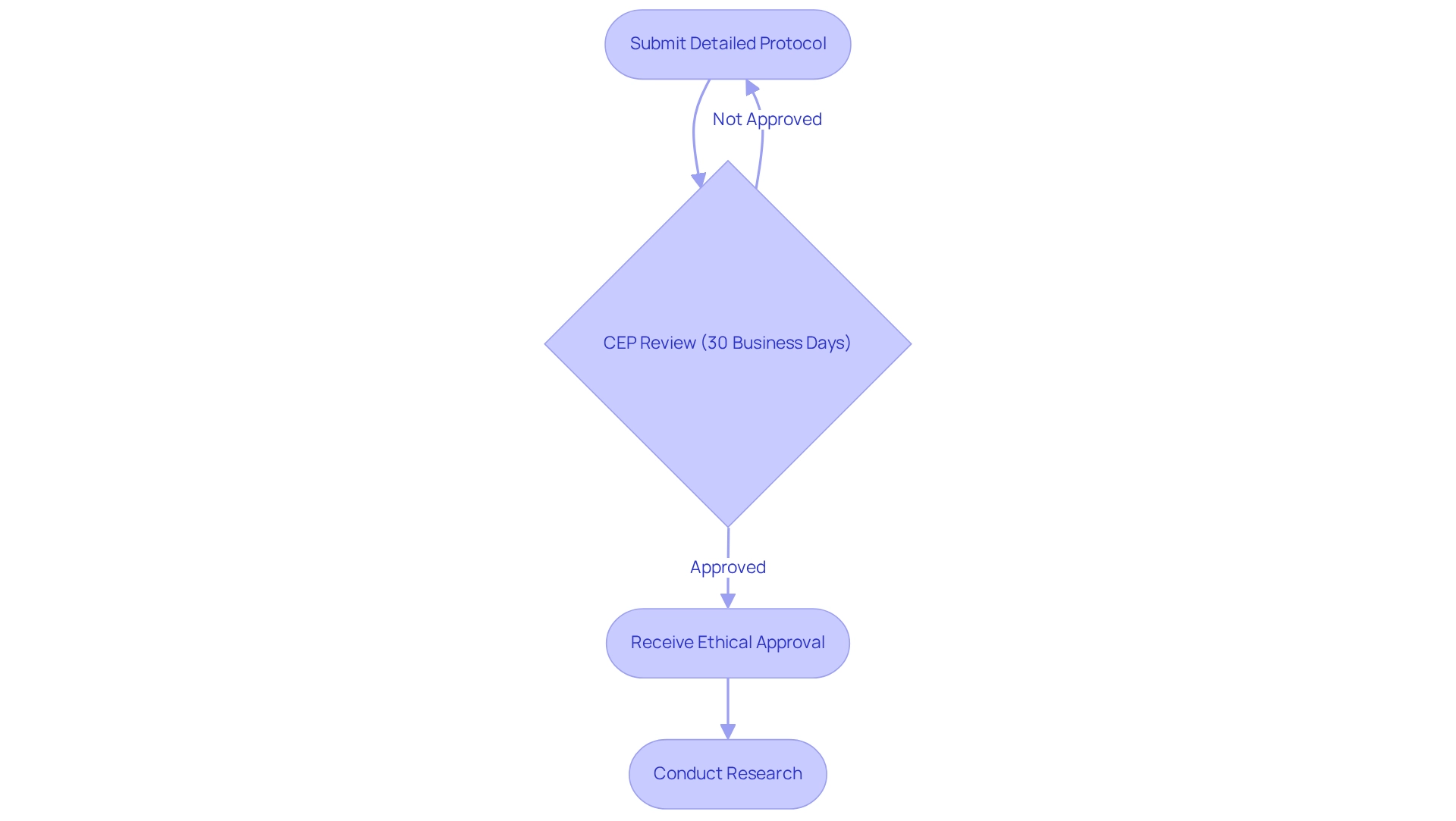
Navigating trial protocols in Brazil underscores the paramount significance of ethical considerations in clinical research. All clinical trials are required to undergo a comprehensive ethical review by a Research Ethics Committee (CEP), which meticulously evaluates the research design, informed consent procedures, and potential risks to participants. Researchers must submit a detailed protocol that outlines the project's objectives, methodology, and ethical safeguards.
