Overview
Latin America stands out as a compelling region for Medtech trials, primarily due to its cost-effectiveness, diverse patient demographics, and an increasingly supportive regulatory environment. This makes it a strategic choice for clinical research. Notably, conducting trials in this region can be up to 60% cheaper than in more developed markets. Recent regulatory improvements, exemplified by Brazil's Law 14.874/24, aim to streamline the approval process, thereby enhancing the overall feasibility and efficiency of Medtech studies.
Introduction
In recent years, Latin America has emerged as a compelling destination for Medtech trials, presenting a unique blend of operational advantages that can significantly enhance clinical research endeavors. The region boasts lower operational costs, a diverse patient population, and a rapidly evolving regulatory landscape, making it an attractive alternative to traditional markets like North America and Europe. As healthcare infrastructure continues to improve and innovative technologies gain traction, Medtech companies are increasingly drawn to Latin America. Here, they can achieve substantial cost savings while tapping into a rich demographic diversity that enriches trial outcomes.
This article delves into the strategic benefits of conducting clinical trials in Latin America, exploring the market landscape, regulatory frameworks, operational efficiencies, and the importance of strategic partnerships, all while highlighting the immense potential that lies ahead for the Medtech sector in this vibrant region.
Strategic Advantages of Conducting Medtech Trials in Latin America
The region offers a vibrant setting for Medtech evaluations, distinguished by reduced operational expenses, diverse patient demographics, and a progressively supportive regulatory framework. With significant enhancements underway in the healthcare infrastructure, it emerges as an attractive choice for clinical research. Businesses can realize substantial cost reductions, with projections indicating that conducting tests in the southern regions can be up to 60% more affordable than in the northern regions or Europe.
This cost reduction is particularly critical, given that patient recruitment expenses can range from $15,000 to $50,000 per participant in more developed markets. Moreover, the diverse demographics of Latin America present a rich pool of participants, enhancing the generalizability of study results. This diversity not only bolsters the scientific validity of studies but also aligns with the increasing demand for improved healthcare access and a commitment to diversity in research enrollment. As Allison Kalloo, founding partner and communications lead of Clinical Ambassador and iParticipate, aptly states, "The current landscape is shaped by a demand for improved healthcare access and a commitment to diversity in research enrollment."
The recent approval of Law 14.874/24 in Brazil exemplifies the region's dedication to promoting medical research. This legislation aims to streamline the evaluation process for medical studies, reducing administrative hurdles and enhancing predictability, which is expected to significantly boost research activities in Brazil.
Furthermore, bioaccess® provides comprehensive study management services, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF). The growth of contract research organizations (CROs) in the region and the high success rates of clinical studies further amplify the strategic advantages of conducting research in the southern continent. By leveraging these factors, Medtech companies can expedite their product development timelines while adhering to high-quality standards.
The combination of cost efficiency, diverse patient populations, and a supportive regulatory environment underscores the compelling reasons to choose Latin America for Medtech trials.

Understanding the Latin American Market Landscape for Medtech
The South American Medtech market is experiencing significant growth, fueled by a rising demand for innovative medical technologies. This surge is largely attributed to the increasing prevalence of chronic diseases and an aging population across the region. In 2025, countries such as Brazil, Mexico, and Argentina are at the forefront of healthcare investments, with government initiatives focused on enhancing healthcare infrastructure to meet these demands.
For instance, Brazil's healthcare sector has seen substantial capital influx, reflecting a commitment to improving patient care and access to advanced medical devices. Significantly, Healthtech startups in the southern region raised considerable capital from 2015 to 2021, indicating a strong investment trend in the sector.
The urbanized population in these countries also presents a strategic advantage for clinical trials, leading us to consider why choose Latin America for medtech trials, as it facilitates easier patient recruitment. However, Medtech companies must navigate a complex landscape of varying healthcare regulations and market access challenges unique to each country. Understanding these regulatory nuances is crucial for firms looking to establish a successful presence in this diverse market.
As George Frazer Moreira, Founder of FRAGIA CORP., observed, "With challenges ranging from logistics to import regulations, establishing an efficient supply chain in the southern continent can appear daunting." But with the right knowledge and strategies, these challenges can be overcome.
Furthermore, the competitive nature of the Medtech sector in South America is highlighted by the presence of numerous participants, with substantial market shares held by prominent firms. For example, the ventilators market showcases over 60 competitors, with Draeger leading at 17%, followed closely by Neumovent and Medtronic. Additionally, Philips leads the American market for C-arms with a 38% share of the installed base, followed by GE Healthcare at 29% and Siemens at 20%.
