Overview
Investing in Latin American Contract Research Organizations (CROs) presents a strategic advantage, primarily due to lower operational costs, a diverse patient population, and an evolving regulatory environment that enhances research efficiency. These factors, combined with the region's growing infrastructure and the increasing demand for innovative medical technologies, position Latin America as a pivotal hub for clinical studies. This landscape not only attracts investors but also underscores the potential for significant returns in the Medtech sector.
As the Medtech landscape evolves, the role of bioaccess becomes increasingly critical in addressing key challenges faced in clinical research. The collaboration between stakeholders in this sector fosters an environment conducive to innovation and efficiency. By leveraging the unique advantages offered by Latin America, investors can tap into a market that is ripe with opportunity and poised for growth.
In conclusion, the importance of collaboration in this dynamic environment cannot be overstated. Investors are encouraged to consider the next steps in engaging with Latin American CROs, capitalizing on the region's strengths to drive forward the future of clinical research.
Introduction
Latin America is rapidly emerging as a powerhouse in the realm of clinical research, attracting global attention for its unique advantages and growing capabilities. With a diverse patient population, lower operational costs, and an expanding infrastructure, the region is positioning itself as a strategic hub for innovative medical technologies. As the demand for efficient clinical trials rises, organizations like bioaccess® are stepping up to deliver high-quality research that meets international standards.
This article delves into the evolving landscape of Clinical Research Organizations (CROs) in Latin America, exploring the myriad opportunities, regulatory considerations, and future trends that make this region a focal point for investors and stakeholders alike in the quest for groundbreaking healthcare solutions.
The Emerging Landscape of Clinical Research Organizations in Latin America
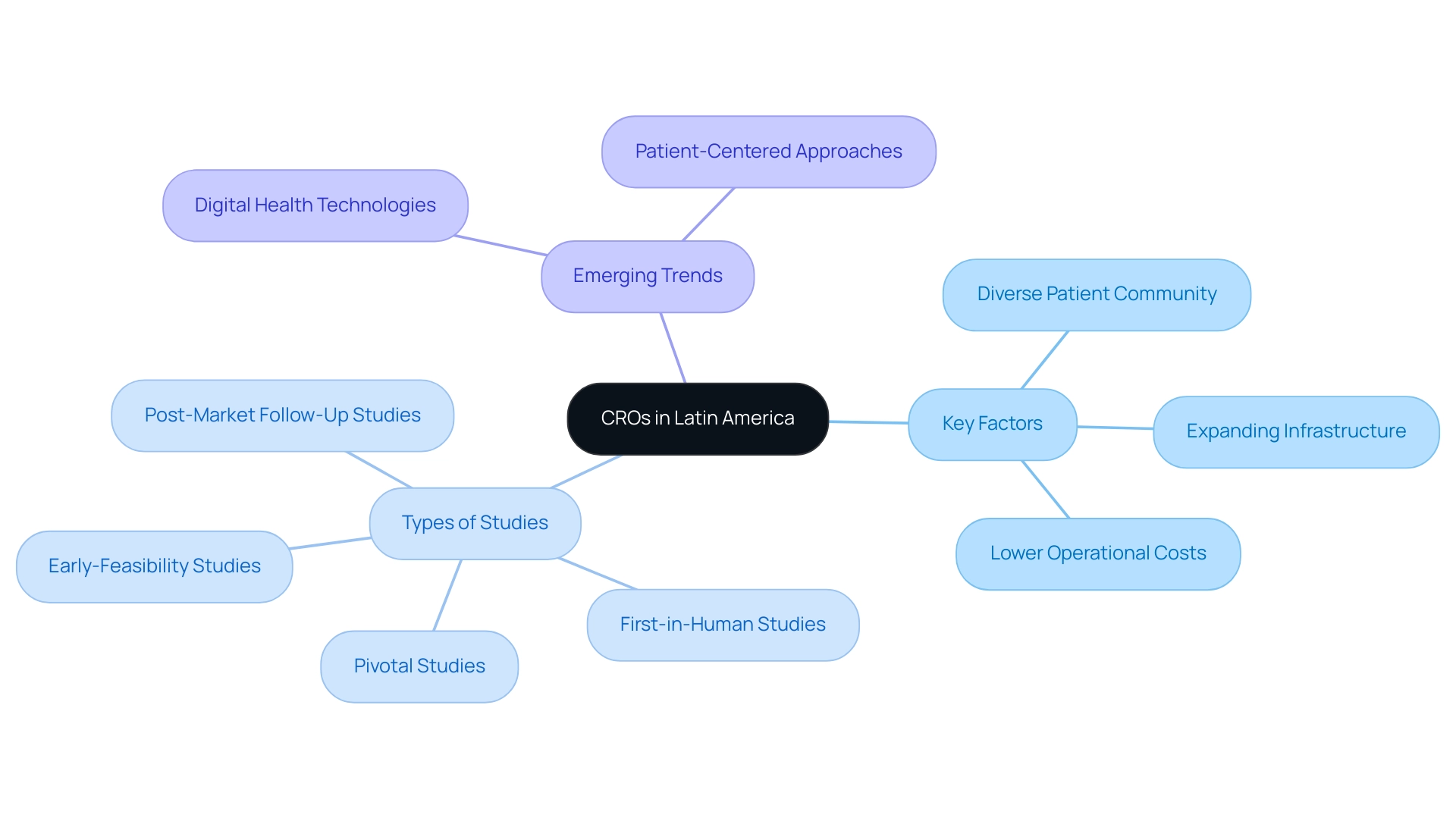
