Overview
The article emphasizes the critical need to understand future trends in medical device trials, particularly the role of adaptive study designs, digital health technologies, and participant-centered approaches. These trends are not merely innovative; they are essential for enhancing trial efficiency and effectiveness. By incorporating real-time data and patient feedback, they aim to improve outcomes and streamline the research process. This focus on adaptability and digital integration positions researchers to tackle existing challenges more effectively, ultimately leading to more successful clinical outcomes.
Introduction
The medical device trial landscape is undergoing a seismic shift, propelled by rapid technological advancements and evolving patient needs. Researchers navigating this dynamic environment must adapt their methodologies to ensure compliance and effectiveness. With the integration of digital health technologies and the rise of adaptive trial designs, there is a heightened focus on enhancing patient engagement and optimizing outcomes. This article explores the transformative trends reshaping medical device trials, examining how innovations such as:
- artificial intelligence
- digital therapeutics
- wearable technologies
streamline processes and pave the way for more inclusive and efficient clinical research. By understanding these developments, clinical researchers can better position themselves to lead successful trials in an ever-changing landscape.
The Evolving Landscape of Medical Device Trials
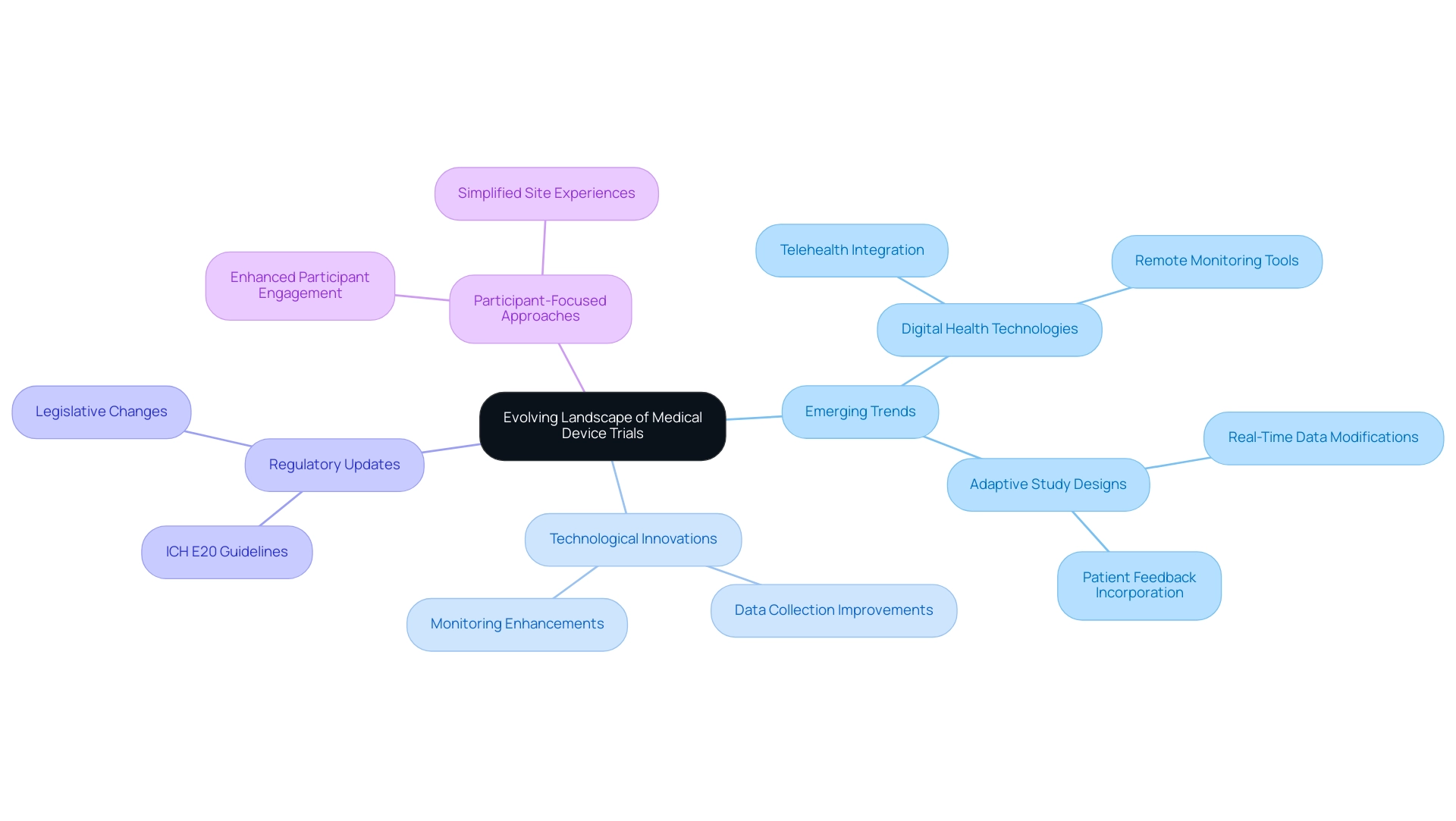
The landscape of medical device studies is undergoing a significant transformation, shaped by emerging trends in medical device trials, technological innovations, regulatory updates, and evolving individual requirements. To ensure compliance and enhance study designs, clinical researchers must remain informed about these developments. Key elements driving this evolution include:
- The integration of digital health technologies that improve data collection and monitoring
- A growing emphasis on participant-focused approaches that prioritize experience and outcomes
These factors are indicative of future trends in medical device trials.
As we look ahead to 2025, the emphasis on adaptive study designs is becoming increasingly critical. These designs empower researchers to modify experimental parameters based on real-time data and patient feedback, thereby enhancing the relevance and efficiency of investigations. The anticipated implementation of the ICH E20 guidelines on Adaptive Design is expected to further encourage the use of complex study elements, such as Bayesian analysis, particularly in early-phase studies, thus shaping future trends in medical device trials.