This competitive environment not only fosters innovation but also encourages companies to develop effective strategies to overcome logistical and regulatory hurdles.
As the region continues to evolve, the demand for medical technologies is expected to rise, driven by the urgent need to address chronic diseases. This trend offers a distinctive chance for Medtech companies to utilize their knowledge and aid in enhancing healthcare outcomes, highlighting why choose Latin America for medtech trials. Moreover, bioaccess® provides extensive management services for studies, including:
- Feasibility assessments
- Site selection
- Compliance evaluations
- Setup
- Import permits
- Project oversight
- Reporting
This ensures a streamlined process for advancing medical device evaluations.
The partnership between bioaccess® and Caribbean Health Group seeks to establish Barranquilla as a prominent location for medical studies in the region, backed by Colombia's Minister of Health, improving the area's appeal for first-in-human research and other health-related initiatives.

Navigating Regulatory Frameworks for Clinical Trials in Latin America
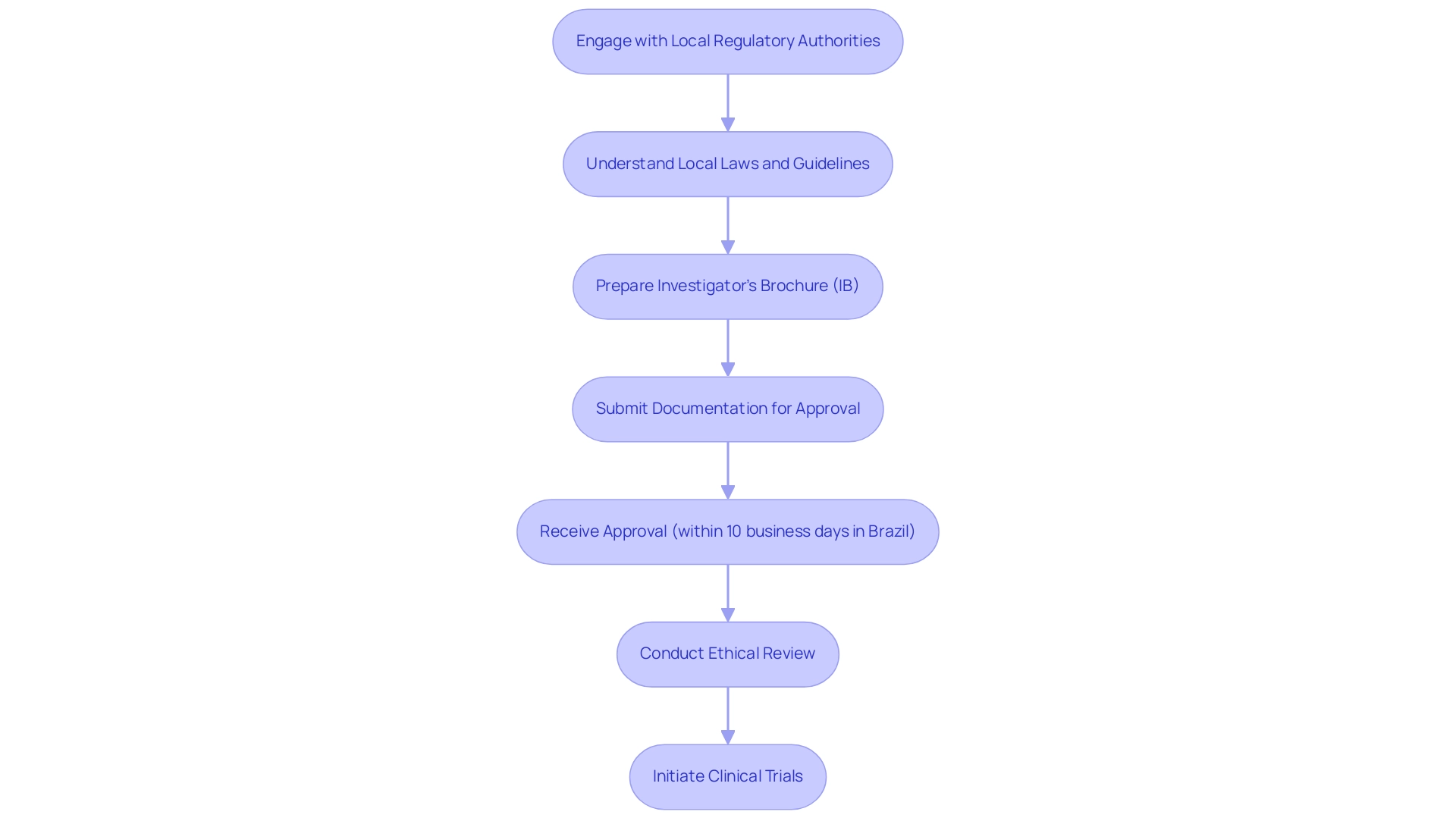
As the regulatory landscape for research studies in Latin America undergoes notable change, it prompts a critical inquiry: why choose Latin America for medtech trials? Numerous nations are enacting policies aimed at attracting foreign investment and simplifying procedures. In Brazil, recent legislative changes have focused on streamlining the approval process for clinical studies, effectively reducing bureaucratic obstacles that previously impeded progress. For instance, the Brazilian regulatory authority now commits to reviewing submitted documents within 10 business days, significantly accelerating the timeline for initiating the process.