The region of South America has swiftly established itself as a dynamic hub for medical studies, driven by several critical factors. It boasts a diverse patient community, enhancing the representativeness of research studies, alongside significantly lower operational costs compared to other regions. Furthermore, the infrastructure for conducting medical studies is expanding, with an increasing number of facilities and skilled professionals dedicated to research.
CROs in South America, such as bioaccess®, are gaining recognition for their capacity to deliver high-quality research that meets international standards. This transformation is primarily fueled by the rising demand for innovative medical technologies and the pressing need for efficient research processes that expedite market entry. By 2025, the growth of medical research in Latin America is expected to continue, focusing on:
- Early-feasibility studies
- First-in-human studies
- Pivotal studies
- Post-market medical follow-up studies
These studies cater to the needs of the Medtech sector.
Investors are increasingly drawn to the potential of Latin American CROs, prompting the question: why invest in Latin American CROs? They are strategically positioned to support the burgeoning Medtech industry. The involvement of CROs is anticipated to drive the expansion of research studies in the region, contributing to the overall enhancement of healthcare solutions. Successful medical studies conducted by CROs in Colombia have demonstrated the effectiveness of leveraging local knowledge and resources, underscoring the region's capability to produce outcomes that align with international standards.
This aligns with bioaccess®'s mission to expedite the development of medical devices through its expertise and tailored strategy, backed by over 20 years of experience in the Medtech sector, reinforcing the rationale behind investing in Latin American CROs as a valuable opportunity.
As the landscape of clinical trials evolves, emerging trends such as the integration of digital health technologies and patient-centered approaches are becoming increasingly prevalent. These trends not only enhance trial efficiency but also improve patient engagement and data collection processes. With literacy rates reported at 95.7% in countries like Chile, the region is well-equipped to embrace these innovations, further solidifying its position as a leader in research.