A notable example is the recent approval of Adaptimmune’s Tecelra (afamitresgene autoleucel) in August 2024 for treating certain forms of synovial sarcoma, which exemplifies the advancements in the medical device sector that are influencing study designs. Furthermore, the shift towards decentralized and hybrid study models is reshaping future trends in medical device trials. This approach not only fosters greater participant engagement but also addresses the logistical challenges often associated with traditional study designs.
As sponsors strive to enhance productivity and reduce costs, the adoption of innovative study designs is essential for aligning with future trends in medical device trials, ultimately increasing the pace and success rates of medical development. Recent trends indicate that sponsors are prioritizing productivity improvements in development through various strategies, which aligns with the need for enhanced efficiency in studies.
Statistics reveal that future trends in medical device trials, particularly the integration of digital health technologies, are set to revolutionize research with a notable increase in the use of telehealth and remote monitoring tools. These advancements not only streamline data management but also enhance care by providing more accessible and flexible participation options. An analysis focused on improving coverage and intricate study designs highlights how legislative and regulatory changes are fostering an environment conducive to innovative designs, aligning with future trends in medical device trials, particularly in addressing unmet patient needs.
As the medical device sector continues to evolve, staying attuned to future trends in medical device trials will be crucial for researchers aiming to conduct successful experiments. With bioaccess® offering comprehensive trial management services—including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting—researchers can effectively navigate the complexities of trials in Latin America. Richard Staines, account director at Optimum Strategic Communications, notes that the research environment is rapidly changing, and adapting to these shifts will be essential for achieving success.
The Rise of Digital Therapeutics in Clinical Trials
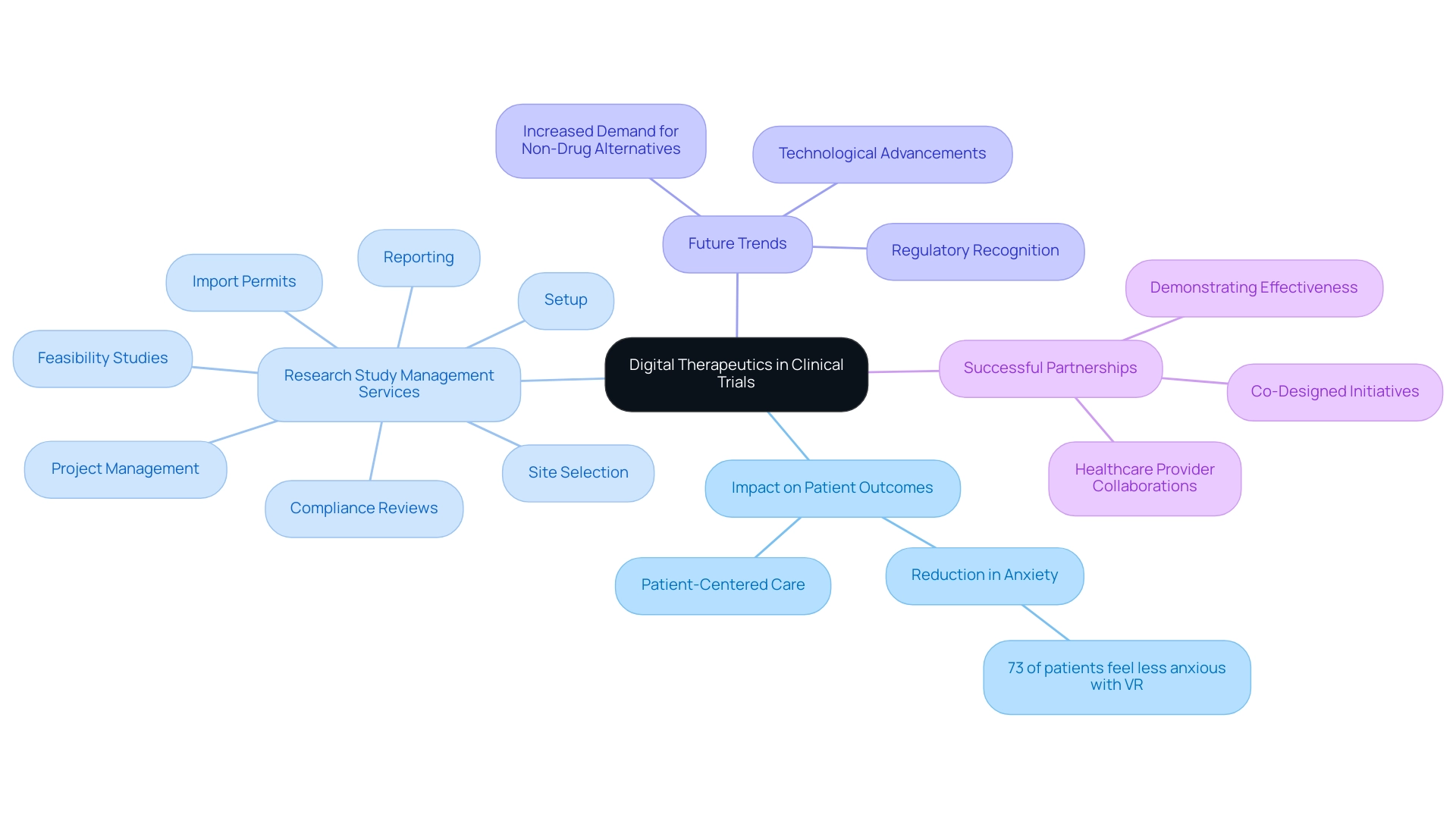
Digital therapeutics (DTx) represent a transformative approach in the field of medical research, offering software-based interventions aimed at preventing, managing, or treating various medical conditions. Their incorporation into research studies is progressively acknowledged as vital, fueled by the need for creative solutions that greatly improve health outcomes. In 2025, future trends in medical device trials are anticipated to advance further, with DTx playing a crucial role in enhancing patient compliance and allowing real-time tracking of progress. This capability not only enriches the data collected on treatment efficacy but also fosters a more patient-centered approach to care.