Despite these advancements, the regulatory landscape remains diverse across the region, necessitating a comprehensive understanding of local laws and guidelines. Companies aiming to conduct tests must engage with local regulatory authorities early in their planning phases to ensure compliance and facilitate quicker approvals. This proactive strategy not only mitigates potential risks but also enhances the likelihood of successful outcomes.
Moreover, the evolving frameworks are designed to support innovative medical technologies. Recent changes in Brazil allow waivers on signed informed consent forms under specific conditions, such as minimal risk research, which can expedite the commencement of studies. However, it is crucial to note that nonviable neonates cannot be involved in research unless vital functions are not artificially maintained, and informed consent is obtained, underscoring the importance of ethical considerations in medical research.
As Alexandre Dalmasso, a partner at Licks Attorneys Compliance, observes, "The evolving regulatory landscape in Brazil is a significant step towards fostering innovation in the Medtech sector, but it requires diligent adherence to ethical standards and local regulations."
In Colombia, the National Food and Drug Surveillance Institute (INVIMA) serves as a Level 4 health authority, overseeing medical device regulation and ensuring compliance with international standards. Partnerships, such as the one between bioaccess™ and Caribbean Health Group, aim to establish Barranquilla as a premier location for research studies in Latin America, supported by Colombia's Minister of Health. This collaboration exemplifies the commitment to enhancing research capabilities in the region.
Case studies illustrate how Medtech companies have effectively navigated these regulatory frameworks. For example, sponsors are now required to provide investigators with an Investigator’s Brochure (IB) that includes detailed information about the investigational new drug (IND), ensuring that all parties are well-informed about the product's formulation, pharmacological effects, and safety data. This essential document promotes safety and compliance during research studies, highlighting the significance of comprehensive preparation and adherence to regulatory standards.
As the regulatory structures for research studies in the region continue to evolve into 2025, understanding the rationale behind choosing Latin America for medtech trials is crucial for Medtech firms seeking to leverage the area's distinctive advantages for medical investigations.
Operational Benefits: Cost-Effectiveness and Patient Access in Latin America
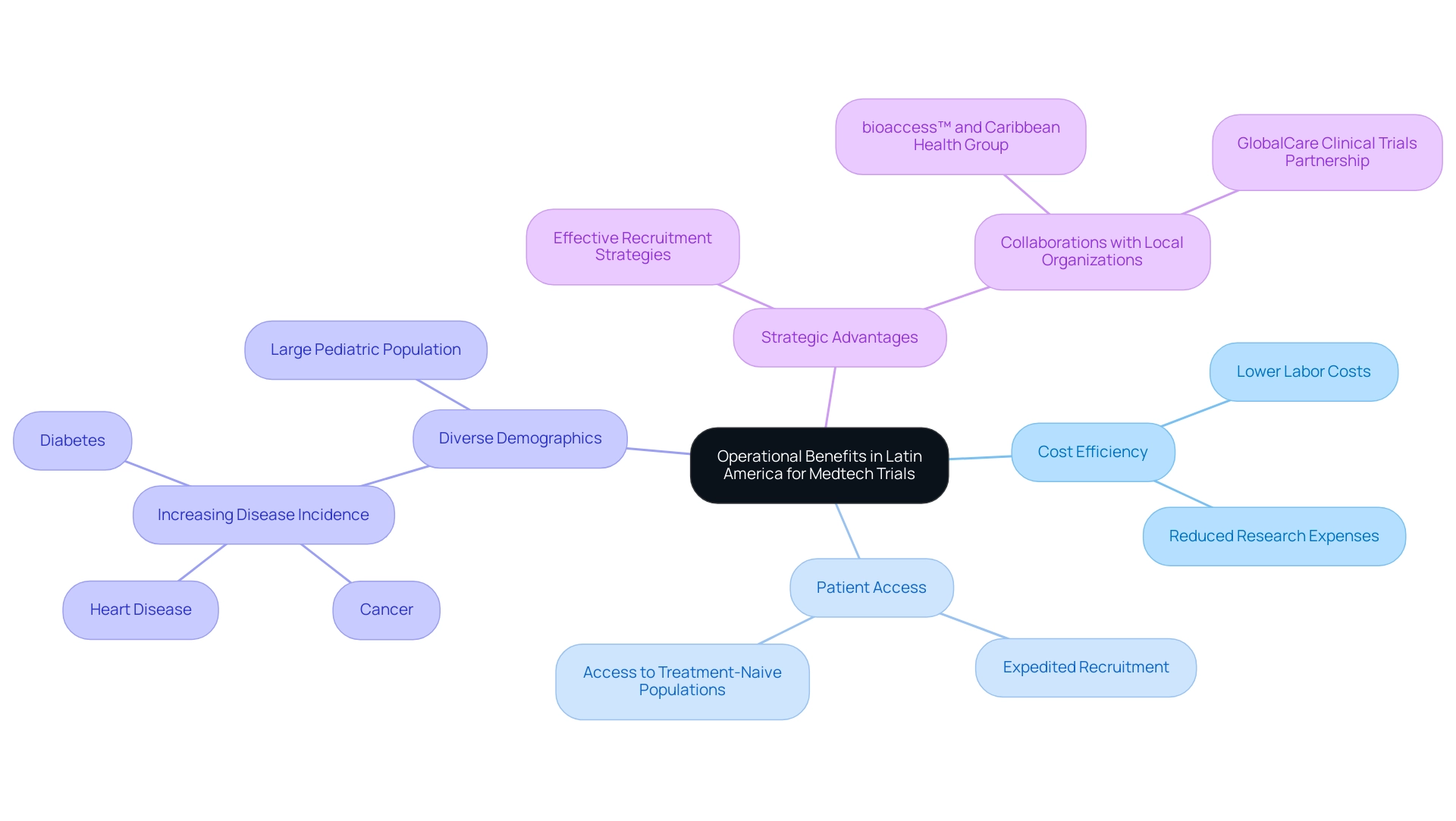