Key Advantages of Investing in Latin American CROs
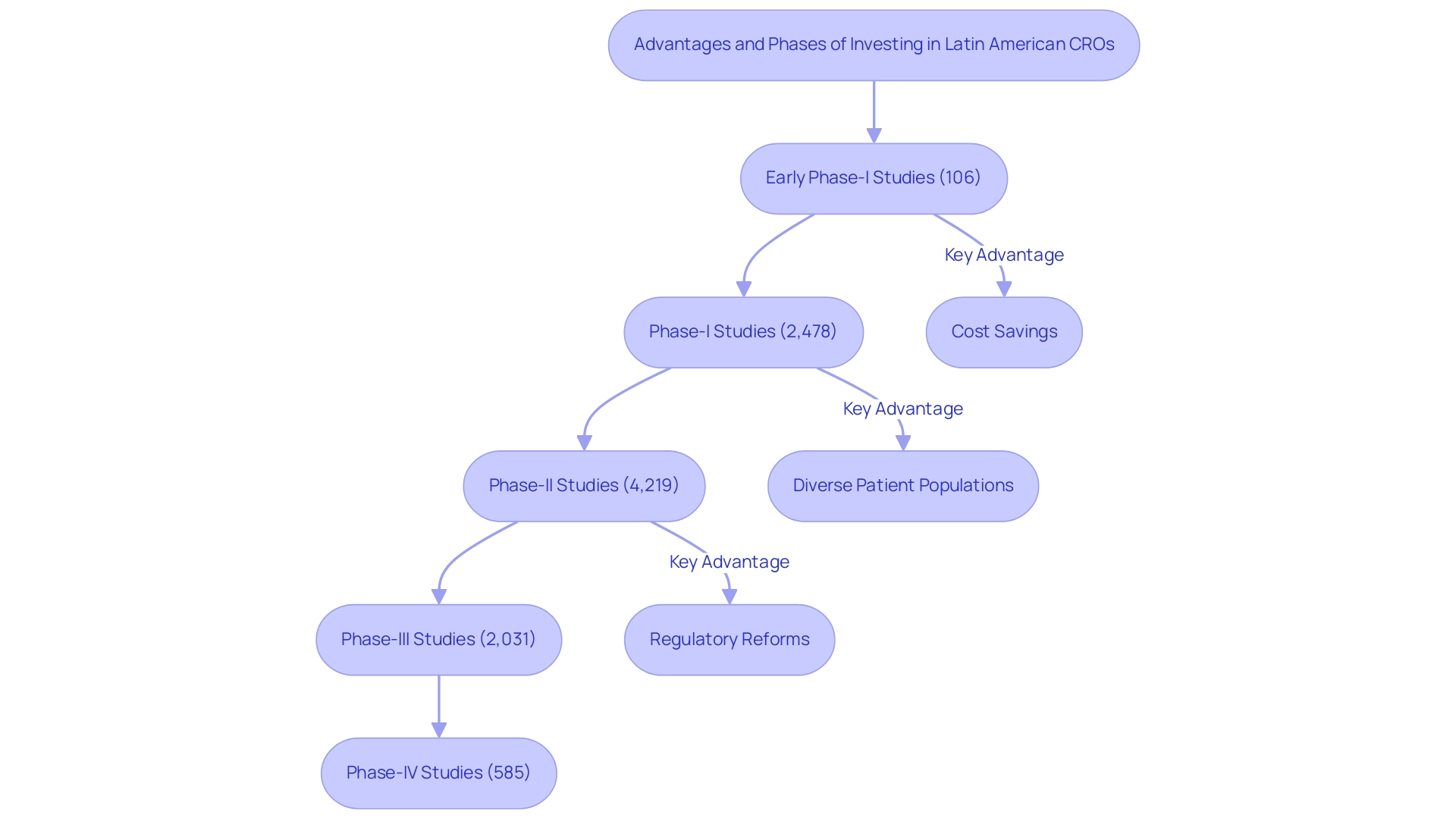
Investing in South American CROs offers numerous advantages, prompting an exploration of why to invest in Latin America CROs, particularly regarding cost savings compared to North America and Europe. The region boasts a substantial and diverse patient population, essential for research requiring varied demographic representation. In 2022, the area recorded a significant number of medical studies across various phases:
- 106 early phase-I interventional studies
- 2,478 phase-I interventional studies
- 4,219 phase-II interventional studies
- 2,031 phase-III interventional studies
- 585 phase-IV interventional studies
This underscores the region's robust scientific activity.