To support these advancements, extensive research study management services are essential. bioaccess offers a variety of capabilities, including:
- Feasibility studies
- Site selection
- Compliance reviews
- Setup
- Import permits
- Project management
- Reporting
These services guarantee that medical studies are carried out efficiently and in accordance with regulatory standards, ultimately aiding the success of DTx integration.
Recent statistics indicate that 73% of individuals reported feeling less anxious when using virtual reality (VR) headsets during surgical procedures. This statistic highlights the potential of DTx to positively affect patient experiences and outcomes, demonstrating how such technologies can improve the overall research experience. As regulatory bodies continue to recognize DTx as valid therapeutic options, researchers must understand their implications for study design and execution, particularly in light of future trends in medical device trials. This comprehension is vital for utilizing DTx to improve the quality of future trends in medical device trials.
Expert opinions highlight a trend where sponsors are increasingly demanding technology that facilitates direct access to live data while still outsourcing certain functions. This dual approach enables a more hands-on management of clinical trials, ensuring high-quality data delivery and enhanced care. As noted by the Head of Clinical Data Engineering, "Traditionally, data management was outsourced to our CRO vendor partners. Part of the initiative is to bring all our research in-house so that our internal teams can start working on it. They can be more involved, and we implement studies internally and we are able to take control of our data, and we provide for those we serve with high quality." This perspective highlights the significance of incorporating DTx into healthcare workflows.
Moreover, successful examples of DTx integration into healthcare workflows demonstrate that partnerships between DTx companies and healthcare providers can lead to co-designed initiatives that address future trends in medical device trials and real-world medical challenges, ultimately resulting in better patient outcomes. The case analysis titled 'The Future Of DTx And Healthcare Provider Partnerships' demonstrates how these collaborations can improve the effectiveness of DTx tools in medical environments.
As the digital therapeutics market continues to grow, fueled by the need for non-drug alternatives and technological progress, future trends in medical device trials emphasize the importance of integrating DTx into research studies. By adopting these innovations along with thorough research management services, researchers can greatly improve the effectiveness of their studies and aid in the development of medical devices that enhance lives.
Artificial Intelligence: Transforming Medical Device Trials
Artificial Intelligence (AI) is revolutionizing medical device testing by significantly enhancing both efficiency and accuracy. By leveraging advanced algorithms, AI can swiftly analyze extensive datasets, uncovering patterns and predicting outcomes that traditionally required considerable time and resources. This ability not only accelerates data examination but also streamlines the overall research process.
A critical application of AI in medical studies lies in its role in enhancing participant recruitment. AI systems can sift through historical data and real-time analytics to identify the most suitable candidates for studies, thereby improving recruitment rates and ensuring a more representative sample. As the landscape of medical studies evolves, the integration of AI-driven tools for monitoring patient adherence and engagement is becoming increasingly vital.
These tools can automate routine tasks, allowing researchers to focus on more complex aspects of trial management. Future trends in medical device trials suggest that the influence of AI in clinical trials is expected to grow even more pronounced by 2025. With the global artificial intelligence in drug discovery market projected to reach USD 12.8 billion by 2032, a compound annual growth rate (CAGR) of 27.5% from 2023 to 2032 underscores the momentum behind AI technologies. Furthermore, a recent survey revealed that while only 25% of healthcare executives had implemented generative AI solutions as of December 2023, there is strong optimism about AI's potential to enhance healthcare outcomes, with 51% of U.S. adults expressing confidence in AI's ability to drive significant advancements in the sector by 2024. Case studies illustrate this trend, with healthcare leaders recognizing AI as an exciting emerging technology.
Approximately 80% of these leaders see substantial opportunities for AI to enhance efficiencies and outcomes, despite existing barriers such as trust and transparency. For AI to achieve widespread acceptance in medical device evaluations, it must demonstrate dependability and effectively improve workflows, decision-making, and patient results.
At bioaccess®, with over 20 years of experience in overseeing medical device assessments, the commitment to advancing medical devices sooner through expertise and a tailored approach aligns perfectly with these trends. By incorporating AI into their research services, bioaccess® not only promotes the effective implementation of studies but also ensures that innovative medical technologies access the market more swiftly. This is further supported by bioaccess's extensive research management services, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies.
Moreover, partnerships with organizations such as Caribbean Health Group aim to establish Barranquilla as a premier location for research studies in Latin America, supported by the assistance of Colombia's Minister of Health.
In conclusion, the incorporation of AI in medical device evaluations is not merely a trend but a groundbreaking change that aligns with future directions in medical device trials, promising to transform how medical research is conducted and ultimately leading to quicker and more efficient medical advancements.
