Overview
This article examines the pivotal role of ANVISA, Brazil's regulatory authority for clinical trials, in shaping the design and execution of clinical research studies. It underscores that ANVISA enforces rigorous guidelines and requirements, including the necessity for comprehensive clinical trial applications and strict adherence to ethical practices. These measures not only safeguard participant safety and ensure compliance but also cultivate an environment that promotes innovative medical research.
Introduction
In the dynamic landscape of clinical research, Brazil's regulatory framework is pivotal in shaping trial conduct, ensuring that innovation aligns with stringent safety and ethical standards. At the forefront of this regulatory oversight is ANVISA, the Brazilian Health Regulatory Agency, tasked with the critical mission of safeguarding public health while facilitating the advancement of medical technology. Recent legislative changes and updates to regulatory guidelines present researchers with both opportunities and challenges in navigating this complex environment.
From expedited approval processes to the necessity of comprehensive ethical reviews, understanding ANVISA's requirements is essential for successful clinical trials. As Brazil positions itself as a burgeoning hub for medical research, insights into ANVISA's evolving role and the implications of these regulations will be invaluable for stakeholders aiming to drive innovation in the healthcare sector.
Overview of ANVISA: Brazil's Regulatory Authority for Clinical Trials
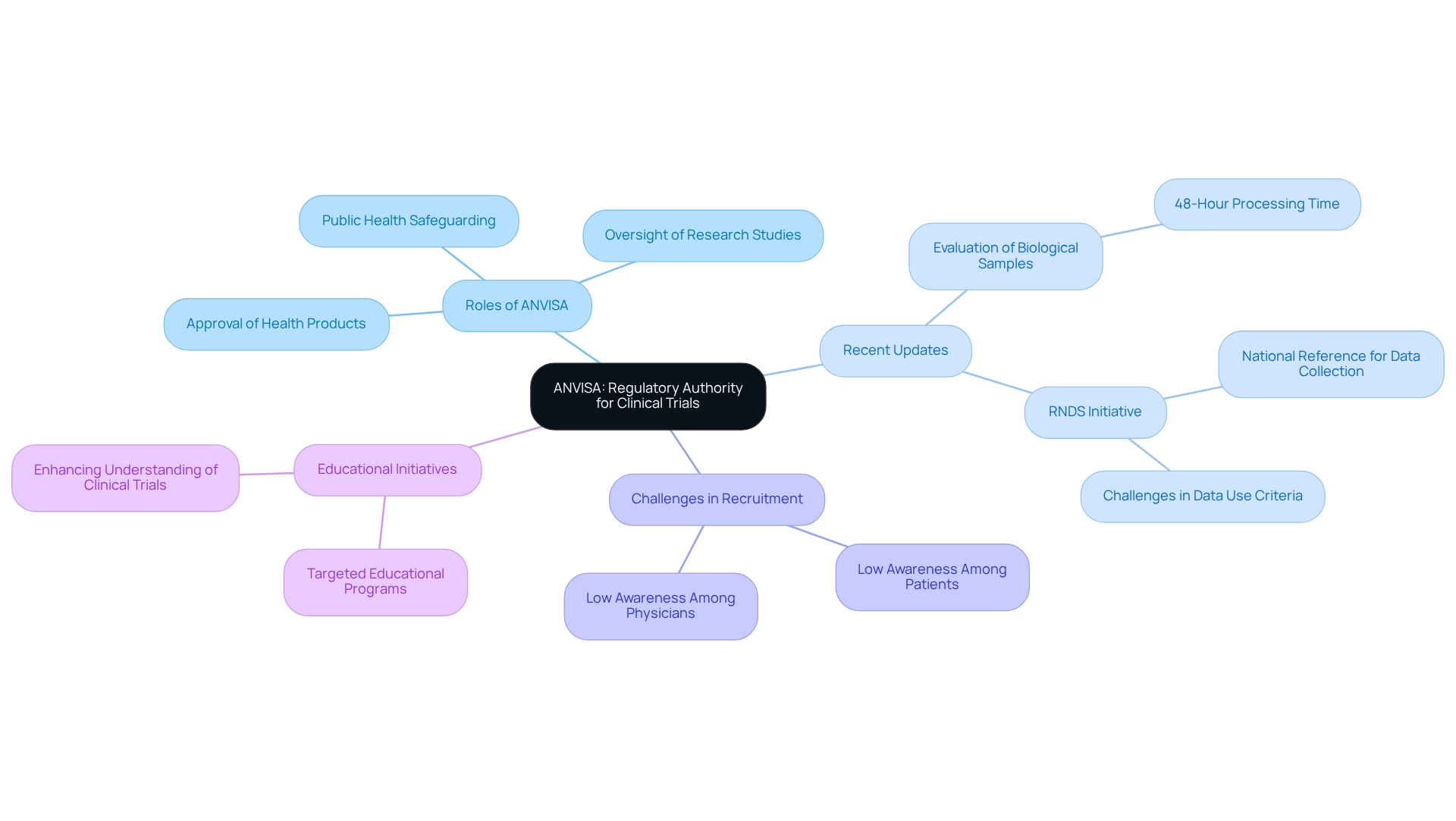
The Brazilian Health Regulatory Agency (ANVISA) is integral to the regulation and oversight of health products, including drugs and medical devices. Established to guarantee the safety and effectiveness of these products, ANVISA plays a crucial role in the approval and oversight of research studies throughout Brazil. Its regulations are meticulously crafted to safeguard public health while promoting the advancement of innovative medical solutions.