Recent advancements in regulatory reforms across the region have streamlined approval processes for medical studies, enhancing operational efficiency. For instance, Uruguay's implementation of fiscal regulations aimed at economic stability has fostered a more favorable environment for medical studies, leading to improved experiment efficiency and data quality. This is particularly relevant as the commitment to enhancing study infrastructure continues to evolve, indicating the potential for high-quality outcomes in medical trials.
The cost benefits of conducting trials in South America are significant. Companies can expect substantial savings on operational expenses, which can be reinvested into further development and innovation. This financial efficiency, coupled with the region's diverse patient demographics, positions Latin America CROs as an attractive option for Medtech firms seeking to conduct medical studies.
The diverse patient populations not only enrich the data collected but also align with the increasing demand for enhanced healthcare access and diversity in research enrollment. As Allison Kalloo noted, "The current landscape is shaped by a demand for improved healthcare access and a commitment to diversity in research enrollment." This positions Central America as a strategic choice for research studies in 2025. Furthermore, partnering with bioaccess® provides access to comprehensive management services for research, including Early-Feasibility Studies, First-In-Human Studies, and Post-Market Follow-Up Studies.
The collaboration between bioaccess® and Caribbean Health Group aims to establish Barranquilla as a leading hub for clinical research in South America, supported by Colombia's Minister of Health. This initiative not only bolsters the region's reputation but also contributes to local economies through job creation and healthcare improvements. However, it is crucial to recognize the challenges that persist in the region, particularly in countries like Nicaragua and Guatemala, where issues related to pension system coverage and poverty alleviation may impact judicial efficiency and data quality.
Despite these challenges, the commitment to enhancing scientific infrastructure in South America has led to advancements in technology and methodologies. The case study titled "Enhancing Research Infrastructure in Latin America" illustrates how these improvements collectively enhance study efficiency and data quality, highlighting the benefits of collaborating with regional CROs like bioaccess® for medical initiatives. This raises the essential question: why invest in Latin America CROs?
Navigating the Regulatory Landscape for Clinical Research in Latin America
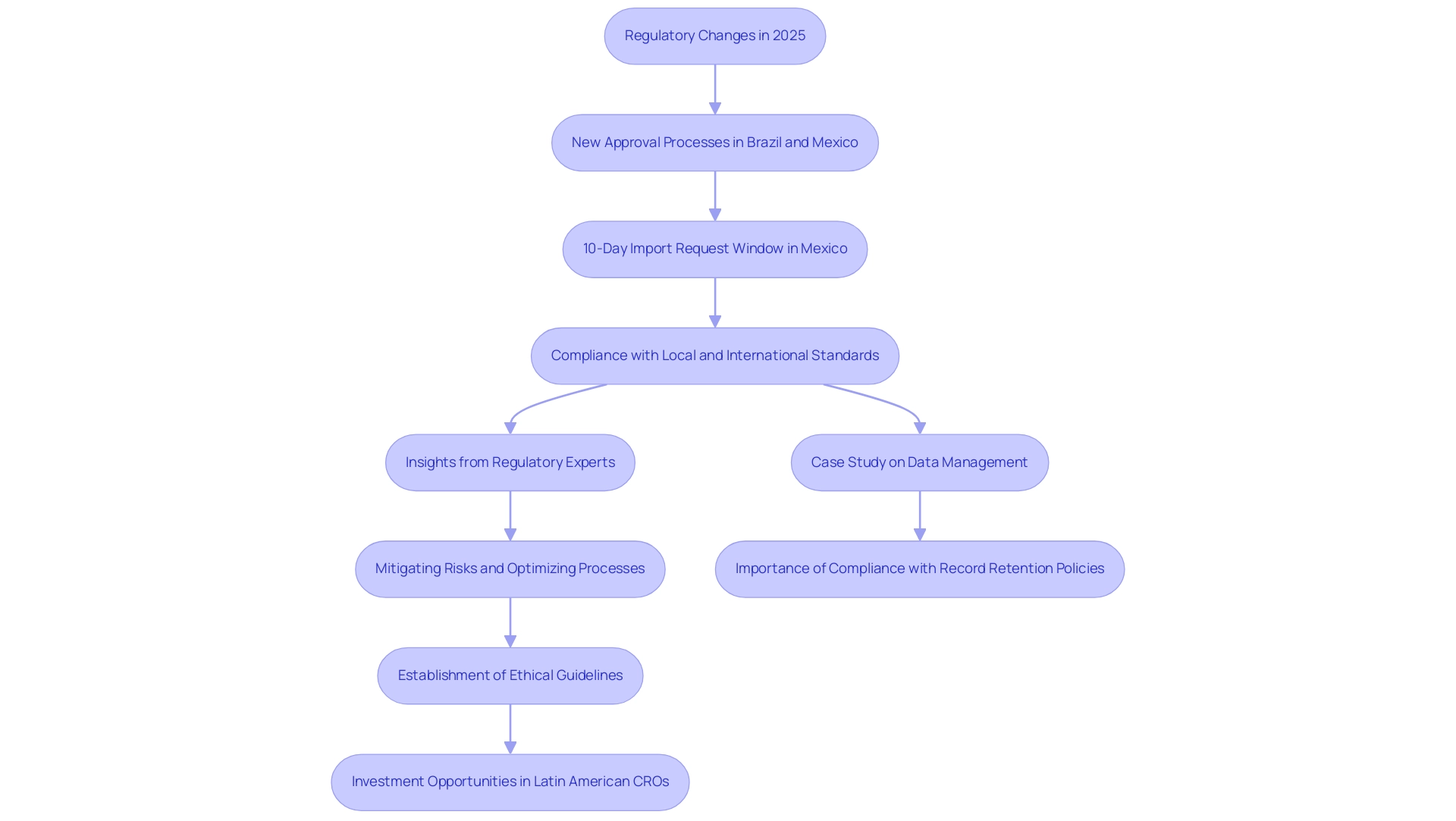