Enhancing Patient Engagement and Diversity in Trials
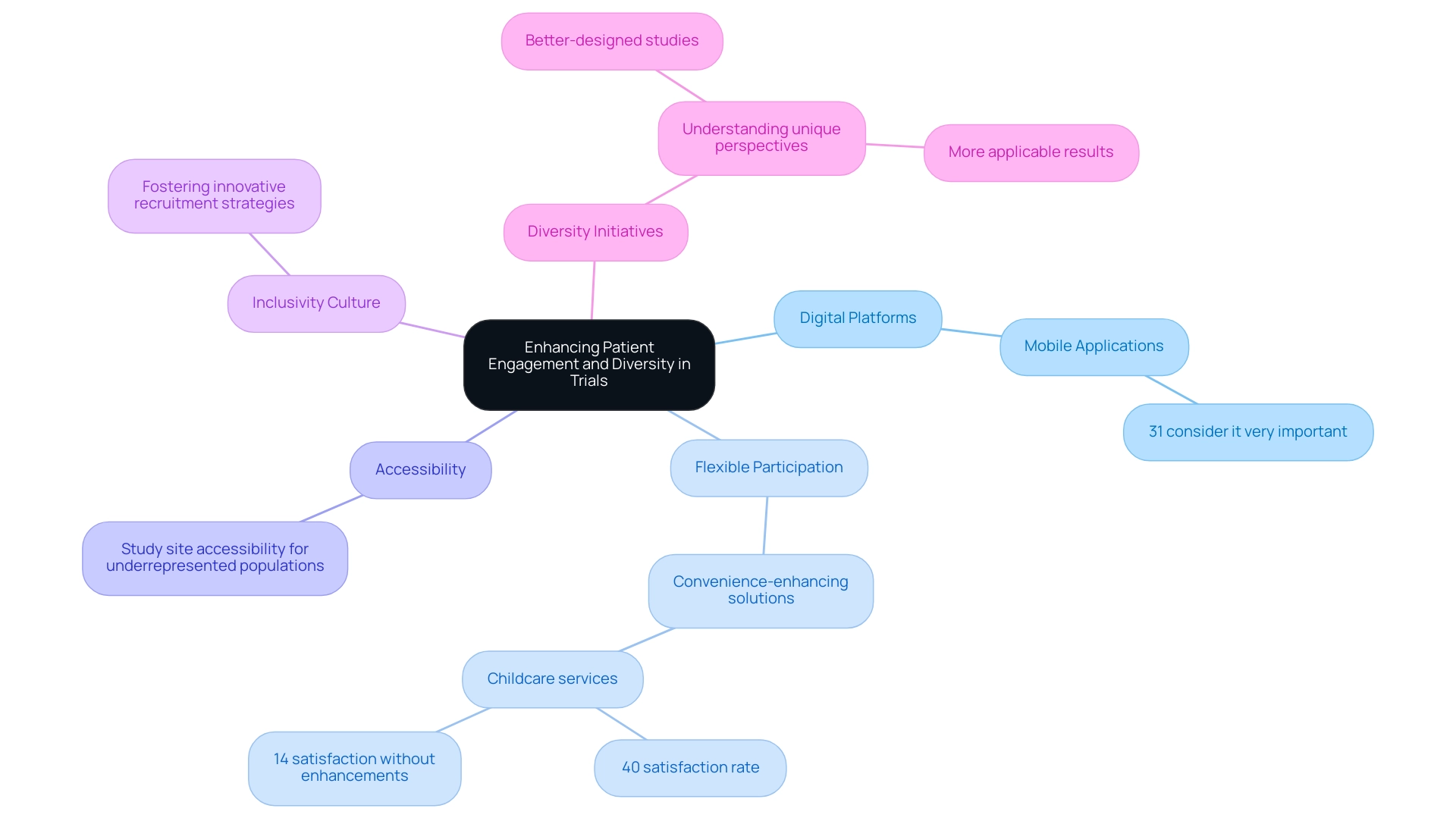
Patient involvement and diversity are essential components of research studies, significantly influencing the validity and relevance of study outcomes. To enhance participant involvement, researchers must implement a multifaceted strategy that leverages digital platforms for effective communication and feedback, offers flexible participation options, and ensures accessibility of study sites for underrepresented populations. Catherine Gregor, chief officer of research studies at Florence, asserts that "to enhance patient involvement with studies and, in the process, boost recruitment and the likelihood of studies concluding successfully, sponsors, research sites, and other stakeholders must combine remote technology with addressing participants’ concerns when developing protocols."
This assertion highlights the necessity of integrating technology into recruitment strategies, as statistics from 2025 reveal that 31% of respondents consider mobile applications to be 'very important' for enhancing engagement.
Furthermore, cultivating a culture of inclusivity within research teams can foster innovative recruitment and retention strategies. For instance, longitudinal research conducted since 2013 indicates that convenience-enhancing solutions, such as childcare services, significantly improved participant satisfaction—40% of those offered such solutions reported that their experience exceeded expectations, compared to only 14% of those without enhancements. This finding underscores the importance of addressing participant needs to bolster engagement and aligns with bioaccess®'s expertise in providing customized strategies to accelerate medical devices through comprehensive clinical management services, including feasibility studies, site selection, and compliance reviews.
Incorporating diversity initiatives in medical device studies transcends regulatory requirements; it serves as a pathway to more comprehensive data. Expert opinions highlight that understanding the unique perspectives of diverse populations can lead to better-designed studies and more applicable results. By implementing these strategies, healthcare researchers can enhance participant involvement, ultimately yielding more reliable data and improved outcomes in the evolving landscape of medical device evaluations, especially considering future trends in medical device trials, supported by bioaccess®'s expedited study services in Latin America.