Recent updates indicate that ANVISA will evaluate and distribute human biological samples intended for research within 48 hours of their arrival in Brazil, provided all legal requirements are fulfilled. This accelerated process is expected to significantly enhance the efficiency of medical studies, particularly for multicenter research, where the coordinating center investigator must prepare and submit consolidated Serious Adverse Event (SAE) reports to the ethics committee.
The establishment of the RNDS (National Health Data Network) is anticipated to serve as a national reference for healthcare data collection. However, challenges persist in establishing data use criteria for compliance purposes, underscoring the significance of this initiative in enhancing approval rates for research studies and ensuring that data gathering aligns with compliance standards.
Despite these advancements, a survey revealed a concerning lack of awareness among both patients and doctors regarding the advantages of research, contributing to low recruitment levels for studies in Brazil. The findings indicate that focused educational programs are essential to enhance awareness and comprehension of research studies, potentially leading to higher participation rates among patients and increased involvement from doctors.
In conclusion, understanding how ANVISA impacts clinical trial design is vital for researchers and sponsors intending to conduct studies in Brazil, as it establishes benchmarks for compliance and ethical behavior in research. The agency's evolving role and policy updates are crucial for comprehending how ANVISA influences clinical trial design in the realm of medical research. With bioaccess®'s expertise in managing extensive research services, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies, researchers can navigate these regulations effectively.
Furthermore, bioaccess® is committed to addressing the challenges of low recruitment through educational initiatives and tailored support, ensuring successful outcomes and adherence to compliance standards.
Key ANVISA Requirements for Clinical Trials in Brazil
Carrying out clinical studies in Brazil necessitates strict adherence to several key stipulations established by the regulatory agency, illustrating the significant impact of ANVISA on clinical trial design to ensure compliance and participant safety. These requirements include:
- Submission of a Clinical Trial Application (CTA): Researchers must submit a comprehensive CTA that encompasses detailed information about the study protocol, informed consent forms, and the qualifications of the investigators involved. This submission is crucial for the initial assessment of the project's feasibility and ethical considerations. At bioaccess®, we leverage over 20 years of Medtech expertise to assist in the preparation and submission of CTAs, underscoring how ANVISA influences clinical trial design to ensure that all necessary documentation meets the agency's stringent standards.
- Ethical Approval: Before initiating any study, researchers must obtain approval from local ethics committees (CEPs). In certain instances, additional approval from the National Commission for Ethics in Research (CONEP) is also mandated. Notably, CONEP prohibits research ethics committees (ECs) from imposing fees for evaluating research protocols, clarifying the ethical approval process. This multi-tiered ethical review process is designed to safeguard participant rights and welfare, reflecting Brazil's commitment to ethical research practices. Bioaccess® provides comprehensive support in navigating these ethical requirements, ensuring timely approvals to facilitate study initiation.
- Adherence to Good Clinical Practices (GCP): All clinical studies must comply with GCP guidelines, which are essential for maintaining the integrity of the data gathered and the safety of participants. Understanding the influence of ANVISA on clinical trial design is not only a regulatory requirement but also a fundamental aspect of conducting high-quality research. Our team at bioaccess® is dedicated to maintaining GCP compliance throughout the research process, enhancing the reliability of study outcomes.
- Reporting of Adverse Events: Researchers are mandated to promptly report any adverse events to the regulatory agency. This requirement is vital for maintaining participant safety and ensuring ongoing regulatory compliance throughout the study's duration. Bioaccess® guarantees that all negative occurrences are meticulously recorded and reported in accordance with regulatory standards, thereby protecting participant welfare.
As we look ahead to 2025, the approval procedure for medical studies in Brazil continues to evolve, demonstrating how ANVISA impacts clinical trial design by emphasizing the importance of timely ethical assessments. Recent statistics indicate that a notable percentage of research study applications receive approval, further illustrating ANVISA's commitment to promoting research while upholding strict ethical standards. Jarbas Barbosa, the current head of the agency, has remarked that "the centralization of analysis by the CEP/Conep System needs to be evaluated and takes as a reference the establishment of the deadline for analysis response, akin to that set by the oversight body by RDC 9/2015."
This highlights ANVISA's ongoing improvements in the approval process.
Case studies exemplify the effectiveness of Brazil's ethical approval process. For instance, the endorsement and oversight of medical studies in Brazil illustrate how ANVISA impacts clinical trial design, as they involve thorough assessments by ethical and governing bodies, ensuring adherence to established standards. Despite some claims from researchers that the approval process may impede new studies, ongoing enhancements in Brazilian standards reflect ANVISA's commitment to refining procedures while maintaining high-quality ethical evaluations.