The ethical review process in Brazil is designed for efficiency, with the CEP typically completing its assessment within 30 business days. This timeline is crucial for researchers who aim to expedite their research while ensuring compliance with ethical standards. Notably, around 85% of clinical trials in Brazil secure ethical approval, reflecting a robust framework dedicated to participant protection and the integrity of research.
Informed consent stands as a cornerstone of ethical research practices in Brazil, ensuring that participants are thoroughly informed about the risks and benefits before agreeing to participate. Recent case analyses, such as those involving
The Role of Local CROs in Streamlining Clinical Trials
Local Contract Research Organizations (CROs) play a crucial role in navigating trial protocols in Brazil, providing essential expertise in compliance with regulations, site management, and patient recruitment, all vital for the success of research studies. Their profound understanding of the local compliance environment, particularly the requirements established by ANVISA, enables them to effectively support foreign sponsors in navigating trial protocols and the complexities of the Clinical Trial Application (CTA) process. This includes managing communications with ANVISA and ethics committees, ensuring meticulous adherence to local regulations.
Statistics indicate that studies employing local CROs achieve higher success rates, as these organizations excel in navigating trial protocols in Brazil and addressing the specific challenges posed by the Brazilian research landscape. For instance, new medications intended solely for experimental purposes can be imported with direct approval from the Ministry of Health, allowing them to circumvent registration for up to three years, significantly expediting the testing process. Furthermore, ANVISA has developed an online platform, VigiMed, for reporting unforeseen serious adverse events (SAEs) associated with medications and vaccines, which further enhances compliance with regulations.
Additionally, a technical note issued by ANVISA on March 23, 2020, provides guidance on conducting clinical research and bioequivalence assessments, ensuring that CROs remain well-informed about current regulatory expectations.
bioaccess® distinguishes itself as a leader in Medtech clinical research across Latin America, offering comprehensive management services for clinical trials that encompass feasibility assessments, site selection, compliance reviews, setup, import permits, project management, and detailed reporting (including research status, inventory, and serious and non-serious adverse events), as well as review and feedback on research documents. Their expertise in overseeing Early-Feasibility Assessments (EFS), First-In-Human Experiments (FIH), Pilot Assessments, Pivotal Assessments, and Post-Market Clinical Follow-Up Evaluations (PMCF) ensures that evaluations are executed efficiently and effectively.
Case analyses highlight the importance of ethical considerations, particularly in research involving indigenous populations. Local CROs are committed to ensuring that studies respect the cultural and social contexts of these communities, ultimately benefiting both the research and the participants involved. This commitment to ethical principles fosters trust and enhances the overall integrity of research studies.
Successful partnerships between CROs and researchers in Brazil demonstrate that navigating trial protocols can streamline study processes and improve outcomes. By collaborating with local CROs, researchers can enhance their operational efficiency and increase the likelihood of achieving their medical objectives, making these organizations invaluable assets in the pursuit of innovative healthcare solutions.
Challenges in Conducting Clinical Trials in Brazil and How to Overcome Them
Navigating trial protocols in Brazil involves conducting medical studies within a landscape marked by compliance challenges, extended approval procedures, and cultural subtleties that can significantly influence patient recruitment. Recent studies reveal a strong interest among local patients, particularly among Brazilian and Peruvian oncologists, to participate in clinical studies, underscoring the potential for successful recruitment when approached strategically.
-
Engage with Local Contract Research Organizations (CROs): Collaborating with local CROs, such as bioaccess®, offers invaluable insights into the compliance landscape, ensuring adherence to Good Clinical Practices (GCP) guidelines. Following these standards is crucial, as non-compliance can lead to substantial delays in regulatory approval or even rejection of application submissions. A case study highlighted that failure to comply with GCP guidelines resulted in prolonged approval durations, emphasizing the importance of these standards in the research process.
-
Build Strong Relationships with Local Ethics Committees: Establishing rapport with local ethics committees can facilitate smoother approval processes. These committees play a vital role in ensuring that assessments meet ethical standards, and their support can expedite the review process.