The regulatory environment for medical research in Latin America is intricate yet rapidly evolving. In 2025, Brazil and Mexico have enacted significant changes designed to streamline the approval process for research studies, thereby potentially reducing the time required to launch new medical devices. For example, in Mexico, the Federal Commission for the Protection against Sanitary Risk (COFEPRIS) has instituted a 10-day window for approving import requests; if these requests are not addressed within this timeframe, they are automatically considered approved.
This reform exemplifies a commitment to enhancing efficiency within the regulatory framework.
Understanding these regulations is paramount for investors, as adherence to local laws and international standards is essential for the successful execution of research studies. The complexities of navigating these regulations can be daunting; however, insights from industry experts such as Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, and Katherine Ruiz, a specialist in Regulatory Affairs for Medical Devices and In Vitro Diagnostics in Colombia, can significantly assist companies in mitigating risks and optimizing their development processes. A recent case study on data management and record retention in medical studies underscored the critical importance of maintaining compliance with regulatory standards.
It demonstrated that effective data management not only safeguards the integrity of medical study data but also ensures compliance with record retention policies, which are vital for regulatory adherence, including the specific durations for which data must be retained and the responsibilities of sponsors and investigators.
Moreover, the establishment of operational guidelines by Hospital Bioethics Committees, which are required to convene at least six times annually, underscores the growing emphasis on ethical oversight in medical investigations. This evolving regulatory landscape, coupled with the requirement for approval from parents, guardians, or legal representatives for terminally ill patients unable to provide consent, reflects a broader commitment to ethical standards in clinical trials.
As these reforms continue to unfold, they present a unique opportunity for companies to explore the rationale behind investing in Latin American Contract Research Organizations (CROs) to harness the advantages of conducting research in South America. By implementing effective compliance strategies, such as staying updated on regulatory changes and collaborating with local experts, investors can adeptly navigate this dynamic environment, emphasizing the necessity of investing in Latin American CROs. This ultimately accelerates the development and commercialization of innovative medical devices, showcasing the benefits of partnering with bioaccess®, a leading Contract Research Organization that offers comprehensive clinical study management services, including Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies.