Moreover, entities such as bioaccess® have effectively conducted research for regulatory submission, offering guidance for clinical device development initiatives and further demonstrating the practical elements of compliance. Additionally, bioaccess® specializes in various types of studies, including Early-Feasibility Studies (EFS) and First-In-Human Studies (FIH), providing a customized approach to trial management to ensure that each study is tailored to meet specific regulatory and participant needs.
The Role of Bioequivalence Studies in ANVISA's Approval Process
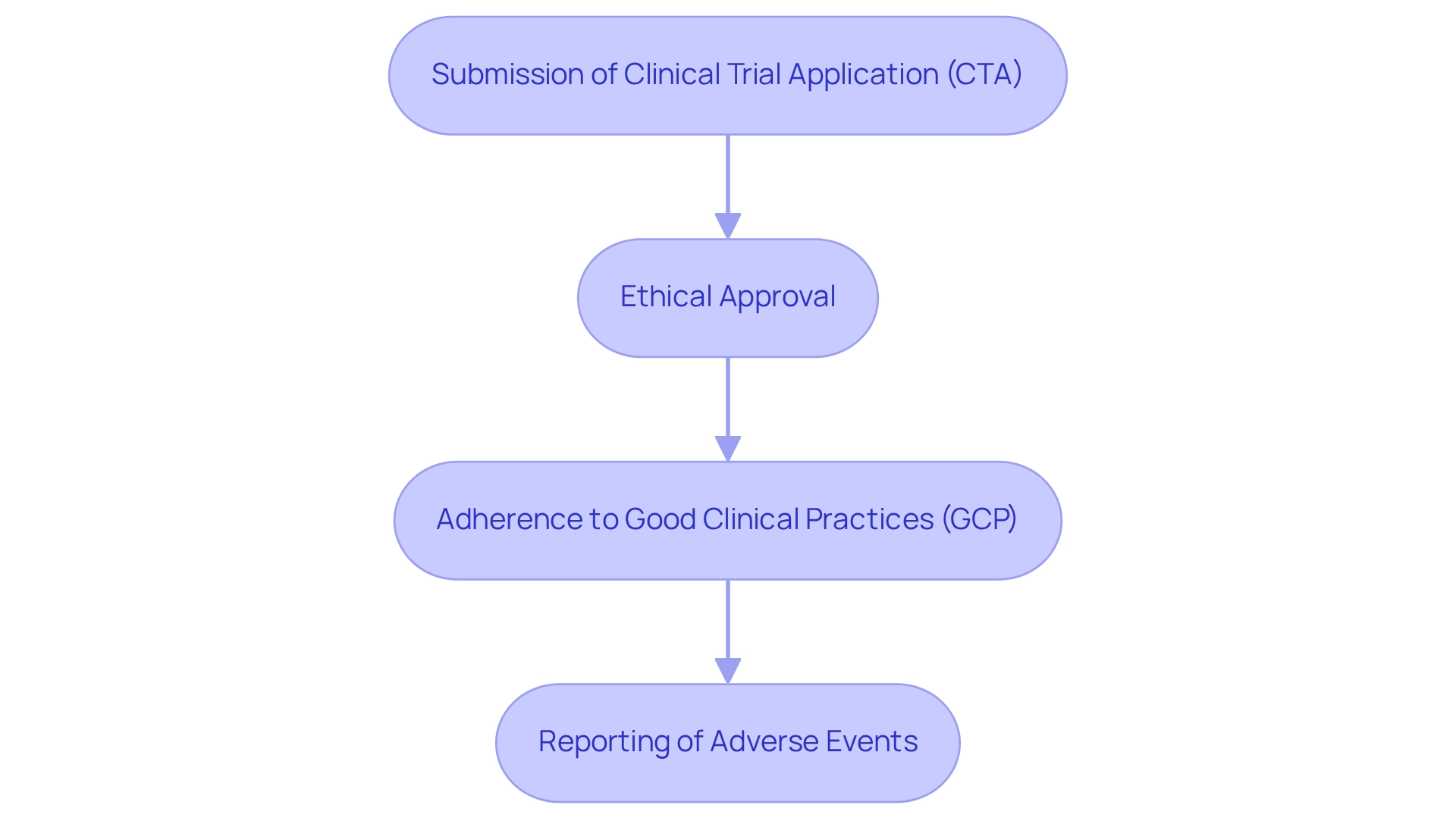
Bioequivalence studies are crucial in the approval process for generic medications in Brazil, as mandated by the regulatory agency. These studies aim to demonstrate that a generic product is therapeutically equivalent to its branded counterpart, ensuring both products exhibit similar pharmacokinetic profiles and therapeutic effects in the body. The following key aspects are essential for compliance with ANVISA's stringent guidelines:
- Study Design: The structure of bioequivalence studies must be meticulously planned to comply with regulatory standards. This includes determining appropriate sample sizes and employing robust statistical analyses to validate the findings.
- Regulatory Submission: The results of bioequivalence studies are a critical component of the Clinical Trial Application (CTA) submitted to ANVISA. Thorough documentation of the study's methodology and outcomes is essential for review and approval. As emphasized by Negar Gharavi, Senior Director of Medical Writing & Regulatory Affairs, "BioPharma Services can assist in guiding your next drug development project from concept to clinic," highlighting the significance of expert advice in managing regulatory submissions.