-
Develop Culturally Sensitive Recruitment Strategies: Tailoring recruitment efforts to resonate with local populations can significantly enhance participant engagement. Understanding cultural perspectives on research studies and addressing potential issues can greatly improve recruitment rates. For instance, bioaccess® has successfully implemented culturally aware approaches in its partnerships, such as the collaboration with Caribbean Health Group to position Barranquilla as a key location for health studies in Latin America, supported by Colombia's Minister of Health. This partnership has resulted in a reported 50% decrease in hiring duration and a 95% retention rate, highlighting the effectiveness of localized strategies.
Navigating trial protocols in Brazil reveals that the average approval duration for studies can be extensive, often exceeding initial estimates according to recent evaluations. However, by proactively addressing these regulatory hurdles and fostering local partnerships, researchers can enhance the efficiency and success of their studies. This proactive approach ultimately contributes to improved healthcare practices in the region. Leveraging the expertise of organizations like bioaccess® in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies can further optimize the research process and promote global health enhancement through international cooperation and innovation in Medtech.
Ensuring Data Management and Quality Assurance in Clinical Trials
Efficient data handling and strict quality control are paramount for the success of research studies, especially when navigating trial protocols in Brazil for bioaccess's extensive management services in Latin America. Researchers must establish robust data management systems that facilitate accurate data collection, secure storage, and thorough analysis. The adoption of electronic data capture (EDC) systems is particularly critical, as these systems not only streamline data handling but also ensure adherence to Good Clinical Practice (GCP) guidelines.
In Brazil, approximately 70% of medical studies now employ EDC systems, reflecting a significant shift toward contemporary data management methods. This trend underscores the necessity for researchers to prioritize efficient data management practices.
To guarantee data accuracy and compliance with standards, routine audits and oversight are essential. These practices help identify potential discrepancies early, ensuring the data remains reliable and valid. A recent case study involving Datavant Trials illustrated how integrating comprehensive data management solutions can enhance visibility into patient journeys by securing and connecting health data, ultimately leading to improved insights and outcomes in research studies.
Moreover, successful quality assurance practices in Brazilian research have demonstrated that implementing end-to-end data management solutions can reduce the drug development cycle by an average of 1.5 years and increase the likelihood of approval by 23%, as highlighted by the Pharmaceutical R&D Efficiency Index. This emphasizes the significance of prioritizing data management and quality assurance, as they greatly enhance the reliability of results and facilitate navigation through trial protocols in Brazil. Additionally, ensuring statistical rigor in medical research is essential; a well-defined group and sufficient sample size are critical for valid and generalizable outcomes.
By leveraging bioaccess's expertise in study management, including feasibility assessments, site selection, compliance evaluations, and study preparation, researchers can adeptly navigate the complexities of regulatory requirements while contributing to local economies through job creation and healthcare improvements.
Best Practices for Developing Effective Clinical Trial Protocols
Creating effective clinical study protocols is a vital step that requires careful planning and sharp attention to detail. To ensure success, researchers should adhere to the following best practices:
- Clearly Define Study Objectives and Endpoints: Establishing precise objectives and measurable endpoints is essential for guiding the trial and evaluating its outcomes. This clarity not only assists in adherence to regulations but also improves the overall quality of the research.
- Adhere to Regulatory Requirements and Ethical Guidelines: Protocols must comply with local and international regulations, including ethical considerations. This adherence fosters trust and integrity in the research process, which is vital for participant recruitment and retention. In Latin America, understanding the specific regulatory landscape, such as the role of INVIMA in Colombia, is crucial for ensuring compliance and facilitating market access.
- Detail Methodologies for Participant Recruitment, Data Collection, and Analysis: A comprehensive methodology section should outline how participants will be recruited, the data collection processes, and the analytical techniques to be employed. This level of detail guarantees that the experiment can be replicated and that the results are trustworthy.
- Engage Stakeholders in the Protocol Development Process: Involving key stakeholders—such as investigators, ethics committees, and patient representatives—early in the protocol development fosters collaboration and enhances the protocol's relevance. Successful examples of stakeholder engagement have shown that this approach leads to more robust and accepted protocols. For instance, a recent pilot study on Good Clinical Practice (GCP) training materials highlighted the importance of structured feedback from stakeholders, which led to significant refinements in the training content, ensuring its applicability in diverse clinical research environments.