Wearable Technologies and Remote Monitoring in Clinical Trials
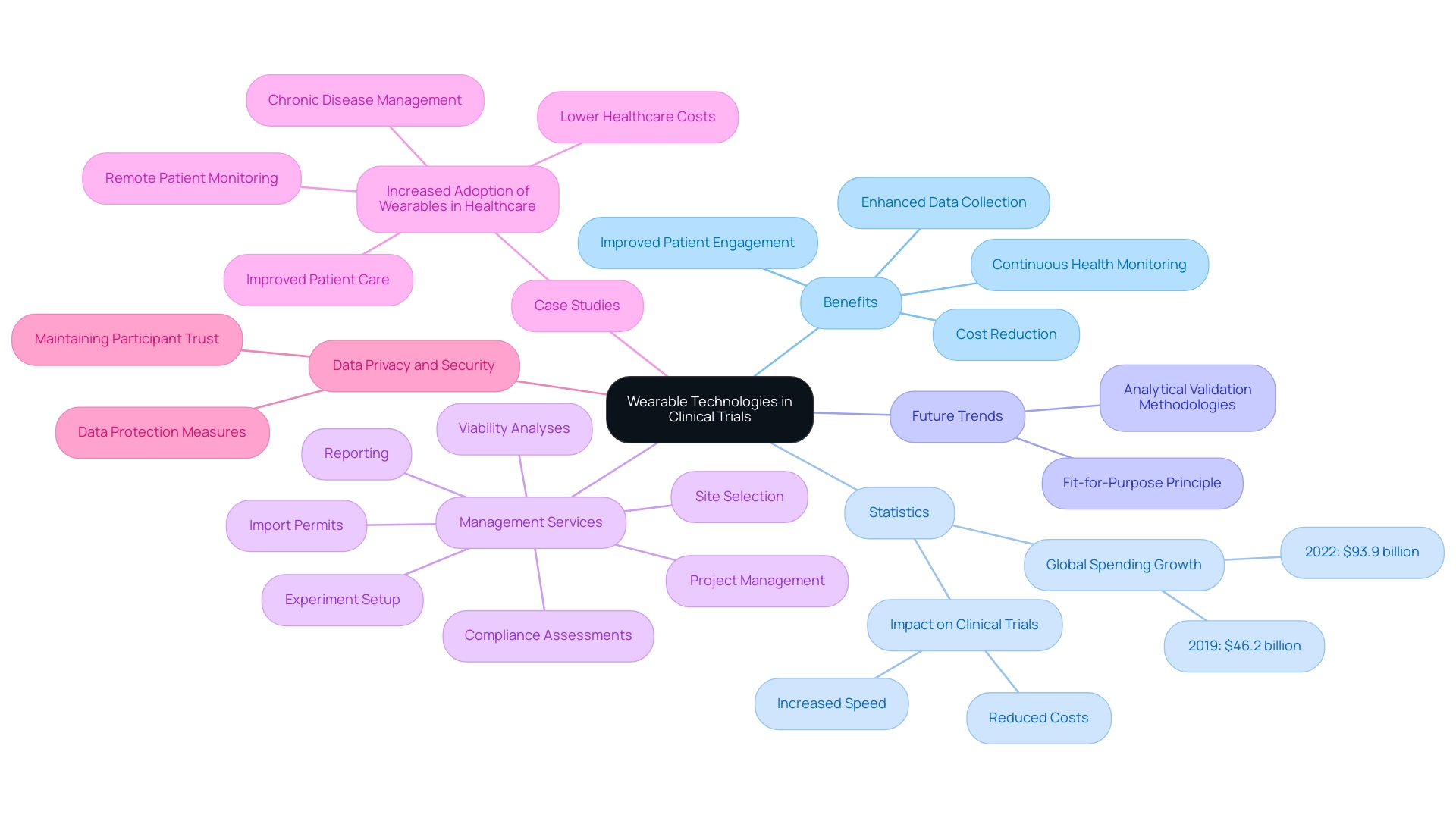
Wearable technologies are revolutionizing the landscape of research studies by facilitating continuous observation of individuals' health metrics outside of traditional medical settings. These devices adeptly track vital signs, medication adherence, and symptom progression, providing researchers with real-time data that significantly enhances the understanding of treatment effects. For instance, global end-user spending on wearable devices surged from $46.2 billion in 2019 to nearly $93.9 billion in 2022, reflecting an increasing reliance on these technologies in healthcare.
Moreover, wearable technology can dramatically accelerate the pace of clinical studies while simultaneously reducing the cost of data acquisition, positioning it as a valuable asset in clinical research. The integration of wearables into trial designs not only boosts participant engagement and retention but also allows individuals to remain in their natural environments, leading to more accurate and relevant data collection. A notable case study illustrates the growing adoption of wearables in healthcare, particularly for remote individual monitoring and chronic disease management. This trend has been shown to improve patient care while concurrently lowering healthcare costs, underscoring the potential of wearables in the medical field.
As we look toward 2025, future trends in medical device trials are expected to amplify the impact of wearable technologies on research studies, with an emphasis on developing analytical validation methodologies and adhering to the fit-for-purpose principle, both of which are crucial for future success. Statistics indicate that remote monitoring can significantly enhance research data collection, with studies demonstrating that participants utilizing wearable devices report higher levels of engagement and adherence to study protocols. Tajammul Pangarkar, CMO at Prudour Pvt Ltd, emphasizes the importance of understanding technology trends, stating, "The incorporation of wearable technology into medical studies is not merely a trend; it’s essential for enhancing patient care and research effectiveness."
To effectively harness the potential of wearable technologies, comprehensive management services for studies, such as those offered by bioaccess, play a pivotal role. These services encompass:
- Viability analyses
- Site selection
- Compliance assessments
- Experiment setup
- Import permits
- Project management
- Reporting
These services ensure that experiments are conducted efficiently and in accordance with regulatory standards. Furthermore, the impact of Medtech trials extends beyond individual experiments, supporting local economies through job creation, economic development, and improved healthcare outcomes, while fostering international collaboration.
However, researchers must remain vigilant regarding data privacy and security implications when implementing wearable technologies in their studies. Ensuring robust data protection measures will be critical to maintaining participant trust and the integrity of clinical research outcomes.