- Compliance with GCP: Adhering to Good Clinical Practice (GCP) standards is essential for all research studies, including bioequivalence examinations. This compliance ensures the integrity of the data collected and the safety of participants involved in the research.
At bioaccess, we offer comprehensive clinical trial management services that encompass feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting. Our expertise, especially through professionals like Katherine Ruiz, who focuses on compliance matters for medical devices and in vitro diagnostics in Colombia, ensures that our clients navigate the intricacies of ANVISA's guidelines effectively.
In 2025, the significance of bioequivalence studies in Brazil is anticipated to rise, driven by a growing demand for generic medications and the necessity for strict oversight. The U.S. FDA's endorsement of 917 Abbreviated New Drug Applications (ANDAs) in 2022 indicates a growing trend in compliance that is likely to influence Brazil's market as well. Additionally, the case study titled "Strategic Initiatives in Bioequivalence Studies" illustrates how major players in the market are focusing on inorganic growth strategies, such as mergers and partnerships, to enhance their presence.
As the market evolves, it will be essential for stakeholders to understand how ANVISA impacts clinical trial design and the intricacies of effective bioequivalence study designs to navigate the complexities of drug development in this region. Furthermore, the anticipated growth in the Asia Pacific region's generics market, projected to register a CAGR of 8.8%, underscores the global trend towards increased demand for bioequivalence studies, further contextualizing their importance in Brazil.
Navigating Regulatory Challenges in Clinical Trials with ANVISA
Navigating the oversight framework of health authorities presents various challenges for researchers in Brazil, particularly in the realm of medical studies. Key issues include:
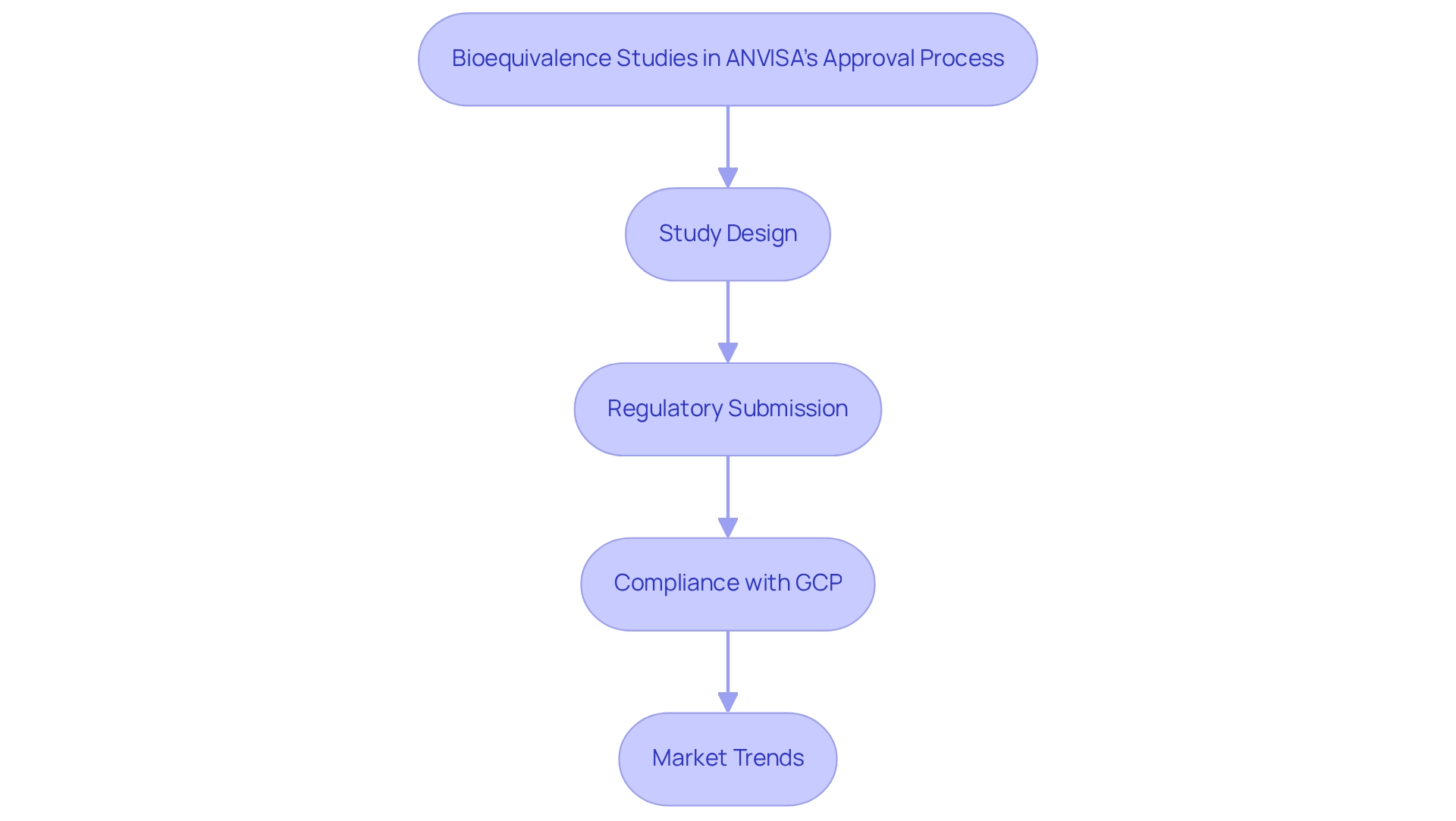
- Lengthy Approval Processes: The approval timeline for clinical study applications by the agency can often exceed 90 days, resulting in significant delays in research timelines. To mitigate this, researchers should incorporate buffer periods into their project plans, allowing for potential setbacks in the approval process. The implementation of accelerated review procedures for COVID-19 research protocols on February 27, 2020, exemplifies how changes in oversight can influence study timelines and methodologies.