Challenges and Considerations for Investing in Latin American CROs
Why invest in Latin America CROs? Investing in Latin American CROs presents a wealth of opportunities, yet it is accompanied by distinct challenges that require careful navigation. Recruitment difficulties remain a significant concern, as cultural differences in patient-physician relationships can complicate the informed consent process. Patients frequently accept medical advice without questioning it, which can lead to misunderstandings in research protocols.
Furthermore, the differing levels of infrastructure throughout the region can affect the efficiency of medical research. For example, all evaluated research sites in Peru have successfully met regulatory standards since 2000, emphasizing a strong compliance record, while other nations may not possess the same degree of operational preparedness.
In 2025, the landscape is further complicated by the political and economic instability that can influence the operational environment for CROs. Investors must conduct thorough due diligence to understand these dynamics and develop strategic plans that account for potential disruptions. Recent legislative changes, such as Brazil's Law 14.874/24, which aims to simplify the assessment process for research studies, signal a positive shift towards reducing bureaucratic obstacles and enhancing predictability in the region.
Moreover, effective communication and collaboration with CROs like bioaccess are crucial for overcoming recruitment challenges. bioaccess provides extensive trial management services, including:
- feasibility studies
- site selection
- compliance reviews
- trial setup
- import permits
- project management
- reporting of study status
- inventory
- adverse events
Establishing clear communication channels and maintaining regular updates can align stakeholders on project goals and timelines, ultimately leading to higher success rates in study completion.
Research indicates that strong partnerships characterized by open dialogue can significantly benefit healthcare advancements and bolster local economies. A case study titled "Fostering Effective Communication and Collaboration with Your CRO" illustrates that effective communication is vital for achieving successful results.
As prospective investors examine the South American market, understanding these challenges and utilizing expert knowledge will be crucial for addressing why invest in Latin America CROs to take advantage of the region's promising prospects in medical studies. Significantly, Julio G. Martinez-Clark, CEO, highlights that "the government of Colombia appears to be the sole nation in South America with an active initiative to draw more trials as part of its strategy to evolve into a knowledge economy by 2031," emphasizing the regional endeavors to improve medical study capabilities. With bioaccess at the forefront of Medtech medical studies, their emphasis on innovation and regulatory excellence establishes them as a vital ally in navigating this changing landscape.