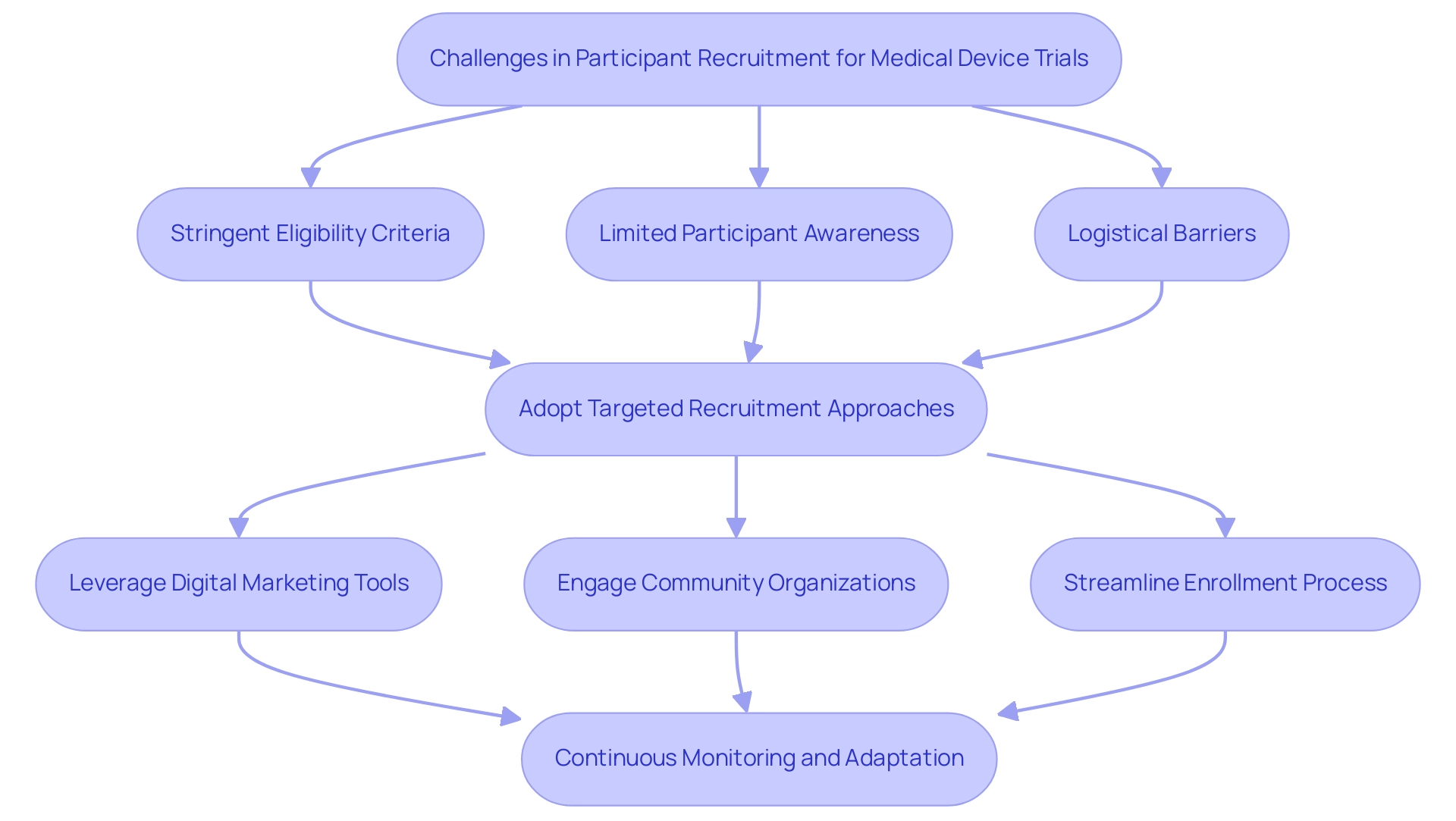
Challenges in Participant Recruitment for Medical Device Trials
Recruiting participants for medical device studies presents numerous challenges, including stringent eligibility criteria, limited participant awareness, and significant logistical barriers. Notably, only 55% of studies achieve their originally specified recruitment targets, underscoring the urgent need for more effective strategies. To navigate these hurdles, researchers must adopt targeted recruitment approaches that leverage digital marketing tools, engage with community organizations, and streamline the enrollment process.
Effective communication is paramount; providing clear information about the study's purpose, potential benefits, and associated risks fosters trust and encourages participation. For instance, a recent case analysis illustrated the success of a multimodal recruitment strategy that combined in-person recruitment, advertising, direct referrals, and community service events. This approach not only heightened awareness but also demonstrated that in-person recruitment was the most efficient and cost-effective method, highlighting the value of diverse tactics.
Community engagement significantly contributed to this success, as it effectively attracted potential participants. Additionally, addressing the financial burdens associated with participation—such as time off work and childcare—can markedly enhance enrollment rates. Logistical obstacles often hinder involvement, particularly when studies are conducted in specific medical facilities requiring long-distance travel; thus, simplifying access to studies is essential. Continuous monitoring of recruitment efforts and adapting strategies based on real-time feedback can further bolster enrollment success.
At bioaccess, we understand the intricacies of research management, offering extensive services that encompass feasibility studies, site selection, compliance evaluations, setup, import permits, project oversight, and reporting. These services are designed to directly address the challenges of participant recruitment by ensuring studies are established efficiently and that potential participants are well-informed and engaged. As Nicola Armitstead, Vice President of Site Clinical Operations, asserts, "With a focus on individualized care and adaptive solutions, we continuously redefine the standards of participant recruitment and retention in research."
By concentrating on these principles, researchers can reshape participant recruitment and retention criteria in medical studies, ultimately leading to more successful experiments.