- Complex Documentation Requirements: The submission process necessitates extensive and detailed documentation, which can be daunting. Researchers must ensure that all necessary documents are meticulously prepared and submitted in accordance with the regulatory body's guidelines. This attention to detail is crucial for facilitating a smoother approval process and avoiding unnecessary rejections or requests for additional information.
- Changing Guidelines: The evolving guidelines underscore how ANVISA impacts clinical trial design, as its oversight framework is subject to frequent updates that can affect both ongoing and future clinical studies. Staying abreast of these changes is vital for maintaining compliance and ensuring that research protocols align with the latest requirements. The fast-tracked review procedures for COVID-19 research protocols implemented in February 2020 illustrate ANVISA's influence on clinical trial design by shaping study timelines and methodologies.
- Common Regulatory Challenges: Researchers frequently encounter obstacles such as the requirement for agreements between sponsors or CROs and involved institutions, along with the obligation to promptly notify all parties of any early termination or suspension of a study. The necessity for the sponsor or CRO to sign an agreement with participating institutions and investigators highlights the importance of formal agreements in the regulatory process. These requirements emphasize the significance of clear communication and robust planning throughout the lifecycle of the experiment.
- Average Approval Time: As of 2025, the average approval duration for research studies through the regulatory agency remains a critical consideration for researchers. Understanding this timeline can assist in planning project timelines and resource allocation efficiently.
By proactively addressing these challenges, researchers can enhance their likelihood of successfully navigating the regulatory environment, ultimately fostering the advancement of cutting-edge medical technologies in Brazil. With over 20 years of experience in Medtech, bioaccess® exemplifies how a leading organization effectively connects innovative Medtech companies with opportunities for conducting research in Latin America. bioaccess® offers a tailored approach to overseeing research studies, ensuring adaptability and adherence to regulatory guidelines while emphasizing data security and client confidence.
With expertise in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies, bioaccess® is committed to accelerating the research process and advancing the development of medical devices.
Designing Clinical Trials: ANVISA's Influence on Methodology and Sample Size
The regulatory body's guidelines underscore the significant influence of ANVISA on clinical trial design, playing a vital role in shaping the framework of medical studies in Brazil. This ensures that such studies maintain high standards of scientific integrity and ethical responsibility. At bioaccess®, we leverage our extensive experience of over 20 years in managing clinical studies to adeptly navigate these guidelines. Our comprehensive clinical study management services encompass feasibility assessments, site selection, compliance reviews, study setup, import permits, project management, and reporting. Our expertise spans various study types, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF).
Key considerations include:
- Researchers must adopt methodologies that reflect ANVISA's impact on clinical trial design. This involves selecting appropriate endpoints and statistical methods that align with regulatory expectations while enhancing the reliability of trial outcomes. The emphasis on methodological soundness is essential for preserving the integrity of clinical research.
- Sample Size Determination: The regulatory agency mandates meticulous calculation of sample sizes to ensure sufficient statistical power for detecting significant differences between treatment groups. At bioaccess®, we employ established statistical methods to validate sample size choices, ensuring studies are sufficiently powered to yield meaningful outcomes. This requirement is critical for the credibility of the findings and the subsequent approval of medical devices.
- Protocol Submission: A detailed study protocol must be submitted to the regulatory agency for approval, encompassing comprehensive descriptions of the trial design, methodology, and statistical analysis plan. This submission process is crucial as it enables the agency to assess how ANVISA influences clinical trial design concerning the proposed study's adherence to standards and its potential impact on public health. Notably, inspections by the health agency are conducted within a maximum span of five working days unless justified otherwise, highlighting the efficiency of the oversight process.
In addition to these foundational components, the agency's influence extends to integrating specific protections in studies involving at-risk groups, such as women of reproductive age or expectant mothers. This commitment reflects ethical research practices and participant protection. For instance, additional safeguards are mandated to ensure awareness of risks and impacts on fertility and pregnancy.
Moreover, ANVISA's enhanced analysis process allows for the inclusion of documentation from comparable foreign oversight bodies, streamlining the assessment of research study applications. This innovation improves the efficiency of the regulatory process, facilitating quicker access to new therapies while upholding rigorous safety and efficacy standards. As noted by Carmen Penido in her article 'Avaliação de ensaios clínicos no Brasil: histórico e atualidades,' grasping these guidelines is vital for navigating the research landscape in Brazil.