There are significant operational benefits, particularly concerning cost efficiency, which elucidates the rationale for choosing Latin America for medtech trials. Labor costs in the region are markedly lower than in many other global areas, allowing companies to substantially reduce overall research-related expenses. This financial efficiency is further enhanced by access to a large, treatment-naive patient population, prompting the question of why Latin America is an optimal choice for medtech trials, as it facilitates expedited patient recruitment and enrollment processes.
As Gotuzzo from Universidad Peruana Cayetano Heredia articulates, "These challenges demand a strategic approach to recruitment," underscoring the necessity of effective strategies in this context.
The diverse demographics of the region not only accelerate trial timelines but also strengthen the data collected. For example, the increasing incidence of diseases such as cancer, heart disease, and diabetes in countries like Chile and Peru creates a fertile environment for clinical studies, particularly for vaccine testing in treatment-naive populations. The case study titled "Disease Trends in South America" illustrates this point, noting the rising prevalence of these diseases and the potential for a burgeoning market, especially considering the substantial pediatric population in the area.
Moreover, when contemplating the strategic advantages, one might inquire why Latin America is a compelling choice for medtech trials, as it transcends mere financial savings. Companies can optimize their research budgets while enhancing their return on investment through effective patient access and recruitment strategies. The collaboration between bioaccess™ and Caribbean Health Group (CHG) to position Barranquilla as a premier site for medical research, endorsed by Colombia's Minister of Health, exemplifies this potential.
Additionally, GlobalCare Clinical Trials' partnership with bioaccess™ has achieved over a 50% reduction in recruitment time and 95% retention rates, showcasing the efficacy of localized strategies.
bioaccess® distinguishes itself as a leading Contract Research Organization supporting medical device studies in the region, offering extensive services such as feasibility assessments, site selection, compliance evaluations, study preparation, import permits, project management, and reporting. While regions like Southeast Europe provide low site expenses and skilled professionals, organizations seeking to expedite their clinical studies and accelerate the introduction of innovative medical devices to market should consider why Latin America is a strategic choice for medtech trials, as its unique landscape presents distinct opportunities. As the Medtech sector continues to evolve, leveraging these operational advantages will be crucial for fostering job creation, economic growth, and healthcare advancements in the region.