Innovative Clinical Trial Designs: Adaptive and Decentralized Approaches
Creative research designs, particularly adaptive and decentralized methods, are gaining significant traction in the medical device sector. With over 20 years of experience in the Medtech industry, bioaccess® is exceptionally positioned to support these advancements through its comprehensive research management services, which include:
- Early-Feasibility Studies (EFS)
- First-In-Human Studies (FIH)
- Pilot Studies
- Pivotal Studies
- Post-Market Follow-Up Studies (PMCF)
Adaptive studies are characterized by their ability to incorporate adjustments throughout the research process based on interim findings, thereby greatly enhancing both flexibility and efficiency. This dynamic approach empowers researchers to make informed decisions that can lead to improved outcomes and optimized resource utilization.
Conversely, decentralized studies leverage technology to facilitate remote research, minimizing the need for participants to travel to research sites. This strategy not only streamlines the recruitment process but also enhances retention by making participation more convenient and accessible. Recent statistics indicate that decentralized methods have led to a notable increase in patient enrollment rates, with some studies reporting up to a 30% improvement in recruitment efficiency.
As we move closer to 2025, the integration of adaptive research designs is expected to continue evolving, with a growing emphasis on patient-reported outcomes (Pros) to enhance decision-making and promote patient-centered care. Christina Yap, a Professor of Clinical Trials Biostatistics, underscores the significance of incorporating Pros in early-phase studies to foster patient-centered care. Expert opinions suggest that these designs will be pivotal in addressing the complexities of medical device evaluations, particularly in the context of multi-regional studies (MRCT), which are anticipated to align with forthcoming trends in medical device trials.
However, researchers must remain vigilant regarding the regulatory implications associated with these innovative designs. Ensuring compliance with evolving guidelines is crucial for the successful implementation of adaptive and decentralized studies. By adequately preparing and comprehending the regulatory landscape, researchers can harness these innovative approaches to advance medical devices more effectively, ultimately benefiting both the industry and patients alike.
Moreover, foundational knowledge in statistics is essential for engaging with these study designs, as emphasized by the optional introduction course on statistics available to participants who lack basic training. This preparation equips researchers to navigate the complexities of adaptive and decentralized studies, supported by bioaccess®'s expertise in feasibility assessments, site selection, setup, monitoring, and reporting.
Key Takeaways for Clinical Researchers: Staying Ahead of Trends
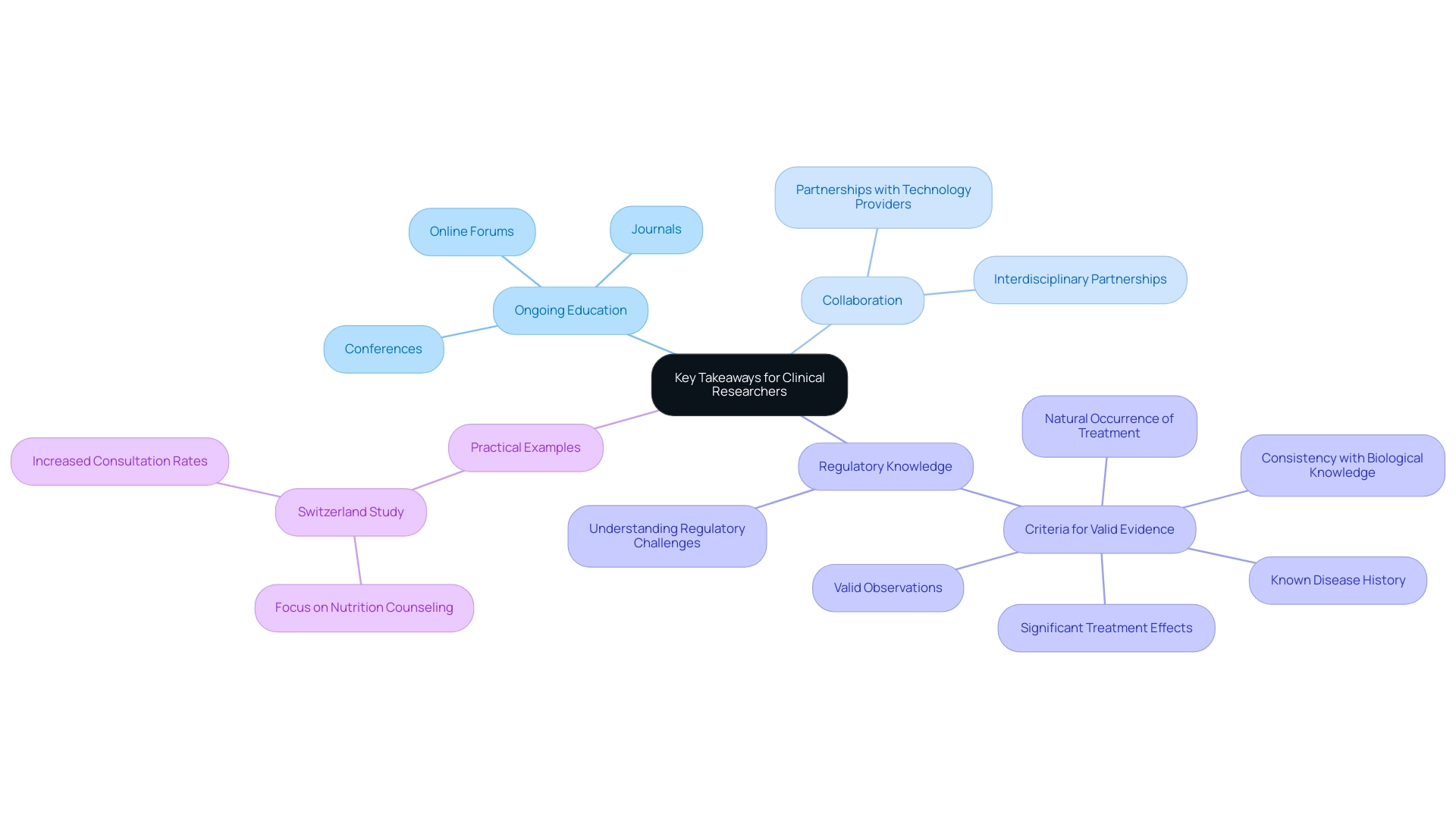
To remain at the forefront of future trends in medical device trials, researchers must prioritize ongoing education and professional development. Participating in industry conferences is particularly influential; these events not only offer networking opportunities but also promote the sharing of knowledge on the latest advancements in research methodologies. Research indicates that involvement in such conferences significantly enhances researchers' comprehension of emerging technologies, with many noting improved outcomes as a result.