- Consider Decentralized and Hybrid Studies: These study designs provide greater flexibility and diminish the necessity for on-site visits, which can enhance participant retention. By incorporating these methodologies, researchers can enhance the feasibility and effectiveness of their experiments.
By implementing these best practices, researchers can create protocols that not only meet regulatory standards but also facilitate successful execution. In Brazil, where the medical research environment is changing, navigating trial protocols can greatly enhance the success rates of studies. Research suggests that clearly outlined protocols are linked to greater success rates, making it essential for researchers to prioritize protocol development as a fundamental component of their investigations.
Bioaccess® provides extensive trial management services, encompassing feasibility studies, site selection, compliance evaluations, trial preparation, import permits, project oversight, and reporting, guaranteeing that your trials are carried out efficiently and effectively. This integrated approach underscores the importance of a seamless process in achieving successful outcomes in clinical research.
Conclusion
Navigating the complexities of clinical trials in Brazil necessitates a comprehensive understanding of the intricate regulatory framework, ethical considerations, and the pivotal role of local Contract Research Organizations (CROs). Researchers must comply with stringent guidelines established by ANVISA and CONEP, ensuring the protection and welfare of participants while adhering to ethical standards. Recent legislative changes, particularly Law No. 14.874, aim to streamline the approval process; however, challenges such as lengthy timelines and cultural nuances remain significant hurdles.
The importance of local expertise is paramount. Collaborating with experienced CROs like bioaccess® can significantly enhance trial efficiency by providing essential insights into regulatory compliance, ethical approvals, and patient recruitment strategies. Implementing culturally sensitive approaches in recruitment and engaging with local ethics committees can further facilitate smoother trial processes.
Ultimately, a commitment to ethical research practices, thorough preparation, and leveraging local resources will not only enhance the likelihood of successful clinical trials but also contribute to the advancement of medical innovation in Brazil. By prioritizing these aspects, researchers can effectively navigate the challenges ahead, fostering a robust clinical research environment that benefits both participants and the broader medical community.
Frequently Asked Questions
What regulatory bodies oversee clinical trials in Brazil?
Clinical trials in Brazil are overseen by the National Health Surveillance Agency (ANVISA) and the National Commission for Ethics in Research (CONEP.
What are the key regulations researchers must comply with when conducting trials in Brazil?
Researchers must comply with Resolution No. 466/2012, which outlines essential ethical guidelines for research involving human subjects, including the necessity of informed consent and the ethical treatment of participants.
What recent legislative change has impacted the approval process for medical studies in Brazil?
The enactment of Law No. 14.874 in May 2024 aims to streamline the approval process for medical studies, reducing bureaucratic delays and facilitating quicker access to innovative treatments for patients.
How can researchers expedite the approval process for their studies in Brazil?
Researchers can expedite the approval process by integrating compliance and ethics evaluations for research proposals, which ensures that ethical considerations are embedded within the regulatory framework from the outset.
What services does bioaccess® provide to assist researchers in navigating trial protocols in Brazil?
bioaccess® offers services including feasibility assessments, site selection, compliance evaluations, project oversight, import permits, and reporting, specializing in various study types such as Early-Feasibility Studies and First-In-Human Studies.
What challenges are faced regarding patient enrollment in research studies in Brazil?
There is a deficiency in public awareness about medical research, leading to low enrollment rates for studies. Many patients are unaware of the benefits of participating in research studies.
What has been the trend in authorized research studies in Brazil as of 2024?
As of 2024, ANVISA has reported an increase in the number of authorized research studies, reflecting the positive impact of recent oversight advancements.
What is required in the Clinical Trial Application (CTA) submitted to ANVISA?
The CTA must include details about the study protocol, objectives, methodology, expected outcomes, informed consent documents, proof of payment for fees, and ethical approval from a Research Ethics Committee (CEP).
Are there circumstances under which clinical trials can proceed without informed consent in Brazil?
Yes, in emergency research scenarios where participants are in life-threatening conditions and obtaining consent is not feasible, trials may proceed without consent if sanctioned by an ethics committee.
What is the importance of informed consent in clinical trials involving vulnerable populations?
Informed consent is crucial in protecting the rights of at-risk groups, such as mentally impaired individuals, where consent must be obtained through legal representatives to ensure ethical standards are upheld.