Challenges and Risks of Medtech Trials in Latin America
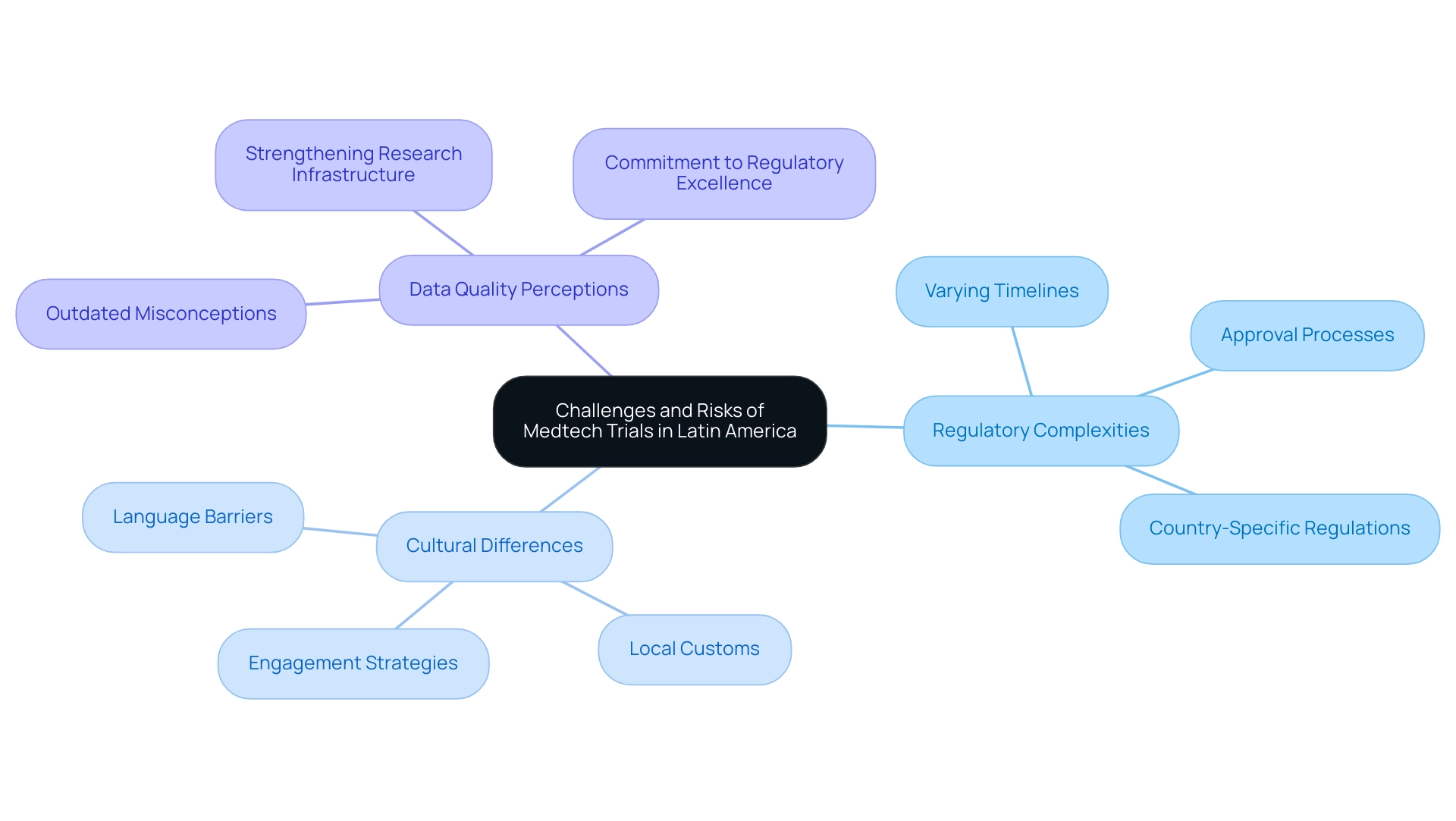
When evaluating the reasons to choose Latin America for medtech trials, it is crucial to acknowledge the myriad advantages it presents alongside the unique challenges companies must navigate. Regulatory complexities pose a significant concern, as each country maintains its own set of rules and approval processes. Despite recent advancements, the regulatory environment can still be daunting, characterized by varying timelines and requirements that may lead to delays in study initiation.