Future Trends and Opportunities in Latin American Clinical Research
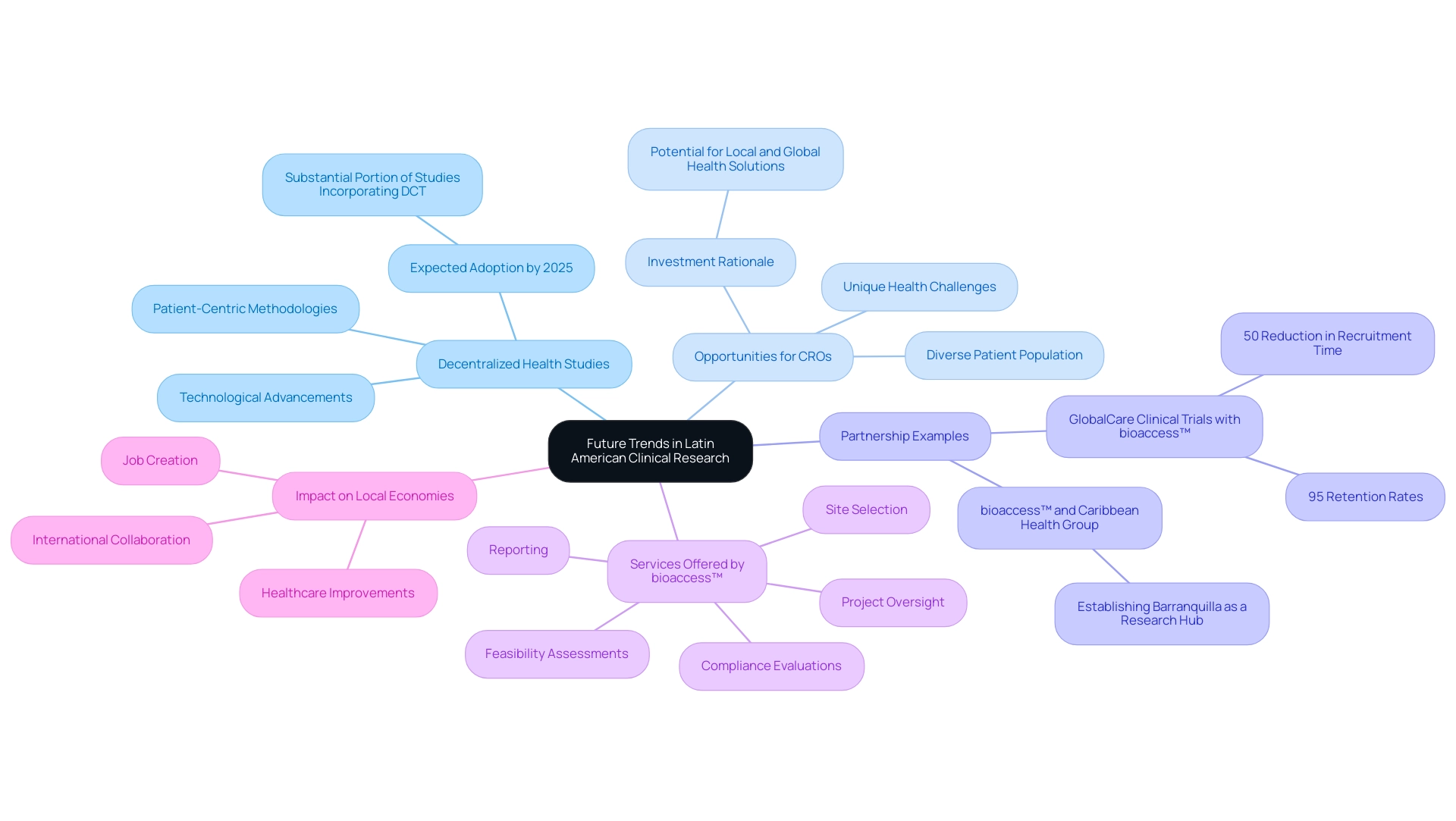
The future of medical research in Latin America is on the brink of significant transformation, driven by technological advancements and an increasing focus on patient-centric methodologies. Decentralized health studies (DCTs) are at the forefront of this evolution, leveraging digital tools to enhance patient involvement and streamline data gathering processes. This innovative approach not only enhances the participant experience but also boosts the efficiency and precision of studies.
As Medtech firms progressively concentrate on emerging markets, a crucial consideration arises: why invest in Latin America CROs? The region is poised to become a vital hub for research studies. Its diverse patient population and unique health challenges present a wealth of opportunities for research that can address both local and global health needs. By 2025, the adoption of decentralized research studies is expected to rise significantly, with statistics indicating that a substantial portion of studies will incorporate DCT components, reflecting a broader trend towards adaptability and accessibility in study design.
A notable example of this shift is the partnership between bioaccess™ and Caribbean Health Group, aimed at establishing Barranquilla as a key location for research studies in Latin America. Supported by Colombia's Minister of Health, this initiative seeks to enhance the region's attractiveness for research endeavors. Furthermore, collaborations such as that of GlobalCare Clinical Trials with bioaccess™ have yielded tangible benefits, achieving over a 50% reduction in recruitment time and impressive retention rates of 95%.
bioaccess™ offers comprehensive management services for research studies, encompassing:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Setup
- Import permits
- Project oversight
- Reporting
These services are essential for ensuring that medical studies are conducted effectively and in alignment with local regulations. The impact of these medical studies extends beyond research; they contribute to local economies through job creation and healthcare improvements, fostering international collaboration.
Investors who grasp the rationale behind investing in Latin America CROs and adapt their strategies accordingly will find themselves well-positioned to capitalize on the myriad opportunities the region presents. Notably, approximately 65% of Horizon Databook's revenue is generated from competitive intelligence and market intelligence teams, underscoring the importance of data in trials. The shift towards enhanced data ownership and transparency among sponsors, as illustrated by the case study titled "Increased Data Ownership and Transparency Among Sponsors," highlights the trend of sponsors seeking greater control over their data, aligning with the discourse on DCTs and serving as a real-world example of the evolving landscape in clinical research.