Furthermore, sponsors are required to ensure access to post-study medication supply programs for research participants, including those in early-terminated studies, underscoring their ethical obligations in medical research. Overall, understanding ANVISA's impact on clinical trial design is essential for compliance with the agency's guidelines, which guarantees the successful design and implementation of research studies in Brazil. Bioaccess® is committed to leading the way in Medtech research in Latin America, emphasizing innovation and regulatory excellence.
Statistical Analysis in Clinical Trials: Meeting ANVISA's Standards
ANVISA enforces stringent statistical analysis requirements for clinical studies to uphold the integrity and validity of research findings. Key components of these requirements include:
- Statistical Methods: Researchers are mandated to utilize appropriate statistical techniques for data analysis, such as Analysis of Variance (ANOVA) and regression analysis. These methods are essential for precisely interpreting test results and ensuring that conclusions derived from the data are statistically valid.
- Analysis Plan: A comprehensive statistical analysis plan must be incorporated into the study protocol. This plan should detail the statistical methods that will be employed for data analysis and interpretation, ensuring transparency and reproducibility in the research process.
- Interim Analyses: Should interim analyses be part of the study design, they must be explicitly outlined in the protocol. This encompasses establishing the criteria for halting the study early if needed, which is crucial for safeguarding participant safety and maximizing resource allocation.
The significance of following these statistical standards cannot be overstated, as they not only improve the credibility of the results but also illustrate how ANVISA impacts clinical trial design through strict regulatory oversight. As Negar Gharavi, Senior Director of Medical Writing & Regulatory Affairs, mentions, "BioPharma Services can assist in navigating your next drug development project from concept to clinic," emphasizing the essential role of compliance in successful research studies.
Additionally, researchers need to recognize the obligation to submit a yearly progress report within 60 days of the effective date of the investigational new drug, highlighting the significance of prompt reporting in research studies. This aligns with bioaccess's commitment to thorough trial management services, including reporting on study status and adverse events.
The successful certification of bioaccess by ANVISA exemplifies adherence to these standards, highlighting how ANVISA impacts clinical trial design to ensure that research conducted in Brazil meets the highest quality benchmarks. This certification process emphasizes how ANVISA impacts clinical trial design by underscoring the importance of adhering to statistical analysis criteria, which are vital for the approval of research studies, including the import permit and nationalization of investigational devices.
As Brazil progresses in its research study environment, particularly with the introduction of new regulations effective January 1, 2025, understanding how ANVISA impacts clinical trial design and incorporating these statistical requirements will be crucial for researchers managing the intricacies of studies in the region. Furthermore, Brazil's centralized registration procedure for research ethics committees (ECs) further influences the oversight framework, requiring institutional level EC approval for each research site, coordinated by the National Research Ethics Commission (CONEP).
Safety Monitoring and Adverse Events: Compliance with ANVISA Regulations
Safety oversight is a crucial element of research studies, and the regulatory agency has established strict guidelines concerning adverse event documentation to guarantee participant safety and adherence to regulatory standards. At bioaccess®, we leverage over 20 years of expertise in managing medical device clinical trials, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), to ensure that our clients meet these critical requirements. Key components include:
- Adverse Event Reporting: Researchers are required to notify any serious adverse events to the relevant authority within specified time limits. This requirement is crucial for maintaining participant safety and ensuring that regulatory bodies are informed of any potential risks associated with the medical device under investigation. The sensitivity of passive surveillance systems for health-related events has been calculated at 71.9% for health hazards and 78.9% for seizures, underscoring the importance of timely reporting.
- Safety Monitoring Plans: A detailed safety monitoring plan must be developed and incorporated into the study protocol. This plan should outline the procedures for monitoring and reporting adverse events, ensuring that all potential risks are systematically addressed. The regulatory agency requires that these plans comply with international standards, including those established by the International Council for Harmonization (ICH), which Brazil became a member of in 2016 to improve its pharmacovigilance practices. Furthermore, ANVISA requires detailed instructions for blood or plasma sampling procedures, including collection time points after drug administration, to ensure reliable results.
- Data Safety Monitoring Boards (DSMBs): For larger clinical studies, the establishment of a Data Safety Monitoring Board may be necessary. These boards are responsible for overseeing participant safety throughout the study and making informed recommendations regarding the continuation or modification of the research based on safety data. The implementation of such boards is a critical step in ensuring compliance with ANVISA regulations and understanding how ANVISA impacts clinical trial design, which is essential for enhancing the overall safety monitoring framework.
Alongside these criteria, it is essential for researchers to recognize that research ethics committees cannot impose charges for evaluating medical study protocols. This highlights the dedication to ethical supervision in medical research. As highlighted by Josiany Carlos de Souza, "the risks are common to medical devices; therefore, it is crucial that there are preventative measures to avoid them, for example, training users to use the products, maintenance, improving quality, and reporting adverse events to manufacturers."
This proactive method not only complies with regulatory standards but also promotes a culture of safety and responsibility in research studies. With bioaccess®'s extensive management services for studies, including feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting, we are well-prepared to handle the complexities of research in Latin America.