Nevertheless, countries such as Brazil, Argentina, and Mexico have made substantial progress in aligning their regulations with international standards, significantly reducing approval timelines to as little as six months in certain instances. This progress is underscored by the expertise of bioaccess®, which boasts over 20 years of experience in the Medtech sector, specializing in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF). This ensures a comprehensive and tailored approach to research management in the region.
Cultural differences further complicate the landscape. Understanding local customs and practices is essential for effective engagement with participants and stakeholders. Language barriers can hinder communication with regulatory entities and research sites, potentially leading to misunderstandings that may affect study outcomes.
Moreover, perceptions of data quality in the region remain a challenge, as outdated misconceptions about the reliability of research in South America persist. It is important to highlight that the notion of low-quality data is not only outdated but also inaccurate, as the region's research infrastructure continues to strengthen, reflecting a commitment to regulatory excellence.
To contextualize these cultural challenges, it is noteworthy that Peru has reportedly improved its literacy rate to 7.1% illiteracy, which can significantly impact communication and engagement strategies. To address these challenges, Medtech companies should prioritize establishing robust local partnerships. Collaborating with experienced local organizations can provide invaluable insights into navigating the regulatory environment and cultural nuances.
As Dushyanth Surakanti, Founder and CEO of Sparta Biomedical, emphasizes, the commitment to regulatory excellence and innovation in Medtech research is paramount. Furthermore, investing in comprehensive training for teams can enhance their ability to effectively manage these complexities. Companies can explore the reasons to choose Latin America for medtech trials by cultivating strong connections and understanding the regional context, thereby reducing risks and capitalizing on the distinct opportunities that the region offers.
Building Strategic Partnerships for Successful Trials in Latin America
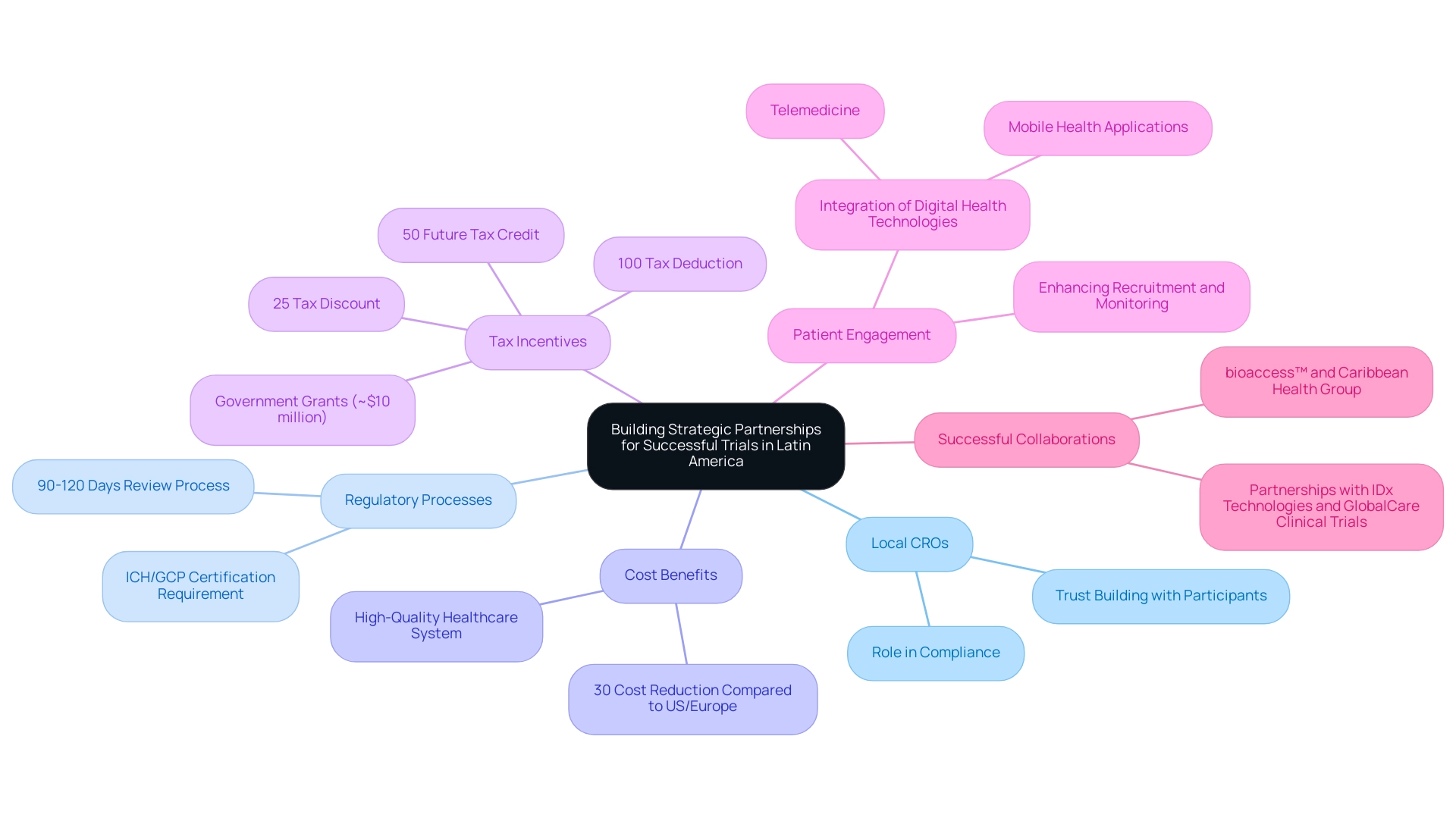
Forming strategic alliances is crucial for Medtech companies, prompting a closer examination of why Latin America is an ideal choice for Medtech trials. Collaborating with local Contract Research Organizations (CROs), healthcare institutions, and regulatory experts provides essential insights into the regional landscape. These alliances not only simplify the navigation of intricate regulatory processes but also significantly enhance patient recruitment initiatives—an essential factor, given that 85 percent of patients in the region are unaware of their participation options in studies.