Moreover, as Roshan Deshmukh aptly stated, "The future of medical exploration in South America is bright, with unique opportunities for innovation that can address both local and global health needs." By considering the reasons for investing in Latin America CROs, stakeholders can not only enhance their operational capabilities but also contribute to the advancement of medical innovations that have the potential to improve patient outcomes worldwide.
Conclusion
Latin America is emerging as a leading destination for clinical research, driven by a combination of diverse patient populations, lower operational costs, and evolving regulatory frameworks. The region's Clinical Research Organizations (CROs), such as bioaccess®, are gaining traction for their ability to conduct high-quality trials that meet international standards, thereby attracting significant interest from investors and stakeholders in the Medtech sector.
The advantages of investing in Latin American CROs are compelling. With a robust number of clinical trials being conducted across all phases, coupled with streamlined regulatory processes, the region presents significant cost savings and diverse demographic representation. These factors not only enhance the quality of data collected but also align with the growing demand for improved healthcare access and diversity in research enrollment.
While there are challenges to navigate, such as recruitment difficulties and varying infrastructure across countries, the region's commitment to enhancing research capabilities continues to evolve. The integration of digital health technologies and patient-centric approaches signifies a promising future for clinical trials, making Latin America an attractive hub for innovative medical research.
As the landscape of clinical research in Latin America transforms, investors and stakeholders have a unique opportunity to leverage the region’s strengths. By partnering with experienced CROs like bioaccess®, they can capitalize on the potential for groundbreaking healthcare solutions that benefit not only local populations but also contribute to global health advancements. The future of clinical research in Latin America is bright, marked by innovation and a commitment to high-quality outcomes that can significantly improve patient care worldwide.
Frequently Asked Questions
Why has South America become a hub for medical studies?
South America has become a hub for medical studies due to its diverse patient community, lower operational costs, and expanding infrastructure for research, including more facilities and skilled professionals.
What types of studies are expected to grow in Latin America by 2025?
By 2025, the growth of medical research in Latin America is expected to focus on early-feasibility studies, first-in-human studies, pivotal studies, and post-market medical follow-up studies.
What advantages do Latin American CROs offer to investors?
Latin American CROs offer cost savings compared to North America and Europe, a diverse patient population for varied demographic representation, and a favorable regulatory environment that enhances operational efficiency.
What recent advancements have improved the efficiency of medical studies in South America?
Recent advancements include regulatory reforms that streamline approval processes, such as Uruguay's fiscal regulations aimed at economic stability, which foster a more favorable environment for medical studies.
How many medical studies were recorded in South America in 2022?
In 2022, South America recorded 106 early phase-I interventional studies, 2,478 phase-I interventional studies, 4,219 phase-II interventional studies, 2,031 phase-III interventional studies, and 585 phase-IV interventional studies.
What is the significance of diverse patient populations in medical research?
Diverse patient populations enrich the data collected during studies and align with the increasing demand for enhanced healthcare access and diversity in research enrollment.
What role does bioaccess® play in the medical research landscape in South America?
Bioaccess® provides access to comprehensive management services for research studies and aims to expedite the development of medical devices, leveraging over 20 years of experience in the Medtech sector.
What challenges does South America face in the medical research sector?
Challenges include issues related to pension system coverage and poverty alleviation in countries like Nicaragua and Guatemala, which may impact judicial efficiency and data quality.
How are collaborations like that between bioaccess® and Caribbean Health Group beneficial?
Such collaborations aim to establish leading hubs for clinical research, bolster the region's reputation, and contribute to local economies through job creation and healthcare improvements.
What emerging trends are influencing clinical trials in South America?
Emerging trends include the integration of digital health technologies and patient-centered approaches, which enhance trial efficiency and improve patient engagement and data collection processes.