Notably, a USA study revealed that the follow-up period was just one month, underscoring the importance of timely follow-up in clinical research. Subscribing to relevant journals and engaging in online forums further enriches researchers' insights into innovative practices and regulatory updates. The significance of continuous education is underscored by recent statistics, which show that researchers who actively pursue ongoing training are more likely to implement successful study designs and methodologies. Bioaccess® is dedicated to advancing medical devices sooner through its expertise and customized approach, emphasizing the necessity of understanding future trends in medical device trials and the imperative for continuous learning in this field. Furthermore, fostering collaborations with technology providers and regulatory experts is essential.
These partnerships can streamline the navigation of the complex regulatory landscape in Latin America, ensuring that trials are not only compliant but also strategically positioned for success. A notable example is the Switzerland Study, which involved 424 patients and primary care physicians focusing on nutrition counseling. This research aimed to increase the number of consultations that included nutritional advice, demonstrating how collaborative efforts can lead to enhanced consultation rates and showcasing the potential benefits of interdisciplinary partnerships.
As discussed by Steve Garchow in the LATAM Medtech Leaders Podcast, understanding the unique regulatory challenges in Latin America is vital for successful market access. By consciously strengthening knowledge in these areas, researchers can elevate their careers and contribute to future trends in medical device trials within the region. Moreover, grasping the criteria for valid evidence in non-experimental comparative studies—such as the natural occurrence of treatment, valid observations, known disease history, significant treatment effects, and consistency with biological knowledge—further emphasizes the significance of collaboration and adherence in medical studies.
Through its comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting, bioaccess® is leading the way in Medtech clinical research in Latin America, with a focus on innovation and regulatory excellence.
Conclusion
The medical device trial landscape is rapidly evolving, shaped by advancements in technology, regulatory shifts, and an increasing focus on patient-centric methodologies. The integration of digital health technologies, adaptive trial designs, and innovative strategies—such as wearable devices and digital therapeutics—are fundamentally transforming the conduct of clinical research. These advancements not only streamline trial processes but also enhance patient engagement, optimize recruitment, and improve overall outcomes.
As we approach 2025, the significance of embracing these transformative trends cannot be overstated. The transition towards decentralized trials and the utilization of artificial intelligence in recruitment and data management are crucial for navigating the complexities of modern clinical research. By adapting to these changes, researchers can ensure their trials are efficient, inclusive, and capable of delivering meaningful results.
Ultimately, remaining informed about the latest advancements and actively engaging in ongoing education and collaboration will be essential for clinical researchers. By leveraging innovative trial designs and technologies, researchers can significantly contribute to the success of medical device trials, ultimately leading to improved patient care and outcomes. Embracing this dynamic landscape will not only enhance the quality of clinical research but also position researchers as leaders in the pursuit of effective medical solutions.
Frequently Asked Questions
What are the key trends shaping the future of medical device trials?
Key trends include the integration of digital health technologies for improved data collection and monitoring, a focus on participant-centered approaches, and the adoption of adaptive study designs that allow real-time modifications based on data and patient feedback.
How will adaptive study designs impact medical device trials by 2025?
Adaptive study designs will empower researchers to adjust experimental parameters in real-time, enhancing the relevance and efficiency of investigations. The anticipated implementation of ICH E20 guidelines is expected to promote the use of complex study elements like Bayesian analysis, particularly in early-phase studies.
Can you provide an example of a recent advancement in the medical device sector?
A notable example is the approval of Adaptimmune’s Tecelra (afamitresgene autoleucel) in August 2024 for treating certain forms of synovial sarcoma, showcasing advancements that influence study designs in the medical device sector.
What is the significance of decentralized and hybrid study models in medical device trials?
Decentralized and hybrid study models enhance participant engagement and address logistical challenges associated with traditional study designs, reshaping trends in medical device trials.
How are sponsors adapting to improve productivity in medical device trials?
Sponsors are adopting innovative study designs to enhance productivity and reduce costs, aligning with future trends to increase the pace and success rates of medical development.
What role do digital health technologies play in future medical device trials?
Digital health technologies, including telehealth and remote monitoring tools, are set to revolutionize research by streamlining data management and providing more accessible and flexible participation options.
Why is it important for researchers to stay updated on future trends in medical device trials?
Staying informed about future trends is crucial for researchers to conduct successful experiments and effectively navigate the evolving landscape of medical device trials.
What services does bioaccess offer to support medical research studies?
bioaccess provides comprehensive trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting.
How are digital therapeutics (DTx) influencing medical device trials?
DTx are becoming vital in research studies by enhancing patient compliance, allowing real-time tracking of progress, and improving health outcomes, thus enriching the data collected on treatment efficacy.
What is the impact of partnerships between DTx companies and healthcare providers?
Collaborations between DTx companies and healthcare providers can lead to co-designed initiatives that address medical challenges and improve patient outcomes, highlighting the effectiveness of DTx tools in clinical settings.