Colombia emerges as a prime destination for first-in-human clinical studies, boasting competitive advantages. The country offers cost reductions exceeding 30% compared to trials in the United States or Western Europe, with the overall IRB/EC and MoH (INVIMA) review process typically requiring only 90-120 days. Moreover, Colombia's healthcare system is highly esteemed, ranked #22 by the World Health Organization among 191 countries, and recognized for housing some of the best hospitals in Latin America, according to America Economia magazine.
In Colombia, hospitals are authorized to conduct research with pharmaceutical drugs only after undergoing a rigorous ICH/GCP certification process, ensuring adherence to high-quality standards.
Additionally, the R&D tax incentives in Colombia provide substantial financial advantages for conducting clinical studies. Investments in science, technology, and innovation projects can benefit from:
- A 100% tax deduction
- A 25% tax discount
- A 50% future tax credit
- Approximately $10 million in government grants
Leveraging local knowledge enables firms to gain a deeper understanding of cultural nuances and patient needs, resulting in more effective study designs and outcomes. For instance, integrating digital health technologies, such as telemedicine and mobile health applications, has been shown to enhance patient engagement and monitoring processes, facilitating more efficient remote testing and expanding access to diverse patient populations. This evolution allows Medtech companies to connect with a wider range of participants, ultimately improving study outcomes.
The collaboration between bioaccess™ and Caribbean Health Group (CHG) exemplifies the potential of strategic partnerships in Colombia. This initiative aims to position Barranquilla as a leading site for medical studies in South America, supported by the Colombian Minister of Health. Furthermore, partnerships with organizations like IDx Technologies and GlobalCare Clinical Trials enhance bioaccess™'s capabilities, achieving significant reductions in recruitment time and high retention rates.
The role of local CROs in this ecosystem is indispensable; they act as vital conduits between Medtech companies and the healthcare landscape, ensuring compliance with local standards and fostering trust among participants. As Sandra Grosworsel Schefler, Managing Director in Brazil, emphasizes, "It benefits to heed these regional specialists for the optimal result of a South American study." By forging these strategic alliances, Medtech firms can effectively explore why Latin America is a compelling choice for Medtech trials, navigating the region's complexities and unlocking its vast research potential.
The Future of Medtech Trials: Opportunities in Latin America
The outlook for Medtech experiments in Latin America is exceptionally bright, prompting the question: why choose this region for medtech trials? The answer lies in a surge in healthcare spending and evolving regulatory frameworks that favor innovation. By 2025, healthcare spending in the area is projected to increase substantially, creating a favorable environment for research investments. This growth is further enhanced by advancements in technology, particularly in telemedicine and decentralized clinical studies, which are set to transform patient engagement and streamline study processes.