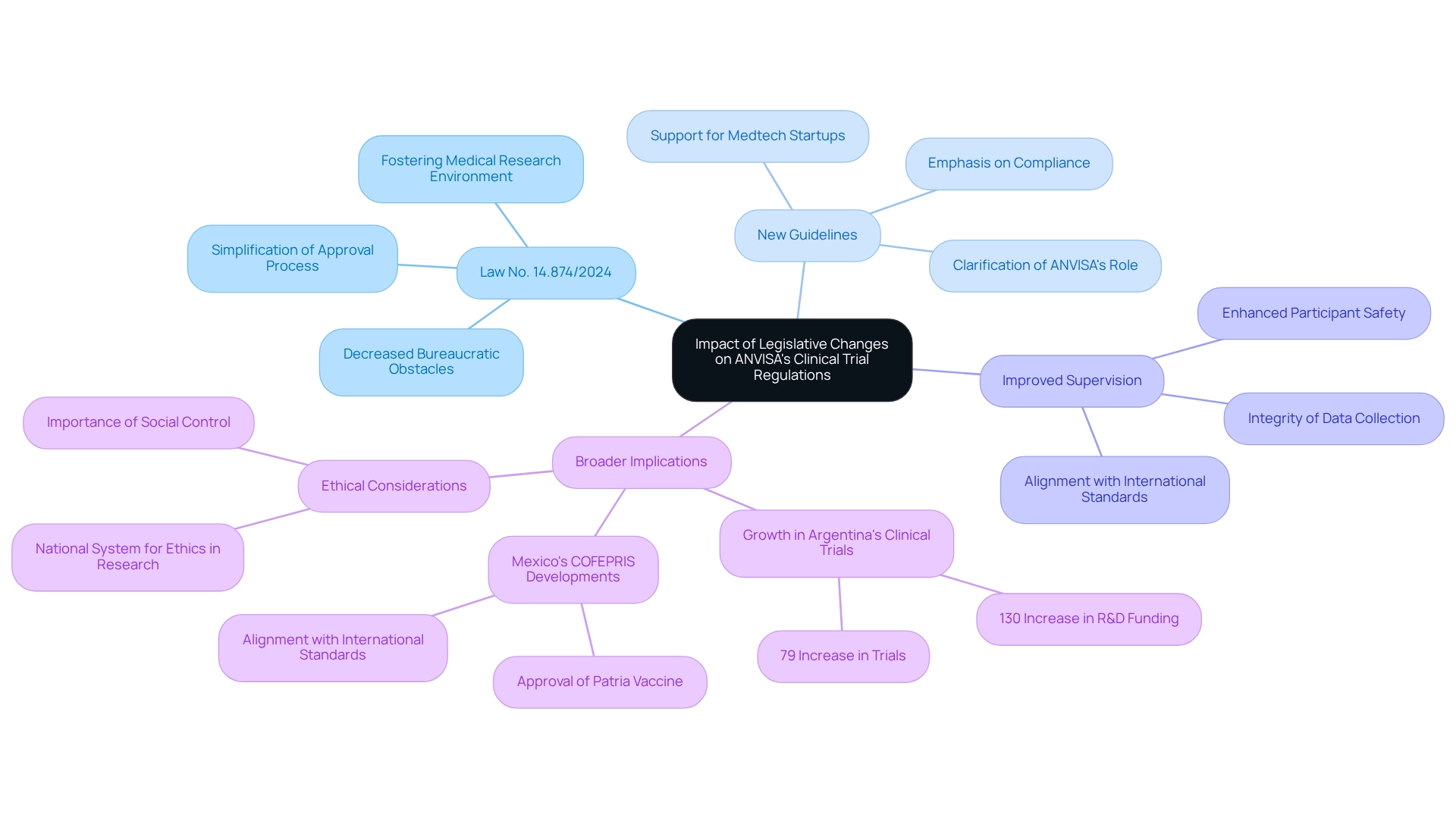
Impact of Recent Legislative Changes on ANVISA's Clinical Trial Regulations
Recent legislative changes in Brazil have profoundly influenced ANVISA's clinical trial regulations, with several key developments shaping the landscape:
- Law No. 14.874/2024: This crucial legislation is designed to simplify the approval process for research trials, effectively decreasing bureaucratic obstacles and improving predictability for researchers. By streamlining procedures, the law aims to foster a more favorable environment for medical research, ultimately accelerating how ANVISA impacts clinical trial design and advances innovative healthcare technologies. Bioaccess® stands at the forefront of this transformation, offering accelerated medical device clinical study services that leverage over 20 years of expertise in managing Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies. Our services guarantee swift compliance approval and extensive data management for studies, providing clients with the assurance required to advance their projects.
- New Guidelines: In response to these legislative changes, ANVISA has issued updated guidelines that emphasize the importance of compliance with the new standards. These guidelines not only reflect the recent changes but also clarify how ANVISA influences clinical trial design, ensuring that researchers understand the expectations and adhere to the latest regulatory requirements. Bioaccess® is committed to navigating these guidelines efficiently, offering customized solutions for Medtech startups to ensure compliance and successful testing results. Our clients have lauded our ability to simplify complex processes, as evidenced by [insert client testimonial].
- Improved Supervision: The recent modifications also incorporate measures for enhanced supervision of clinical studies. This focus on supervision is vital for preserving participant safety and ensuring the integrity of data collected during experiments. As Brazil aligns its regulatory framework with international standards, the emphasis on rigorous oversight illustrates how ANVISA impacts clinical trial design, fostering greater confidence among sponsors and researchers alike. Bioaccess®'s comprehensive management services for studies include compliance assessments and project coordination to facilitate this improved supervision, ensuring that our clients can manage these changes seamlessly.
The impact of Law No. 14.874/2024 illustrates how ANVISA influences clinical trial design. The research sector is increasingly recognizing this influence. For instance, Argentina has experienced an extraordinary 79% growth in research studies and a 130% increase in healthcare R&D funding from 2017 to 2021, driven by comparable government reforms.
This trend underscores Latin America's growing capacity as a hub for medical research, attracting international studies and funding in innovation. Furthermore, Mexico's COFEPRIS recently authorized the first Mexico-developed COVID-19 vaccine, highlighting the advancements in oversight frameworks across the region.
As Jucelia Maria Guedert noted, "It is essential that the new 'National System for Ethics in Research with Human Beings' considers social control, works in line with Anvisa and is adequately funded to fulfill its mandate." This statement underscores the significance of ethical considerations within the context of the new regulations. As Brazil continues to adapt its regulatory environment, understanding how ANVISA impacts clinical trial design holds significant implications, paving the way for more efficient and effective research practices, supported by Bioaccess®'s expertise in regulatory navigation and tailored solutions.
Conclusion
In conclusion, the evolving regulatory landscape for clinical trials in Brazil, propelled by ANVISA's unwavering commitment to safety, efficiency, and innovation, is crucial for the future of medical research. Recent legislative changes, including Law No. 14.874/2024, are designed to streamline approval processes, thereby reducing bureaucratic hurdles for researchers and enhancing the predictability of trial outcomes. This pivotal shift positions Brazil as a competitive hub for clinical research in Latin America, emphasizing the necessity for stakeholders to stay informed and engaged.
For researchers and sponsors aiming to conduct successful clinical trials, understanding ANVISA's requirements is essential. Compliance with comprehensive Clinical Trial Applications, Good Clinical Practices, and timely reporting of adverse events is paramount. Additionally, the implementation of bioequivalence studies and rigorous safety monitoring highlights the agency's dedication to public health standards while fostering an innovative environment.
As Brazil navigates the complexities of clinical trial regulations, collaboration with experienced partners, such as bioaccess®, becomes increasingly vital. This partnership can provide the necessary support to manage clinical trials efficiently, ensuring adherence to regulatory standards while addressing challenges like low recruitment rates. By prioritizing education and awareness initiatives, stakeholders can unlock the potential for increased participation in clinical research, ultimately benefiting public health and advancing medical science.
Frequently Asked Questions
What is the role of ANVISA in Brazil?
The Brazilian Health Regulatory Agency (ANVISA) is responsible for the regulation and oversight of health products, including drugs and medical devices, ensuring their safety and effectiveness. It plays a crucial role in the approval and oversight of research studies throughout Brazil.
How has ANVISA improved the process for evaluating human biological samples for research?
ANVISA will evaluate and distribute human biological samples intended for research within 48 hours of their arrival in Brazil, provided all legal requirements are met. This accelerated process is designed to enhance the efficiency of medical studies, especially for multicenter research.
What is the RNDS and its significance?
The RNDS (National Health Data Network) is expected to serve as a national reference for healthcare data collection. It aims to improve approval rates for research studies and ensure that data gathering aligns with compliance standards, although challenges remain in establishing data use criteria.
What challenges does Brazil face regarding research study recruitment?
A survey indicated a lack of awareness among patients and doctors about the advantages of research, contributing to low recruitment levels for studies in Brazil. Focused educational programs are necessary to enhance awareness and comprehension of research studies.
What are the key requirements for conducting clinical studies in Brazil?
Key requirements include: 1. Submission of a Clinical Trial Application (CTA) with detailed study protocol and investigator qualifications. 2. Obtaining ethical approval from local ethics committees and, in some cases, the National Commission for Ethics in Research (CONEP). 3. Adherence to Good Clinical Practices (GCP) to ensure data integrity and participant safety. 4. Prompt reporting of adverse events to maintain participant safety and regulatory compliance.
How does ANVISA influence clinical trial design?
ANVISA establishes benchmarks for compliance and ethical behavior in research, impacting clinical trial design significantly. Understanding ANVISA's role is vital for researchers and sponsors conducting studies in Brazil.
What support does bioaccess® provide to researchers in navigating ANVISA regulations?
Bioaccess® offers expertise in managing research services, including assistance in preparing and submitting CTAs, navigating ethical requirements, ensuring GCP compliance, and managing adverse event reporting, all aimed at facilitating successful study outcomes.
What is the current state of the approval process for medical studies in Brazil?
The approval process for medical studies in Brazil is evolving, with a notable percentage of research study applications receiving approval. ANVISA is committed to promoting research while upholding strict ethical standards, and ongoing improvements are being made to the approval process.