As organizations increasingly recognize the potential of alternative care delivery models, the landscape for Medtech studies is undergoing a significant transformation. Notably, sixty-three percent of survey participants anticipate their organizations will invest in alternative care locations, new facilities, or innovative care-delivery methods by 2025. Expert forecasts indicate that companies aligning strategically with these trends will not only reap immediate benefits from conducting studies in the southern region but also lay the groundwork for long-term growth and innovation within the Medtech sector.
The Horizon Databook reveals that over 1 million market statistics and more than 20,000 reports are available, offering invaluable insights into market trends and customer preferences. This wealth of data empowers companies to make informed decisions and gain a competitive edge in the medical research technology landscape.
Moreover, the ongoing updates to regulations throughout Latin America are designed to foster innovation while maintaining safety and quality standards, particularly in digital health and telemedicine. Consequently, the region is becoming an increasingly attractive destination for Medtech studies, raising the question of why choose Latin America for medtech trials. This region presents unique opportunities for companies poised to invest in this burgeoning market. bioaccess® plays a pivotal role in this landscape by providing comprehensive research study management services, including:
- feasibility assessments
- site selection
- compliance evaluations
- study setup
- import permits
- project management
- reporting
Their expertise in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF) ensures that organizations can navigate these opportunities effectively. Additionally, the collaboration between bioaccess® and Caribbean Health Group aims to position Barranquilla as a premier destination for clinical trials in Latin America, with the support of Colombia's Minister of Health, further enhancing the region's appeal for Medtech investments.
Conclusion
Latin America is swiftly emerging as a premier destination for Medtech trials, presenting substantial operational advantages that can significantly enhance research outcomes. With potential cost savings of up to 60% compared to traditional markets, the region offers lower operational costs and access to a diverse patient population, enabling companies to maximize their budgets while achieving comprehensive trial results.
The growth of the Medtech market in Latin America is driven by increased healthcare investments and a rising demand for innovative technologies. Recent regulatory reforms, particularly in Brazil, underscore the region's commitment to streamlining trial approval processes, facilitating quicker and more efficient project initiation.
To fully capitalize on these opportunities, Medtech companies must establish strategic partnerships with local organizations and experts. Collaborating with Contract Research Organizations (CROs) and healthcare institutions enhances patient recruitment and provides essential insights into the regulatory landscape and cultural dynamics. This local expertise is crucial for designing effective trials and improving patient outcomes.
In conclusion, the outlook for Medtech trials in Latin America is promising. With ongoing advancements in technology and evolving regulations, the region is poised for significant growth. By investing in this vibrant market, Medtech firms can position themselves for long-term success, leading to improved healthcare solutions that benefit diverse populations across Latin America.
Frequently Asked Questions
What advantages does Latin America offer for Medtech evaluations?
Latin America provides a vibrant setting for Medtech evaluations, characterized by reduced operational expenses, diverse patient demographics, and a supportive regulatory framework, making it an attractive choice for clinical research.
How much can businesses save by conducting tests in southern regions of Latin America compared to northern regions or Europe?
Businesses can realize substantial cost reductions, with projections indicating that conducting tests in the southern regions can be up to 60% more affordable than in the northern regions or Europe.
Why is patient recruitment cost significant in Medtech trials?
Patient recruitment expenses can range from $15,000 to $50,000 per participant in more developed markets, making cost efficiency critical for conducting clinical trials.
What role does demographic diversity play in Medtech studies in Latin America?
The diverse demographics of Latin America present a rich pool of participants, enhancing the generalizability of study results and bolstering the scientific validity of studies.
What recent legislative changes in Brazil support medical research?
The recent approval of Law 14.874/24 in Brazil aims to streamline the evaluation process for medical studies, reducing administrative hurdles and enhancing predictability, which is expected to boost research activities.
What services does bioaccess® provide for Medtech studies?
Bioaccess® offers comprehensive study management services, including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies (PMCF).
What is the current trend in the South American Medtech market?
The South American Medtech market is experiencing significant growth due to rising demand for innovative medical technologies, driven by the increasing prevalence of chronic diseases and an aging population.
What challenges do Medtech companies face in Latin America?
Medtech companies must navigate a complex landscape of varying healthcare regulations and market access challenges unique to each country, which can include logistics and import regulations.
How does the competitive environment in the South American Medtech sector influence innovation?
The presence of numerous participants and substantial market shares held by prominent firms fosters innovation and encourages companies to develop effective strategies to overcome logistical and regulatory hurdles.
What is the partnership between bioaccess® and Caribbean Health Group aimed at achieving?
The partnership seeks to establish Barranquilla as a prominent location for medical studies in the region, improving the area’s appeal for first-in-human research and other health-related initiatives.




